Leishmaniasis laboratory tests
|
Leishmaniasis Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Leishmaniasis laboratory tests On the Web |
|
American Roentgen Ray Society Images of Leishmaniasis laboratory tests |
|
Risk calculators and risk factors for Leishmaniasis laboratory tests |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alejandro Lemor, M.D. [2]
Overview
Diagnosis of visceral leishmaniasis requires a blood sample and/or taking a biopsy from the bone marrow to observe the parasite. Diagnosis of cutaneous leishmaniasis will require a small biopsy or scraping of the ulcer. Diagnosis of mucocutaneous leishmaniasis requires a biopsy of the affected tissues. Biopsy samples are examined by microscopy, culture and other methods to look for the parasite and identify the specific kind of Leishmania causing the ulcer. Some of these methods will give results within a few days, but culture may take 2-4 weeks to demonstrate the parasite. Examination of Giemsa stained slides of the relevant tissue is still the technique most commonly used to detect the parasite. Additional routine tests, such as complete blood count and serum protein, are also used for diagnosis. Other diagnostic techniques exist that allow parasite detection and/or species identification using biochemical (isoenzymes), immunologic (immunoassays), and molecular (PCR) approaches.
Laboratory Findings
| List of laboratory test used to diagnose Leishmaniasis |
|---|
|
|
|
|
|
|
|
|
|
- Various laboratory methods can be used to diagnose leishmaniasis,to detect the parasite and to identify the Leishmania species.[1]
- Leishmaniasis is diagnosed by detecting Leishmania parasites (or DNA) in tissue specimens—such as from skin lesions, for cutaneous leishmaniasis, or from bone marrow, for visceral leishmaniasis via light-microscopic examination of stained slides, specialized culture techniques, or molecular methods.
- Identification of the Leishmania species also can be important, particularly if more than one species is found where the patient lived or traveled and if they can have different clinical and prognostic implications.
- The species can be identified by various approaches, such as molecular methods and biochemical techniques (isoenzyme analysis of cultured parasites).
- For visceral leishmaniasis, serologic testing can provide supportive evidence for the diagnosis.
- The performance of various serologic assays may vary by geographic region and by host factors (for example, the sensitivity of serologic testing generally is lower in HIV-coinfected patients, particularly if the HIV infection predated the Leishmania infection).
- Most serologic assays do not reliably distinguish between active and quiescent infection.
- No leishmanin skin-test preparation has been approved for use in the United States
Isoenzyme analysis
- Isolation can be done using the biphasic medium which includes a solid phase composed of blood agar base (e.g., NNN medium), with defribinated rabbit blood.
- After isolation parasites can be characterized to the complex and sometimes to the species level using isoenzyme analysis, which is the conventional diagnostic approach for Leishmania species identification.
- Diagnostic identification of Leishmania using this approach may take several weeks.
Serology
- Antibody detection can prove useful in visceral leishmaniasis but is of limited value in cutaneous disease, since most patients do not develop a significant antibody response.
- In addition, cross reactivity can occur with Trypanosoma cruzi, a fact to consider when investigating Leishmania antibody response in patients who have been in Central or South America.
Molecular Diagnosis
- Molecular approaches have the potential to be more sensitive and rapid; e.g., the results can be available within days versus weeks. CDC has incorporated molecular methods in the algorithm for the laboratory diagnosis of leishmaniasis.
- The method is based on PCR amplification using generic primers that amplify a segment of the rRNA internal transcribed spacer 2 (ITS2) from multiple Leishmania species. DNA sequencing analysis is performed on the amplified fragment for species identification.
- This approach allows the differentiation among Viannia spp., namely, L. (V.) braziliensis, L. (V.) guyanensis, and L. (V.) panamensis as well as L. (L.) aethiopica, L. (L.) amazonensis, L. (L.) donovani, L. (L.) infantum/chagasi, (L.) major, L. (L.) mexicana and L. L. (L.) tropica
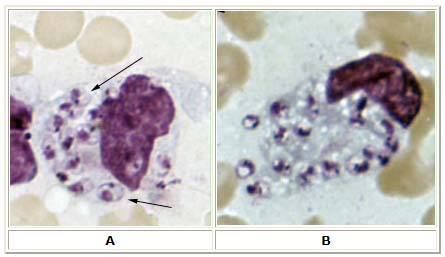
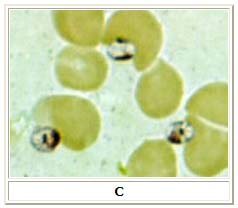
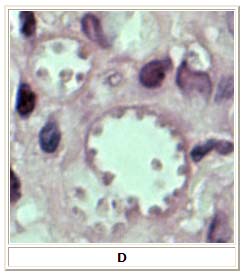
Microscopy
- In the human host, only the amastigotes stage is seen upon microscopic examination of tissue specimens.
- Amastigotes can be visualized with both Giemsa and hematoxylin and eosin (H&E) stains.
- The amastigotes of Leishmania spp. are morphologically indistinguishable from those of Trypanosoma cruzi.
- Amastigotes are ovoid and measure 1-5 micrometers long by 1-2 micrometers wide. They possess both a nucleus and kinetoplast.
- Isolation of the organism in culture (using for example the diphasic NNN medium) constitutes another method for confirmation of the diagnosis, and in addition can provide material for further investigations (e.g., isoenzyme analysis).