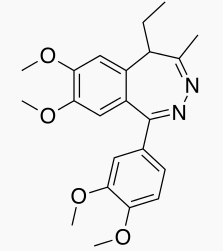
Tofisopam
 | |
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | Hepatic |
| Elimination half-life | 6-8 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C22H26N2O4 |
| Molar mass | 382.5 |
|
WikiDoc Resources for Tofisopam |
|
Articles |
|---|
|
Most recent articles on Tofisopam |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Tofisopam at Clinical Trials.gov Clinical Trials on Tofisopam at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Tofisopam
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Tofisopam Discussion groups on Tofisopam Directions to Hospitals Treating Tofisopam Risk calculators and risk factors for Tofisopam
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Tofisopam |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Tofisopam (marketed under brand names Emandaxin and Grandaxin) is a drug which is a benzodiazepine derivative. Like other benzodiazepines, it possesses anxiolytic properties but unlike other benzodiazepines it does not have anticonvulsant, sedative,[1] skeletal muscle relaxant, motor skill-impairing or amnestic[2] properties. While it may not be an anticonvulsant in and of itself, it has been shown to enhance the anticonvulsant action of classical 1,4-benzodiazepines such as diazepam (but not sodium valproate, carbamazepine, phenobarbital, or phenytoin).[3] Tofisopam is indicated for the treatment of anxiety and alcohol withdrawal, and is prescribed in a dosage of 50 - 300mg per day divided into three doses. Peak plasma levels are attained two hours after an oral dose. Tofisopam is not reported as causing dependance to the same extent as other benzodiazepines, but is still recommended to be prescribed for a maximum of 12 weeks. [4]
Tofisopam is not approved for sale in the United States or Canada. However, Vela Pharmaceuticals of New Jersey is developing the D- enantiomer (dextofisopam) as a treatment for irritable bowel syndrome.[5]
References
- ↑ Bond, A (1982). "A comparison of the psychotropic profiles of tofisopam and diazepam". European Journal of Clinical Pharmacology. 22 (2): 137–42. PMID 6124424. Unknown parameter
|coauthors=ignored (help) - ↑ Seppala, T (1980). "Tofisopam, a novel 3,4-benzodiazepine: multiple-dose effects on psychomotor skills and memory. Comparison with diazepam and interactions with ethanol". Psychopharmacology (Berlin). 69 (2): 209–18. PMID 6109345. Unknown parameter
|coauthors=ignored (help) - ↑ Saano, V. (1986). "Tofizopam selectively increases the action of anticonvulsants". Medical Biology. 64 (4): 201–6. PMID 3023768.
- ↑ http://www.biam2.org/www/Sub1469.html
- ↑ Vela Pharmaceuticals (2005). "Vela Announces Positive Phase 2 Results for Dextofisopam in Treating Irritable Bowel Syndrome - IBS: Results Show Effects of Dextofisopam Both in Women and in Men". VelaPharm - News. Retrieved 21 February. Unknown parameter
|accessyear=ignored (|access-date=suggested) (help); Check date values in:|accessdate=(help)
External links
- Pages with script errors
- Pages with citations using unsupported parameters
- CS1 errors: dates
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Drug
- Anxiolytics
- Benzodiazepines