Morphine (rectal)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Adeel Jamil, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Morphine (rectal) is a analgesic opioid that is FDA approved for the treatment of the relief of severe chronic pain, and severe acute pain. Common adverse reactions include lightheadedness, dizziness, sedation,somnolence, nausea, vomiting, constipation, and sweating.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Morphine is indicated for the relief of severe chronic pain, and severe acute pain.
Dosing Information
Usual Adult Dose:
- 10 to 20 mg every 4 hours or as directed by physician.
- Dosage is a patient dependent variable, therefore increased dosage may be required to achieve adequate analgesia.
- Note: Medication may suppress respiration in the elderly, the very ill, and those patients with respiratory problems, therefore lower doses may be required.
Morphine Dosage Reduction
- During the first two to three days of effective pain relief, the patient may sleep for many hours. This can be misinterpreted as the effect of excessive analgesic dosing rather than the first sign of relief of a pain exhausted patient. The dose, therefore, should be maintained for at least three days before reduction, if respiratory activity and other vital signs are adequate.
- Following successful relief of severe pain, periodic attempts to reduce the narcotic dose should be made. Smaller doses or complete discontinuation of the narcotic analgesic may become feasible due to a physiologic change or the improved mental state of the patient.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Morphine (rectal) in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Morphine (rectal) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Morphine (rectal) FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Morphine (rectal) in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Morphine (rectal) in pediatric patients.
Contraindications
- Hypersensitivity to morphine; respiratory insufficiency or depression; severe CNS depression; attack of bronchial asthma; heart failure secondary to chronic lung disease; cardiac arrhythmias; increased intracranial or cerebrospinal pressure; head injuries; brain tumor; acute alcoholism, delerium tremens; convulsive disorders; after biliary tract surgery; suspected surgical abdomen; surgical anastomosis; concomitantly with MAO inhibitors or within 14 days of such treatment.
Warnings
- Morphine can cause tolerance, psychological and physical dependence. Withdrawal will occur on abrupt discontinuation or administration of a narcotic antagonist.
PRECAUTIONS
General:
Head Injury and Increased Intracranial Pressure=
- The respiratory depressant effects of morphine and its capacity to elevate cerebrospinal-fluid pressure may be markedly exaggerated in the presence of increased intracranial pressure. Furthermore, narcotics produce side effects that may obscure the clinical course of patients with head injuries. In such patients, morphine must be used with caution and only if it is deemed essential.
Asthma and Other Respiratory Conditions
- Morphine should be used with caution in patients having an acute asthmatic attack, in those with chronic obstructive pulmonary disease or cor pulmonale, and in individuals with substantially decreased respiratory reserve, preexisting respiratory depression, hypoxia, or hypercapnia. In such patients, even usual therapeutic doses of narcotics may decrease respiratory drive while simultaneously increasing airway resistance to the point of apnea.
Hypotensive Effect
- The administration of morphine may result in severe hypotension in an individual whose ability to maintain his blood pressure has already been compromised by a depleted blood volume or concurrent administration of such drugs as the phenothiazines or certain anesthetics.
Supraventricular Tachycardias
- Caution should be used in patients with atrial flutter and other supraventricular tachycardias due to a possible vagolytic action which may produce a significant increase in the ventricular response rate.
Special Risk Patients
- Morphine should be given with caution and the initial dose should be reduced in certain patients, such as the elderly or debilitated and those with severe impairment of hepatic or renal function, hypothyroidism, Addison's disease, prostatic hypertrophy, or urethral stricture.
Convulsions
- Morphine may aggravate preexisting convulsive disorders. Convulsions may occur in individuals without a history of convulsive disorders if dosage is substantially escalated above recommended levels because of tolerance development.
Acute Abdominal Conditions
- The administration of morphine or other narcotics may obscure the diagnosis or clinical course in patients with acute abdominal conditions.
DRUG ABUSE AND DEPENDENCE
- Morphine Sulfate is a Schedule II controlled substance. As with other narcotics, some patients may develop a physical and psychological dependence on morphine. They may increase dosage without consulting a physician and subsequently may develop a physical dependence on the drug. In such cases, abrupt discontinuance may precipitate typical withdrawal symptoms, including convulsions. Therefore, the drug should be withdrawn gradually from any patient known to be taking excessive dosages over a long period of time.
- In treating the terminally ill patient the benefit of pain relief may outweigh the possibility of drug dependence. The chance of drug dependence is substantially reduced when the patient is placed on scheduled narcotic programs instead of a "pain to relief of pain" cycle typical of a PRN regimen.
Adverse Reactions
Clinical Trials Experience
- THE MAJOR HAZARDS OF MORPHINE, AS WITH OTHER NARCOTIC ANALGESICS, ARE RESPIRATORY DEPRESSION AND, TO A LESSER DEGREE, CIRCULATORY DEPRESSION, RESPIRATORY ARREST, SHOCK, AND CARDIAC ARREST HAVE OCCURRED.
- The most frequently observed adverse reactions include lightheadedness, dizziness, sedation, nausea, vomiting and sweating. These effects seem to be more prominent in ambulatory patients and in those who are not suffering severe pain. In such individuals, lower doses are advisable. Some adverse reactions may be alleviated in the ambulatory patient who lies down.
Other adverse reactions include the following:
Central Nervous System
- Euphoria, dysphoria, weakness, headache, insomnia, agitation, disorientation, and visual disturbances.
Gastrointestinal
- Dry mouth, anorexia, constipation, and biliary tract spasm.
Cardiovascular
- Flushing of the face, bradycardia, palpitation, faintness and syncope.
Genitourinary
- Urinary retention or hesitancy, anti-diuretic effect, and reduced libido and/or potency.
Allergic
- Pruritus, urticaria, other skin rashes, edema, and rarely hemorrhagic urticaria.
Treatment of the most frequent adverse reactions:
Constipation
- Ample intake of water or other liquids should be encouraged.
- Concomitant administration of a stool softener and a peristaltic stimulant with the narcotic analgesic can be an effective preventive measure for those patients in need of therapeutics. If elimination does not occur for two days, an enema should be administered to prevent impaction.
- In the event diarrhea occurs, seepage around a fecal impaction is a possible cause to consider before antidiarrheal measures are employed.
Nausea and Vomiting
- Phenothiazines and antihistamines can be effective treatments for nausea of the medullary and vestibular sources respectively.
- However, these drugs may potentiate the side effects of the narcotic or the antinauseant.
Drowsiness (sedation)
- Once pain control is achieved, undesirable sedation can be minimized by titrating the dosage to a level that just maintains a tolerable pain or pain free state.
Postmarketing Experience
There is limited information regarding Morphine (rectal) Postmarketing Experience in the drug label.
Drug Interactions
Interaction with Other Central-Nervous-System Depressants
- Morphine should be used with caution and in reduced dosage in patients who are concurrently receiving other narcotic analgesics, general anesthetics, phenothiazines, other tranquilizers, sedative-hypnotics, tricyclic antidepressants, and other CNS depressants (including alcohol). Respiratory depression, hypotension, and profound sedation or coma may result.
Other Drug Interactions
- Generally, effects of morphine may be potentiated by alkalizing agents and antagonized by acidifying agents. Analgesic effect of morphine is potentiated by chlorpromazine and methocarbamol, CNS depressants such as anesthetics, hypnotics, barbiturates, phenothiazines, chloral hydrate, glutethimide, sedatives, MAO inhibitors (including procarbazine hydrochloride), antihistamines, beta-blockers (propranolol), alcohol, furazolidone and other narcotics may enhance the depressant effects of morphine.
- Morphine may increase anticoagulant activity of Coumadin® and other anticoagulants.
Use in Specific Populations
Pregnancy
- Animal reproduction studies have not been conducted with morphine. It is also not known whether morphine can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Morphine should be given to a pregnant woman only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Morphine (rectal) in women who are pregnant.
Labor and Delivery
- Morphine readily crosses the placental barrier and if administered during labor may lead to respiratory depression in the neonate.
Nursing Mothers
- Morphine has been detected in human milk. For this reason, caution should be exercised when morphine is administered to a nursing woman.
Pediatric Use
There is no FDA guidance on the use of Morphine (rectal) in pediatric settings.
Geriatic Use
There is no FDA guidance on the use of Morphine (rectal) in geriatric settings.
Gender
There is no FDA guidance on the use of Morphine (rectal) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Morphine (rectal) with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Morphine (rectal) in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Morphine (rectal) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Morphine (rectal) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Morphine (rectal) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Rectal
Monitoring
There is limited information regarding Morphine (rectal) Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Morphine (rectal) and IV administrations.
Overdosage
Signs and Symptoms:
- Serious overdosage is characterized by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, maximally constricted pupils, skeletal muscle flaccidity, cold and clammy skin, and sometimes bradycardia and hypotension. In severe overdosage, particularly by the IV route, apnea, circulatory collapse, cardiac arrest, and death may occur.
Treatment of Overdose:
- Primary attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. If depressed respiration is associated with muscular rigidity, an IV neuromuscular blocking agent may be required to facilitate assisted or controlled respiration.
- The narcotic antagonists-nalorphine, naloxone, and levallorphan are specific antidotes against respiratory depression resulting from overdosage or unusual sensitivity to narcotics. Thus, an antagonist should be administered, preferably by the IV route, simultaneously with efforts at respiratory resuscitation. Since the duration of action of morphine may exceed that of the antagonist, repeated doses of the antagonist may be required to maintain adequate respiration; the patient must be kept under surveillance.
- Oxygen, intravenous fluids, vasopressors, and other supportive measures should be employed as indicated. In cases of oral overdose, the stomach should be evacuated by emesis or gastric lavage if treatment can be instituted within 2 hours following ingestion. The patient should be observed closely for a rise in temperature or pulmonary complications that may signal the need for institution of antibiotic therapy.
Pharmacology
Mechanism of Action
- Morphine sulfate, an opioid agonist, is relatively selective for the mu receptor, although it can interact with other opioid receptors at higher doses. In addition to analgesia, the widely diverse effects of morphine include drowsiness, changes in mood, respiratory depression, decreased gastrointestinal motility, nausea, vomiting, and alterations of the endocrine and autonomic nervous system.
- Morphine produces both its therapeutic and its adverse effects by interaction with one or more classes of specific opioid receptors located throughout the body. Morphine acts as a full agonist, binding with and activating opioid receptors at sites in the peri-aqueductal and peri-ventricular grey matter, the ventro-medial medulla and the spinal cord to produce analgesia
- Morphine is a potent narcotic analgesic; it's principal therapeutic effect is relief of pain. In addition to analgesia, narcotics produce drowsiness, changes in mood, and mental clouding; although neither sensory modalities nor motor activity are blocked at therapeutic doses. There is no intrinsic limit to the analgesic effect. Clinically, however, dosage limitations are imposed by the adverse effects, primarily respiratory depression, nausea, and vomiting, which can result from high doses.
- Morphine has diverse additional actions. It depresses the respiratory center, stimulates the vomiting center, depresses the cough reflex, constricts the pupils, increases the tone of gastrointestinal and genitourinary tracts, and produces mild vasodilation.
Structure
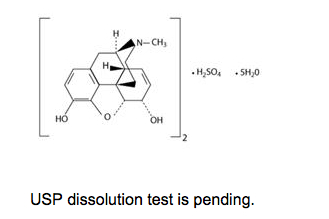
- Each suppository for rectal administration contains 5 mg, 10 mg, 20 mg, or 30 mg of Morphine Sulfate in a bland, specially formulated Hydrogenated Vegetable Oil Base with BHA and BHT as preservatives, as well as other ingredients.
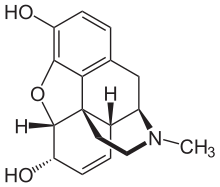
- Chemically, Morphine Sulfate is, Morphinan-3, 6-diol, 7,8-didehydro-4,5-epoxy-17-methyl-, (5a,6a)-, sulfate (2:1) (salt), pentahydrate, which can be represented by the following structural formula:
Pharmacodynamics
There is limited information regarding Morphine (rectal) Pharmacodynamics in the drug label.
Pharmacokinetics
- Morphine is detoxified in the liver by means of conjugation with glucuronic acid. Small amounts of the free drug and larger amounts of conjugated morphine are present in the urine, and these account for most of the administered drug. Ninety percent of the total excretion occurs within the first 24 hours.
- Morphine is about two-thirds absorbed from the gastrointestinal tract with the maximum analgesic effect occuring 20-60 minutes post administration.
Nonclinical Toxicology
There is limited information regarding Morphine (rectal) Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Morphine (rectal) Clinical Studies in the drug label.
How Supplied
Morphine Sulfate Suppositories are available in cartons of 12. Easy-to-open, color-coded:
5 mg Suppositories (Red) 12's NDC 0574-7110-12
10 mg Suppositories (Light Blue) 12's NDC 0574-7112-12
20 mg Suppositories (Light Green) 12's NDC 0574-7114-12
30 mg Suppositories (Purple) 12's NDC 0574-7116-12
Storage
There is limited information regarding Morphine (rectal) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Morphine (rectal) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
PRINCIPAL DISPLAY PANEL - 5 MG SUPPOSITORY CARTON
Rx Only
NDC 0574-7110-12
Morphine Sulfate Suppositories 5 mg CII
FOR RECTAL USE ONLY Warning: May be habit forming.
12 Suppositories UNIT DOSE
Perrigo®
PRINCIPAL DISPLAY PANEL - 10 MG SUPPOSITORY CARTON
Rx Only
NDC 0574-7112-12
Morphine Sulfate Suppositories 10 mg CII
FOR RECTAL USE ONLY Warning: May be habit forming.
12 Suppositories UNIT DOSE
Perrigo®
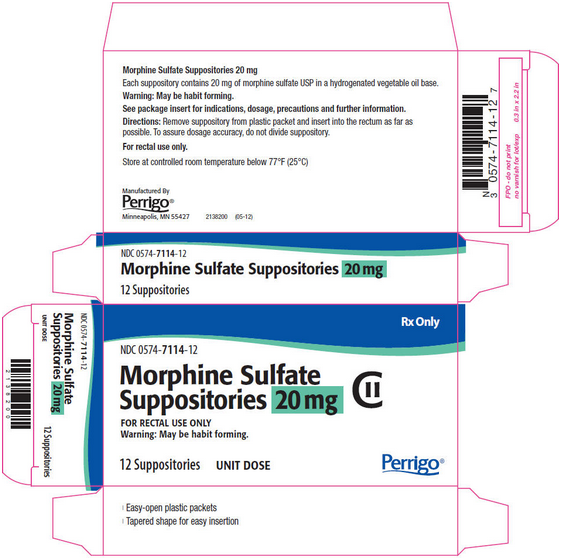
PRINCIPAL DISPLAY PANEL - 20 MG SUPPOSITORY CARTON
Rx Only
NDC 0574-7114-12
Morphine Sulfate Suppositories 20 mg CII
FOR RECTAL USE ONLY Warning: May be habit forming.
12 Suppositories UNIT DOSE
Perrigo®
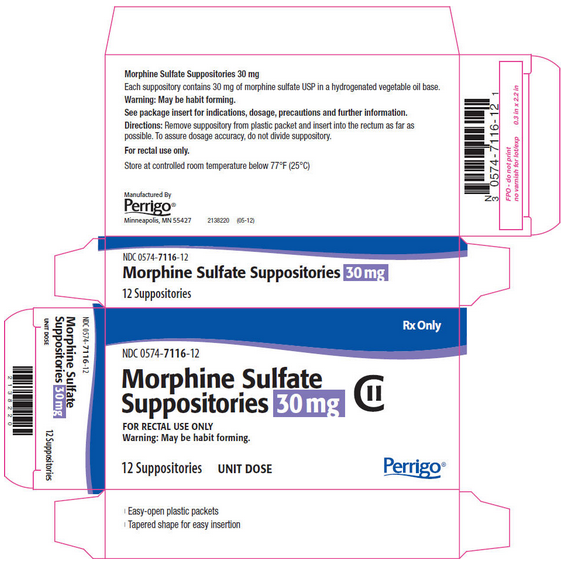
PRINCIPAL DISPLAY PANEL - 30 MG SUPPOSITORY CARTON
Rx Only
NDC 0574-7116-12
Morphine Sulfate Suppositories 30 mg CII
FOR RECTAL USE ONLY Warning: May be habit forming.
12 Suppositories UNIT DOSE
Perrigo®
{{#ask: Label Page::Morphine (rectal) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Use in Ambulatory Patients
- Morphine may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a car or operating machinery. The patient should be cautioned accordingly.
- Morphine, like other narcotics, may produce orthostatic hypotension in ambulatory patients.
- Patients should be cautioned about the combined effects of alcohol or other central nervous system depressants with morphine.
Precautions with Alcohol
Alcohol-Morphine (rectal) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Morphine (rectal) Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Morphine (rectal) Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Jonsson T, Christensen CB, Jordening H, Frølund C (April 1988). "The bioavailability of rectally administered morphine". Pharmacol. Toxicol. 62 (4): 203–5. doi:10.1111/j.1600-0773.1988.tb01872.x. PMID 3387374.