Tretinoin (topical)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Deepika Beereddy, MBBS [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING
See full prescribing information for complete Boxed Warning.
Experienced Physician and Institution :
Retinoic Acid-APL Syndrome:
Leukocytosis at Presentation and Rapidly Evolving Leukocytosis During Tretinoin Capsules Treatment:
Teratogenic Effects. Pregnancy Category D:
|
Overview
Tretinoin (topical) is a dermatologic agent, keratolytic agent that is FDA approved for the treatment of acne vulgaris; induction therapy of acute promyelocytic leukemia, FAB M3, in patients who are refractory to, have relapsed from, or are not candidates for anthracycline chemotherapy. There is a Black Box Warning for this drug as shown here. Common adverse reactions include cardiac dysrhythmia, chest discomfort, edema, flushing, peripheral edema, alopecia , dry skin, erythema, injection site reaction, peeling of skin, pruritus, rash, sensation of burning of skin, skin irritation , stinging of skin, hypercholesterolemia, hypertriglyceridemia, decreased , weight gain, abdominal pain, constipation, diarrhea, loss of appetite, mucous membrane dryness, nausea and vomiting, increased liver function test, bone pain, dizziness, headache, paresthesia, visual disturbance, otalgia, agitation, anxiety, hallucinations, disorder of upper respiratory system, dyspnea, respiratory insufficiency, fever,infectious disease, malaise, pain, and shivering.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Tretinoin cream:
Acne vulgaris
- Tretinoin gel and cream are indicated for topical application in the treatment of acne vulgaris. The safety and efficacy of the long - term use of this product in the treatment of other disorders have not been established.
- Dosing Information
- Tretinoin gel or cream should be applied once a day, before retiring, to the skin where acne lesions appear, using enough to cover the entire affected area lightly. Gel: Excessive application results in " pilling " of the gel, which minimizes the likelihood of over application by the patient.
- Application may cause a transitory feeling of warmth or slight stinging. In cases where it has been necessary to temporarily discontinue therapy or to reduce the frequency of application, therapy may be resumed or frequency of application increased when the patients become able to tolerate the treatment.
- Alterations of vehicle, drug concentration, or dose frequency should be closely monitored by careful observation of the clinical therapeutic response and skin tolerance.
- During the weeks of therapy, an apparent exacerbation of inflammatory lesions may occur. This is due to the action of the medication on deep, previously unseen lesions and should not be considered a reason to discontinue therapy.
- Therapeutic results should be noticed after two to three weeks but more than six weeks of therapy may be required before beneficial effects are seen.
- Once the acne lesions have responded satisfactorily, it may be possible to maintain the improvement with less frequent applications, or other dosage forms.
- Patients treated with tretinoin preparations may use cosmetics, but the areas to be treated should be cleansed thoroughly before the medication is applied.
Hyperpigmentation of skin, Facial mottling; with comprehensive skin care and sunlight avoidance programs; Adjunct(Renova(R) 0.05% cream only, 50 years or younger only)
- Dosing Information
- For palliation of mottled hyperpigmentation, Renova(R) 0.05% cream should be used in conjunction with a comprehensive skin care and sunlight avoidance program. A pea sized amount should be applied topically once daily at bedtime covering the effected area lightly and avoiding the eyes, ears, nostrils and angles of mouth and nose. Excessive amounts will not increase efficacy and may result in marked redness, peeling or discomfort.
Roughness of skin, Facial tactile roughness; with comprehensive skin care and sunlight avoidance programs; Adjunct(Renova(R) 0.05% cream only, 50 years or younger only)
- Dosing Information
- For palliation of tactile roughness of facial skin, Renova(R) 0.05% cream should be used in conjunction with a comprehensive skin care and sunlight avoidance program. A pea sized amount should be applied topically once daily at bedtime covering the effected area lightly and avoiding the eyes, ears, nostrils and angles of mouth and nose. Excessive amounts will not increase efficacy and may result in marked redness, peeling or discomfort.
Tretinoin capsule:
Acute promyelocytic leukemia, FAB M3, Induction therapy in patients who are refractory to, have relapsed from, or are not candidates for anthracycline chemotherapy
- Tretinoin capsules are indicated for the induction of remission in patients with acute promyelocytic leukemia (APL), French-American-British (FAB) classification M3 (including the M3 variant), characterized by the presence of the t(15;17) translocation and/or the presence of the PML/RARα gene who are refractory to, or who have relapsed from, anthracycline chemotherapy, or for whom anthracycline-based chemotherapy is contraindicated. Tretinoin capsules are for the induction of remission only. The optimal consolidation or maintenance regimens have not been defined, but all patients should receive an accepted form of remission consolidation and/or maintenance therapy for APL after completion of induction therapy with tretinoin capsules.
- Dosing Information
- The recommended dose is 45 mg/m2/day administered as two evenly divided doses until complete remission is documented. Therapy should be discontinued 30 days after achievement of complete remission or after 90 days of treatment, whichever occurs first.
- If after initiation of treatment of Tretinoin Capsules the presence of the t(15;17) translocation is not confirmed by cytogenetics and/or by polymerase chain reaction studies and the patient has not responded to tretinoin capsules, alternative therapy appropriate for acute myelogenous leukemia should be considered.
- Tretinoin Capsules are for the induction of remission only. Optimal consolidation or maintenance regimens have not been determined. All patients should, therefore, receive a standard consolidation and/or maintenance chemotherapy regimen for APL after induction therapy with tretinoin capsules, unless otherwise contraindicated.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- Peripheral neuropathy, Chemotherapy-induced; Prophylaxis
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Tretinoin (topical) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Acne vulgaris
- Dosing Information
- For children 12 years or older, apply gel microsphere (0.1%, 0.04%) topically to acne lesions once daily at bedtime to the area of the skin where acne lesions appear. Enough product should be applied to cover the entire area lightly. Excessive amounts will not increase efficacy and may result in marked redness, peeling or discomfort. If irritation or stinging is excessive, frequency of application may be reduced. When the patient becomes tolerant to treatment, the frequency of application may be increased. Efficacy has not been established for dosing frequencies less then once daily. During the first few weeks of therapy, an exacerbation of the lesions may be seen. This is an expected reaction and therapeutic results should be seen after 2 weeks, but more than 7 weeks of therapy may be necessary to see a beneficial response.
Acute promyelocytic leukemia, FAB M3, Induction therapy in patients who are refractory to, have relapsed from, or are not candidates for anthracycline chemotherapy(1 year or older; oral only)
- Dosing Information
- For patients 1 year or older, the recommended tretinoin dosage regimen for the induction of remission in patients with acute promyelocytic leukemia (PML) who are refractory to, or who have relapsed from, anthracycline chemotherapy, or for whom anthracycline-based chemotherapy is contraindicated is 45 mg/m(2)/day orally administered in 2 evenly divided doses. Treatment should be discontinued 30 days after achievement of complete remission or after 90 days of treatment, whichever occurs first. Because optimal tretinoin consolidation or maintenance regimens have not been determined, the manufacturer recommends that all patients receive standard consolidation and/or maintenance following tretinoin induction therapy.
- The safety and efficacy of oral tretinoin have not been established in pediatric patients younger than 1 year.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Tretinoin (topical) in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Tretinoin (topical) in pediatric patients.
Contraindications
- Use of this product should be discontinued if hypersensitivity to any of the ingredients is noted.
Warnings
|
WARNING
See full prescribing information for complete Boxed Warning.
Experienced Physician and Institution :
Retinoic Acid-APL Syndrome:
Leukocytosis at Presentation and Rapidly Evolving Leukocytosis During Tretinoin Capsules Treatment:
Teratogenic Effects. Pregnancy Category D:
|
Tretinoin cream:
- GELS ARE FLAMMABLE. AVOID FIRE, FLAME OR SMOKING DURING USE. Keep out of reach of children.
- Keep tube tightly closed. Do not expose to heat or store at temperatures above 120 F ( 49 C ).
PRECAUTIONS
General
- If a reaction suggesting sensitivity or chemical irritation occurs, use of the medication should be discontinued. Exposure to sunlight, including sunlamos, should be minimized during the use of tretinoin, and patients with sunburns should be advised not to use this product until fully recored because of heightened susceptibility to sunlight as a result of the use of tretinoin. Patients who may be required to have considerable sun exposure due to occupation and those with inherent sensitivity to the sun should exercise particular caution. Use of sunscreen products and protective clothing over treated areas is recommended when exposure cannot be avoided. Weather extremes, such as wind or cold,also may be irritating to patients under treatment with tretinoin.
- Tretinoin preparations for acne treatment should be kept away from the eyes, the mouth, angles of the nose, and mucous membranes. Topical use may induce severe local erythema and peeling at the site of application. If the degree of local irritation warrants, patients should be directed to use the medication less frequently, discontinue use temporarily, or discontinue use altogether. Tretinoin has been reported to cause severe irritation on eczematous skin and should be used with utmost caution in patients with this condition.
Tretinoin capsule:
Pregnancy Category D – See Boxed WARNINGS
- Tretinoin has teratogenic and embryotoxic effects in mice, rats, hamsters, rabbits and pigtail monkeys, and may be expected to cause fetal harm when administered to a pregnant woman. Tretinoin causes fetal resorptions and a decrease in live fetuses in all animals studied. Gross external, soft tissue and skeletal alterations occurred at doses higher than 0.7 mg/kg/day in mice, 2 mg/kg/day in rats, 7 mg/kg/day in hamsters, and at a dose of 10 mg/kg/day, the only dose tested, in pigtail monkeys (about 1/20, 1/4, and 1/2 and 4 times the human dose, respectively, on a mg/m2 basis).
- There are no adequate and well-controlled studies in pregnant women. Although experience with humans administered tretinoin capsules is extremely limited, increased spontaneous abortions and major human fetal abnormalities related to the use of other retinoids have been documented in humans. Reported defects include abnormalities of the CNS, musculoskeletal system, external ear, eye, thymus and great vessels; and facial dysmorphia, cleft palate, and parathyroid hormone deficiency. Some of these abnormalities were fatal. Cases of IQ scores less than 85, with or without obvious CNS abnormalities, have also been reported. All fetuses exposed during pregnancy can be affected and at the present time there is no antepartum means of determining which fetuses are and are not affected.
- Effective contraception must be used by all females during tretinoin capsules therapy and for 1 month following discontinuation of therapy. Contraception must be used even when there is a history of infertility or menopause, unless a hysterectomy has been performed. Whenever contraception is required, it is recommended that two reliable forms of contraception be used simultaneously, unless abstinence is the chosen method. If pregnancy does occur during treatment, the physician and patient should discuss the desirability of continuing or terminating the pregnancy.
Patients Without the t(15;17) Translocation
- Initiation of therapy with tretinoin capsules may be based on the morphological diagnosis of acute promyelocytic leukemia. Confirmation of the diagnosis of APL should be sought by detection of the t(15;17) genetic marker by cytogenetic studies. If these are negative, PML/RARα fusion should be sought using molecular diagnostic techniques. The response rate of other AML subtypes to tretinoin capsules has not been demonstrated; therefore, patients who lack the genetic marker should be considered for alternative treatment.
Retinoic Acid-APL (RA-APL) Syndrome
- In up to 25% of patients with APL treated with tretinoin capsules, a syndrome occurs which can be fatal.
Leukocytosis at Presentation and Rapidly Evolving Leukocytosis During Tretinoin Capsules Treatment
- See boxed WARNINGS.
Pseudotumor Cerebri
- Retinoids, including tretinoin capsules, have been associated with pseudotumor cerebri (benign intracranial hypertension), especially in pediatric patients. The concomitant use of other agents known to cause pseudotumor cerebri/intracranial hypertension, such as tetracyclines, might increase the risk of this condition. Early signs and symptoms of pseudotumor cerebri include papilledema, headache, nausea and vomiting, and visual disturbances. Patients with these symptoms should be evaluated for pseudo - tumor cerebri, and, if present, appropriate care should be instituted in concert with neurological assessment.
Lipids
- Up to 60% of patients experienced hypercholesterolemia and/or hypertriglyceridemia, which were reversible upon completion of treatment. The clinical consequences of temporary elevation of triglycerides and cholesterol are unknown, but venous thrombosis and myocardial infarction have been reported in patients who ordinarily are at low risk for such complications.
Elevated Liver Function Test Results
- Elevated liver function test results occur in 50% to 60% of patients during treatment. Liver function test results should be carefully monitored during treatment and consideration be given to a temporary withdrawal of tretinoin capsules if test results reach >5 times the upper limit of normal values. However, the majority of these abnormalities resolve without interruption of tretinoin capsules or after completion of treatment.
PRECAUTIONS
General
- Tretinoin capsules have potentially significant toxic side effects in APL patients. Patients undergoing therapy should be closely observed for signs of respiratory compromise and/or leukocytosis. Supportive care appropriate for APL patients, eg, prophylaxis for bleeding, prompt therapy for infection, should be maintained during therapy with tretinoin capsules.
- There is a risk of thrombosis (both venous and arterial) which may involve any organ system, during the first month of treatment. Therefore, caution should be exercised when treating patients with the combination of tretinoin capsules and anti-fibrinolytic agents, such as tranexamic acid, aminocaproic acid or aprotinin.
- The ability to drive or operate machinery might be impaired in patients treated with tretinoin capsules, particularly if they are experiencing dizziness or severe headache.
- Microdosed progesterone preparations (“minipill”) may be an inadequate method of contraception during treatment with tretinoin capsules.
Laboratory Tests
- The patient’s hematologic profile, coagulation profile, liver function test results, and triglyceride and cholesterol levels should be monitored frequently.
Adverse Reactions
Clinical Trials Experience
Tretinoin cream:
- The skin if certain sensitive individuals may become excessively red, edamatous, blistered or crusted.
- If these effects occur, the medication should either be discontinued until the integrity of the skin is restored, or the medication should be adjusted to a level the patient can tolerate. True contact allergy to topical tretinoin is rarely encountered. Temporary hyper - or hypopigmentation has been reported with susceptibility to sunlight while under treatment with tretinoin. To date, all adverse effects of tretinoin have been reversible upon discontinuance of therapy.
Tretinoin capsule:
Warnings
- Virtually all patients experience some drug-related toxicity, especially head ache, fever, weakness, and fatigue. These adverse effects are seldom permanent or irreversible nor do they usually require interruption of therapy. Some of the adverse events are common in patients with APL, including hemorrhage, infections, gastrointestinal hemorrhage, disseminated intravascular coagulation, pneumonia, septicemia, and cerebral hemorrhage. The following describes the adverse events, regardless of drug relationship, that were observed in patients treated with tretinoin capsules.
Typical Retinoid Toxicity
- The most frequently reported adverse events were similar to those described in patients taking high doses of vitamin A and included headache (86%), fever (83%), skin/mucous membrane dryness (77%), bone pain (77%), nausea/vomiting (57%), rash (54%), mucositis (26%), pruritus (20%), increased sweating (20%), visual disturbances (17%), ocular disorders (17%), alopecia (14%), skin changes (14%), changed visual acuity (6%), bone inflammation (3%), visual field defects (3%).
RA-APL Syndrome
- APL patients treated with tretinoin capsules have experienced a potentially fatal syndrome characterized by fever, dyspnea, acute respiratory distress, weight gain, radiographic pulmonary infiltrates, pleural and pericardial effusions, edema, and hepatic, renal, and multi-organ failure. This syndrome has occasionally been accompanied by impaired myocardial contractility and episodic hypotension and has been observed with or without concomitant leukocytosis. Some patients have expired due to progressive hypoxemia and multi-organ failure. The syndrome generally occurs during the first month of treatment, with some cases reported following the first dose of tretinoin capsules. The management of the syndrome has not been defined rigorously, but high-dose steroids given at the first signs of the syndrome appear to reduce morbidity and mortality. Treatment with dexamethasone, 10 mg intravenously administered every 12 hours for 3 days or until resolution of symptoms, should be initiated without delay at the first suspicion of symptoms (one or more of the following: fever, dyspnea, weight gain, abnormal chest auscultatory findings or radiographic abnormalities). Sixty percent or more of patients treated with tretinoin capsules may require high-dose steroids because of these symptoms. The majority of patients do not require termination of tretinoin capsules therapy during treatment of the syndrome.
Body as a Whole
- General disorders related to tretinoin capsules administration and/or associated with APL included malaise (66%), shivering (63%), hemorrhage (60%), infections (58%), peripheral edema (52%), pain (37%), chest discomfort (32%), edema (29%), disseminated intravascular coagulation (26%), weight increase (23%), injection site reactions (17%), anorexia (17%), weight decrease (17%), myalgia (14%), flank pain (9%), cellulitis (8%), face edema (6%), fluid imbalance (6%), pallor (6%), lymph disorders (6%), acidosis (3%), hypothermia (3%), ascites (3%).
Respiratory System Disorders
- Respiratory system disorders were commonly reported in APL patients administered tretinoin capsules. The majority of these events are symptoms of the RA-APL syndrome. Respiratory system adverse events included upper respiratory tract disorders (63%), dyspnea (60%), respiratory insufficiency (26%), pleural effusion (20%), pneumonia (14%), rales (14%), expiratory wheezing (14%), lower respiratory tract disorders (9%), pulmonary infiltration (6%), bronchial asthma (3%), pulmonary edema (3%), larynx edema (3%), unspecified pulmonary disease (3%).
Ear Disorders
- Ear disorders were consistently reported, with earache or feeling of fullness in the ears reported by 23% of the patients. Hearing loss and other unspecified auricular disorders were observed in 6% of patients, with infrequent (<1%) reports of irreversible hearing loss.
Gastrointestinal Disorders
- GI disorders included GI hemorrhage (34%), abdominal pain (31%), other gastrointestinal disorders (26%), diarrhea (23%), constipation (17%), dyspepsia (14%), abdominal distention (11%), hepatosplenomegaly (9%), hepatitis (3%), ulcer (3%), unspecified liver disorder (3%).
Cardiovascular and Heart Rate and Rhythm Disorders
- Arrhythmias (23%), flushing (23%), hypotension (14%), hypertension (11%), phlebitis (11%), cardiac failure (6%) and for 3% of patients: cardiac arrest, myocardial infarction, enlarged heart, heart murmur, ischemia, stroke, myocarditis, pericarditis, pulmonary hypertension, secondary cardiomyopathy.
Central and Peripheral Nervous System Disorders and Psychiatric
- Dizziness (20%), paresthesias (17%), anxiety (17%), insomnia (14%), depression (14%), confusion (11%), cerebral hemorrhage (9%), intracranial hypertension (9%), agitation (9%), hallucination (6%) and for 3% of patients: abnormal gait, agnosia, aphasia, asterixis, cerebellar edema, cerebellar disorders, convulsions, coma, CNS depression, dysarthria, encephalopathy, facial paralysis, hemiplegia, hyporeflexia, hypotaxia, no light reflex, neurologic reaction, spinal cord disorder, tremor, leg weakness, unconsciousness, dementia, forgetfulness, somnolence, low speech.
Urinary System Disorders
- Renal insufficiency (11%), dysuria (9%), acute renal failure (3%), micturition frequency (3%), renal tubular necrosis (3%), enlarged prostate (3%).
Miscellaneous Adverse Events
- Isolated cases of erythema nodosum, basophilia and hyperhistaminemia, Sweet’s syndrome, organomegaly, hypercalcemia, pancreatitis and myositis have been reported.
Additional Adverse Reactions Reported With Tretinoin Capsules
Cardiovascular
- Cases of thrombosis (both venous and arterial) involving various sites (eg, cerebrovascular accident, myocardial infarction, renal infarct) have been reported rarely.
Hematologic
- Rare cases of thrombocytosis have been reported.
Skin
Miscellaneous Adverse Events
- Rare cases of vasculitis, predominantly involving the skin, have been reported.
Postmarketing Experience
There is limited information regarding Tretinoin (topical) Postmarketing Experience in the drug label.
Drug Interactions
Tretinoin cream
Drug Interactions
- In a 91 - week dermal study in which CD - 1 mice were administered 0.017 % and 0.035 % formulations of tretinoin, cutaneous squamous cell carcinomas and papillomas in the treatment area were observed in some female mice. A dose - related incidence of liver tumors in male mice was observed at those same doses. The maximum systemic doses associated with the administered 0.017 % and 0.035 % formulations are 0.5 and 1.0 mg /kg / day, respectively. These doses are two and four times the maximum human systemic dose, when adjusted for total body area. The biological significance of these findings is not clear because they occurred at doses that exceeded the dermal maximally tolerated dose ( MTD ) of tretinoin and because they were within the background natural occurrence rate for these tumors in this strain of mice. There was no evidence of carcinogenic potential when 0.025 mg / kg / day of tretinoin was administered topically to mice ( 0.1 times the maximum human systemic dose, adjusted for total body surface area ). For purposes of comparisons of the animal exposure to systemic human exposure, the maximum human systemic dose is defined as 1 gram of 0.1 % tretinoin applied daily to a 50 kg person ( 0.02 mg tretinoin / kg body weight).
- Studies in hairless albino mice suggest that concurrent exposure to tretinoin may enhance the tumorigenic potential of carcinogenic doses of UVB and UVA light form a solar stimulator. This effect has been confirmed in a later study in pigmented mice, and dark pigmentation did not overcome the enhancement of photocarcinogenesis by 0.05 % tretinoin. Although the significance of these studies ton humans is not clear, patients should minimize exposure to sunlight or artificial ultraviolet radiation sources.
- The mutagenic potential of tretinoin was evaluated in the Ames assay and in the invivomouse micronucleus assay, both of which were negative.
- In dermal Segment I fertility studies of tretinoin in rats, slight ( not statistically significant ) decreases in sperm count and motility were seen at 0.5 mg / kg / day ( 4 times the maximum human systemic dose adjusted for total body surface area ), and slight ( not statistically significant) increases in the number and percent of nonviable embryos in females treated with 0.25 mg / kg /day ( 2 times the maximum human systemic dose adjusted for total body surface area ) and above were observed. A dermal Segment III study with tretinoin has not been performed has not been performed in any species. In oral Segment I and Segment III studies in rats with tretinoin, decreased survival of neonates and growth retardation were observed at doses in excess of 2 mg / kg / day (16 times the human topical dose adjusted for total body surface area).
- Limited clinical data on potential drug interactions are available.
Drugs Metabolized By the Hepatic P450 System
- As tretinoin capsules are metabolized by the hepatic P450 enzymes, there is a potential for alteration of pharmacokinetics parameters in patients administered concomitant medications that are also inducers or inhibitors of this system. Medications that generally induce hepatic P450 enzymes include rifampicin, glucocorticoids, phenobarbital and pentobarbital. Medications that generally inhibit hepatic P450 enzymes include ketoconazole, cimetidine, erythromycin, verapamil, diltiazem and cyclosporine. To date there are no data to suggest that co-use with these medications increases or decreases either efficacy or toxicity of tretinoin capsules.
- Agents Known to Cause Pseudotumor Cerebri/Intracranial Hypertension (Such as Tetracyclines)
- Tretinoin capsules may cause pseudotumor cerebri/intracranial hypertension. Concomitant administration of tretinoin capsules and agents known to cause pseudotumor cerebri/intracranial hypertension as well might increase the risk of this condition.
Vitamin A
- As with other retinoids, tretinoin capsules must not be administered in combination with vitamin A because symptoms of hypervitaminosis A could be aggravated.
Anti-fibrinolytic Agents (Such as Tranexamic Acid, Aminocaproic Acid, or Aprotinin)
- Cases of fatal thrombotic complications have been reported rarely in patients concomitantly treated with tretinoin capsules and anti-fibrinolytic agents. Therefore, caution should be exercised when administering tretinoin capsules concomitantly with these agents.
Effect of Food
- No data on the effect of food on the absorption of tretinoin capsules are available. The absorption of retinoids as a class has been shown to be enhanced when taken together with food.
Tretinoin capsule
- Limited clinical data on potential drug interactions are available.
Drugs Metabolized By the Hepatic P450 System
- As tretinoin capsules are metabolized by the hepatic P450 enzymes, there is a potential for alteration of pharmacokinetics parameters in patients administered concomitant medications that are also inducers or inhibitors of this system. Medications that generally induce hepatic P450 enzymes include rifampicin, glucocorticoids, phenobarbital and pentobarbital. Medications that generally inhibit hepatic P450 enzymes include ketoconazole, cimetidine, erythromycin, verapamil, diltiazem and cyclosporine. To date there are no data to suggest that co-use with these medications increases or decreases either efficacy or toxicity of tretinoin capsules.
Agents Known to Cause Pseudotumor Cerebri/Intracranial Hypertension (Such as Tetracyclines)
- Tretinoin capsules may cause pseudotumor cerebri/intracranial hypertension. Concomitant administration of tretinoin capsules and agents known to cause pseudotumor cerebri/intracranial hypertension as well might increase the risk of this condition.
Vitamin A
- As with other retinoids, tretinoin capsules must not be administered in combination with vitamin A because symptoms of hypervitaminosis A could be aggravated.
Anti-fibrinolytic Agents (Such as Tranexamic Acid, Aminocaproic Acid, or Aprotinin)
- Cases of fatal thrombotic complications have been reported rarely in patients concomitantly treated with tretinoin capsules and anti-fibrinolytic agents. Therefore, caution should be exercised when administering tretinoin capsules concomitantly with these agents.
Effect of Food
- No data on the effect of food on the absorption of tretinoin capsules are available. The absorption of retinoids as a class has been shown to be enhanced when taken together with food.
Use in Specific Populations
Pregnancy
- Oral tretinoin ha sbeen shown to be teratogenic in rats, mice, hamsters, and subhuman primates. It was teratogenic and fetotoxic in Wistar rats when given orally or topically in doses greater than 1 mg / kg /day (8 times the maximum human systemic dose adjusted for total body surface area). However, variations in teratogenic doses among various strains of rats have been reported. In the cynomolgus monkey, which metabolically is closer to humans for tretinoin than the other species examined, fetal malfomrations were reported at doses of 10 mg / kr / day(83 times the maximumhuman systemic dose adjusted for total body surface area), although skeletal variations were observed at all doses. A dose - related increase in embryolethality and abortion were reported. Similar reuslts have been reported in pigtail macaques.
- Topical tretinoin in animal teratogenecity tests have generated equivocal results. There is evidence for teratogenicity (shortened or kinked tail) of topical tretinoin in Wistar rats at doses greater than 1 mg / kg /day (8 times e maximum human systemic dose adjusted for total body surface area). Anomalies(humerous: short 13 % , bent 6 %, os parietal incompletely ossified 14 % ) have also been reported when 10 mg / kg / day was topically applied.
- There are other reports in New Zealand White rabbits administered in doses of greater than 0.2 mg / kg / day ( 3.3 times the maximum human systemic dose adjusted for total body surface area ) of an increased incidence of domed head and hydrocephaly, typical of reinoid - induced fetal malformations in this species.
- In contrast, several well - controlled animal studies have shown that dermally applied tretinoin may be fetotoxic, but not overly teratogenic in rats and rabbits at doses of 1.0 and 0.5 mg / kg / day, respectively (8 times the maximum human systemic dose adjusted for total body surface area in both species).
- With widespread use of any drug,a small number of birth defect reprots associated temporally with the administration of the drug would be expected by chance alone. Thirty human cases of temporally associated congenital malformations have been reported during two decades of clinical use of tretinoin.
- Although no definite pattern of teratogenecity and no causal association has been established form these cases, five of the reports describe therare birth defect category holoprosencephaly (defects associated with incomplete midline development of the forebrain). The significance of these spontaneous reports in terms of risk to the fetus is not known.
Nonteratogenic Effects
- Topical tretinoin has been shown to be ferotoxic in rabbits when administered 0.5 mg / kg /day (8 times the maximum human systemic dose adjusted for total body surface area). Oral tretinoin has been shown to be ferotoxic, resulting in skeletal variations and increased intrauterine death in rats when administered 2.5 mg / kg / day ( 20 mes the maximum human systemic dose adjusted for total body surface area).
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
- There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Tretinoin (topical) in women who are pregnant.
Labor and Delivery
- There is no FDA guidance on use of Tretinoin (topical) during labor and delivery.
Nursing Mothers
- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when tretinoin is used by a nursing woman.
Pediatric Use
- Safety and effectiveness in pediatric patients below the age of 12 have not been established.
Geriatic Use
- Safety and effectiveness in a geriatric populationhave not been established. Clinical studies of tretinoin did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger patients.
Gender
- There is no FDA guidance on the use of Tretinoin (topical) with respect to specific gender populations.
Race
- There is no FDA guidance on the use of Tretinoin (topical) with respect to specific racial populations.
Renal Impairment
- There is no FDA guidance on the use of Tretinoin (topical) in patients with renal impairment.
Hepatic Impairment
- There is no FDA guidance on the use of Tretinoin (topical) in patients with hepatic impairment.
Females of Reproductive Potential and Males
- There is no FDA guidance on the use of Tretinoin (topical) in women of reproductive potentials and males.
Immunocompromised Patients
- There is no FDA guidance one the use of Tretinoin (topical) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Tretinoin gel or cream should be applied once a day, before retiring, to the skin where acne lesions appear, using enough to cover the entire affected area lightly. Gel: Excessive application results in " pilling " of the gel, which minimizes the likelihood of over application by the patient.
- Application may cause a transitory feeling of warmth or slight stinging. In cases where it has been necessary to temporarily discontinue therapy or to reduce the frequency of application, therapy may be resumed or frequency of application increased when the patients become able to tolerate the treatment.
- Alterations of vehicle, drug concentration, or dose frequency should be closely monitored by careful observation of the clinical therapeutic response and skin tolerance.
- During the weeks of erapy, an apparent exacerbation of inflammatory lesions may occur. This is due to the action of the medication on deep, previously unseen lesions and should not be considered a reason to discontinue therapy.
- Therapeutic results should be noticed after two to three weeks but more than six weeks of therapy may be required before beneficial effects are seen.
- Once the acne lesions have responded satisfactorily, it may be possible to maintain the improvement with less frequent applications, or other dosage forms.
- Patients treated with tretinoin preparations may use cosmetics, but the areas to be treated should be cleansed thoroughly before the medication is applied
Monitoring
- There is limited information regarding Monitoring of Tretinoin (topical) in the drug label.
IV Compatibility
- There is limited information regarding IV Compatibility of Tretinoin (topical) in the drug label.
Overdosage
- If medication is applied excessively, no more rapid or better results will be obtained and marked redness, peeling, or discomfort may occur. Oral ingestion of the drug may lead to the same side effects as those associated with excessive oral intake of Vitamin A.
Pharmacology
| |
Tretinoin (topical)
| |
| Systematic (IUPAC) name | |
| retinoic acid | |
| Identifiers | |
| CAS number | ? |
| ATC code | D10 L01XX14 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 300.4412 g/mol |
| SMILES | & |
| Physical data | |
| Melt. point | 180 °C (356 °F) |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Protein binding | > 95% |
| Metabolism | ? |
| Half life | 0.5-2 hours |
| Excretion | ? |
| Therapeutic considerations | |
| Licence data |
|
| Pregnancy cat. | |
| Legal status |
Prescription Only (S4)(AU) POM(UK) [[Prescription drug|Template:Unicode-only]](US) |
| Routes | Topical, oral |
Mechanism of Action
- Although the exact mode of action of tretinoin is unknown, current evidence suggest that topical tretinoin decreases cohesiveness of follicular eipthelial cells with decreased micromedo formation.
- Additionally, tretinoin stimulates mitotic activity and increased turnover of follicular epithelial cells causing extrusion of the comedones.
Structure
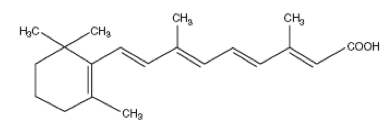
- Tretinoin Gel, USP and Tretinoin Cream, USP are used for the topical treatment of acne vulgaris. Each gram of tretinoin gel contains tretinoin in either of two strengths, 0.025 % (0.25 mg) or 0.01 % (0.1mg) in a gel vehicle of hydroxypropyl cellulose, butylated hydroxytoluene, and alcohol (denatured with tert - butyl alcohol and brucine sulfate) 90 % w / w. Each gram of tretinoin cream contains tretinoin in either of three strengths, 0.1 % (1 mg), 0.05 % (0.5 mg), or 0.025 % (0.25 mg) in a hydrophilic cream vehicle of: stearic acid, isopropyl myristate, polyoxyl 40 stearate, stearyl alcohol, xantham gum, sorbic acid, butylated hydroxytoluene, and purified water. Chemically, tretinoin is all - trans - retinoic acid. It has a molecular weight of 300.44.

Pharmacodynamics
- There is limited information regarding Pharmacodynamics of Tretinoin (topical) in the drug label.
Pharmacokinetics
- There is limited information regarding Pharmacokinetics of Tretinoin (topical) in the drug label.
Nonclinical Toxicology
- There is limited information regarding Nonclinical Toxicology of Tretinoin (topical) in the drug label.
Clinical Studies
- There is limited information regarding Clinical Studies of Tretinoin (topical) in the drug label.
How Supplied
- Tretinoin Cream, USP
Storage
- Store below 27 C ( 80 F )
Images
Drug Images
{{#ask: Page Name::Tretinoin (topical) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel

{{#ask: Label Page::Tretinoin (topical) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information

Precautions with Alcohol
- Alcohol-Tretinoin (topical) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Avita Renova Retin-A, Retin-A Micro, Vesanoid, Tretin-X, Atralin, Refissa, OBAGI.[1]
Look-Alike Drug Names
There is limited information regarding Tretinoin (topical) Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Tretinoin (topical)
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Tretinoin (topical) |Label Name=Tretinoin Package2.png
}}