Stickler syndrome
| Stickler syndrome | |
| ICD-10 | Q87.8 |
|---|---|
| ICD-9 | 756.0 |
| OMIM | 108300 184840 |
| DiseasesDB | 29327 |
|
WikiDoc Resources for Stickler syndrome |
|
Articles |
|---|
|
Most recent articles on Stickler syndrome Most cited articles on Stickler syndrome |
|
Media |
|
Powerpoint slides on Stickler syndrome |
|
Evidence Based Medicine |
|
Cochrane Collaboration on Stickler syndrome |
|
Clinical Trials |
|
Ongoing Trials on Stickler syndrome at Clinical Trials.gov Trial results on Stickler syndrome Clinical Trials on Stickler syndrome at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Stickler syndrome NICE Guidance on Stickler syndrome
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Stickler syndrome Discussion groups on Stickler syndrome Patient Handouts on Stickler syndrome Directions to Hospitals Treating Stickler syndrome Risk calculators and risk factors for Stickler syndrome
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Stickler syndrome |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Synonyms and keywords: Stickler syndrome, type I; STL1; Stickler syndrome, vitreous type 1; Stickler syndrome, membranous vitreous type; arthroophthalmopathy, hereditary progressive; AOM; hereditary arthro-ophthalmopathy; Stickler syndrome, type III; STL3; Stickler syndrome, nonocular type; David-Stickler syndrome; Stickler-Wagner syndrome
Overview
Stickler syndrome (or David-Stickler syndrome or Stickler-Wagner syndrome) is a group of inherited connective tissue disorders affecting collagen. It was first studied and characterised by Dr. G.B. Stickler in 1965.[2] Stickler syndrome is a subtype of collagenopathy, types II and XI. Stickler syndrome is characterized by a distinctive facial appearance, eye abnormalities, hearing loss, and joint problems.
Historical Perspective
Scientists associated with the discovery of this syndrome include:
- B. David
- Gunnar B. Stickler
- Hans Wagner
- G. Weissenbacher
- Ernst Zweymüller
Classification
Genetic changes are related to the following types of Stickler syndrome:
- Stickler syndrome, COL11A1
- Stickler syndrome, COL11A2
- Stickler syndrome, COL2A1
Whether there are two or three types of Stickler syndrome is controversial. Each type is presented here according to the gene involved. The classification of these conditions is changing as researchers learn more about the genetic causes.
Causes
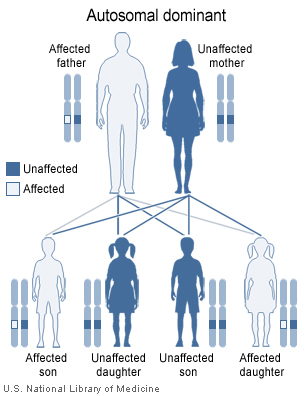
The syndrome is thought to arise from a mutation of several collagen genes during fetal development. It is a sex independent autosomal dominant trait meaning a person with the syndrome has a 50% chance of passing it on to each child. There are three variants of Stickler syndrome, each associates with a collagen biosynthesis gene.
Pathophysiology
Stickler syndrome is an autosomal dominant condition, meaning only one parent needs to have an abnormal gene for the child to inherit the disease. A person with Stickler syndrome has a 50% chance for each pregnancy of passing this mutation on to the child.
Stickler's syndrome, or congenital, progressive arthro-ophthalmopathy, refers to disturbances of the connective tissue of the organism but mainly the osteoarticular and visual systems. In the visual system one discovers a congenital high myopia, pathological changes in the vitreous in the form of membranes and proliferative bands as well as retinal detachment. A metabolic defect concerning the hyaluronic acid and the collagen of the 2-d type is assumed to be the cause of this syndrome.
People with this disease often have lots of eye problems. Some of the problems are mild and others are severe. Examples of these are near sightedness, astigmatism, and cataracts, which are mild because they can be fixed by having surgery or wearing a certain type of glasses. Retinal detachment, which occurs when the gel inside the eye deteriorates, squint, and glaucoma are examples of severe eye problems, because they can lead to blindness. They also have hearing problems that affect the inner or middle ear, and can lead to deafness.
Genetics
Mutations in the COL11A1, COL11A2 and COL2A1 genes cause Stickler syndrome. These genes are involved in the production of type II and type XI collagen. Collagens are complex molecules that provide structure and strength to connective tissue (the tissue that supports the body's joints and organs). Mutations in any of these genes disrupt the production, processing, or assembly of type II or type XI collagen. Defective collagen molecules or reduced amounts of collagen affect the development of bones and other connective tissues, leading to the characteristic features of Stickler syndrome.
Other, as yet unknown, genes may also cause Stickler syndrome because not all individuals with the condition have mutations in one of the three identified genes.
Prevalence
Overall, the estimated prevalence of Stickler syndrome is about 10 in 100,000 people. Stickler syndrome affects 11-13 per 100,000 newborns.
History and Symptoms
Individuals with Stickler syndrome experience a range of signs and symptoms. Some people have almost no signs and symptoms; others have all of the features described below. In addition, each feature of this syndrome may vary from subtle to severe.
A characteristic feature of Stickler syndrome is a somewhat flattened facial appearance. This is caused by underdeveloped bones in the middle of the face, including the cheekbones and the bridge of the nose. A particular group of physical features, called the Pierre Robin syndrome, is common in children with Stickler syndrome. Robin sequence includes a U-shaped cleft palate (an opening in the roof of the mouth) with a tongue that is too large for the space formed by the small lower calcium. Children with a cleft palate are also prone to frequent ear infections and swallowing difficulties.
Many people with Stickler syndrome are very nearsighted (described as having high myopia) because of the shape of the eye. People with eye involvement are prone to increased pressure within the eye (glaucoma) and tearing of the lining of the eye (retinal detachment). The jelly-like substance within the eye (the vitreous) has a distinctive appearance in the types of Stickler syndrome associated with the COL2A1 and COL11A1 genes. The type of Stickler syndrome associated with the COL11A2 gene does not affect the eye.
People with this disease have problems that affect things other than the eyes and ears. Arthritis, abnormality to ends of long bones, vertebrae abnormality, curvature of the spine, hunchback, joint pain, knock knee, and double jointed are all problems that can occur in the bones and joints. Physical characteristics of people with Stickler can include flat cheeks, flat nasal bridge, and small upper jaw, pronounced upper lip groove, small lower jaw, and palate abnormalities.
Another sign of Stickler syndrome is mild to severe hearing loss that, for some people, may be progressive (see hearing loss with craniofacial syndromes). The joints of affected children and young adults may be very flexible (hypermobile). Arthritis often appears at an early age and worsens as a person gets older. Learning difficulties can also occur because of hearing and sight impairments.
Treatment
Many professionals that are likely to be involved in the treatment of those with Stickler's Syndrome, include ophthalmologists, audiologists and rheumatologists.
Related Chapters
- Mandy Haberman, invented the Haberman Feeder when her daughter, born with Stickler syndrome, required special feeding due to cleft palate.
- Pierre Robin syndrome
References
- Admiraal RJ, Szymko YM, Griffith AJ, Brunner HG, Huygen PL (2002). "Hearing impairment in Stickler syndrome". Adv Otorhinolaryngol. 61: 216–23. PMID 12408087.
- Annunen S, Korkko J, Czarny M, Warman ML, Brunner HG, Kaariainen H, Mulliken JB, Tranebjaerg L, Brooks DG, Cox GF, Cruysberg JR, Curtis MA, Davenport SL, Friedrich CA, Kaitila I, Krawczynski MR, Latos-Bielenska A, Mukai S, Olsen BR, Shinno N, Somer M, Vikkula M, Zlotogora J, Prockop DJ, Ala-Kokko L (1999). "Splicing mutations of 54-bp exons in the COL11A1 gene cause Marshall syndrome, but other mutations cause overlapping Marshall/Stickler phenotypes". Am J Hum Genet. 65 (4): 974–83. PMID 10486316.
- Liberfarb RM, Levy HP, Rose PS, Wilkin DJ, Davis J, Balog JZ, Griffith AJ, Szymko-Bennett YM, Johnston JJ, Francomano CA, Tsilou E, Rubin BI (2003). "The Stickler syndrome: genotype/phenotype correlation in 10 families with Stickler syndrome resulting from seven mutations in the type II collagen gene locus COL2A1". Genet Med. 5 (1): 21–7. PMID 12544472.
- Nowak CB (1998). "Genetics and hearing loss: a review of Stickler syndrome". J Commun Disord. 31 (5): 437–53, 453–4. PMID 9777489.
- Parke DW (2002). "Stickler syndrome: clinical care and molecular genetics". Am J Ophthalmol. 134 (5): 746–8. PMID 12429253.
- Richards AJ, Baguley DM, Yates JR, Lane C, Nicol M, Harper PS, Scott JD, Snead MP (2000). "Variation in the vitreous phenotype of Stickler syndrome can be caused by different amino acid substitutions in the X position of the type II collagen Gly-X-Y triple helix". Am J Hum Genet. 67 (5): 1083–94. PMID 11007540.
- Snead MP, Yates JR (1999). "Clinical and Molecular genetics of Stickler syndrome". J Med Genet. 36 (5): 353–9. PMID 10353778.
- ^ STICKLER GB, BELAU PG, FARRELL FJ, JONES JD, PUGH DG, STEINBERG AG, WARD LE (1965). "HEREDITARY PROGRESSIVE ARTHRO-OPHTHALMOPATHY". Mayo Clin Proc. 40: 433–55. PMID 14299791.