Pridinol
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | oral, parenteral |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
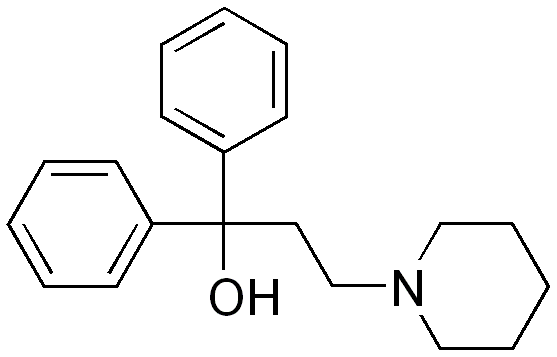
| Formula | C20H25NO |
| Molar mass | 295.419 g/mol |
| |
| | |
|
WikiDoc Resources for Pridinol |
|
Articles |
|---|
|
Most recent articles on Pridinol |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Pridinol at Clinical Trials.gov Clinical Trials on Pridinol at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Pridinol
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Pridinol Risk calculators and risk factors for Pridinol
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Pridinol |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Pridinol is a muscle relaxant.
Synthesis

This is formed by conjugate addition of piperidine to ethyl acrylate to give the aminoester, followed by addition of phenylmagnesium bromide.
Uses
It is used as an antiparkinsonian and anticholinergic drug.
See also
References
- Pages with script errors
- Template:drugs.com link with non-standard subpage
- Articles with changed CASNo identifier
- Articles with changed DrugBank identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Articles without KEGG source
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Piperidines
- Alcohols
- Muscle relaxants
- Antiparkinsonian agents
- Anticholinergics
- Drug