Chemical substance

Overview
A chemical substance is a material with a definite chemical composition. It is a concept that became firmly established in the late eighteenth century after work by the chemist Joseph Proust on the composition of some pure chemical compounds such as basic copper carbonate.[1] He deduced that: "All samples of a compound have the same composition; that is, all samples have the same proportions, by mass, of the elements present in the compound". This is now known as the law of constant composition[2]. Later with the advancement of methods for chemical synthesis particularly in the realm of organic chemistry; the discovery of many more chemical elements and new techniques in the realm of analytical chemistry used for isolation and purification of elements and compounds from chemicals that led to the establishment of modern chemistry, the concept was defined as is found in most chemistry textbooks. However, there are some controversies regarding this definition mainly because the large number of chemical substances reported in chemistry literature need to be indexed.
A common example of a chemical substance is pure water; it has the same properties and the same ratio of hydrogen to oxygen whether it is isolated from a river or made in a laboratory. A pure chemical substance cannot be separated into other substances by a process that does not involve any chemical reaction and is rarely found in nature. Some typical chemical substances can be diamond, gold, salt (sodium chloride) and sugar (sucrose). Generally, chemical substances exist as solid, liquid, or gas, and may change between these phases of matter with changes in temperature or pressure.
Defining
Chemical substances (also sometimes referred to as a pure substances) are often defined as any material with a definite chemical composition in most introductory general chemistry textbooks.[3] According to this definition a chemical substance can either be a pure chemical element or a pure chemical compound. However, there are exceptions to this definition, a pure substance can also be defined as a form of matter that has both definite composition and distinct properties.[4] and the chemical substance index published by CAS also includes several alloys of uncertain composition.[5] Nonstoichiometric compounds are a special case (in inorganic chemistry) that violates the law of constant composition, and for them, it is sometimes difficult to draw the line between a mixture and a compound, as in the case of palladium hydride.
Elements
An element is a chemical substance that is made up of a particular kind of atoms and hence cannot be broken down or transformed by a chemical reaction into a different element, though it can be transmutated into another element through a nuclear reaction. This is so, because all of the atoms in a sample of an element have the same number of protons, though they may be different isotopes, with differing numbers of neutrons.
There are about 120 known elements, about 80 of which are stable, that is, they do not change by radioactive decay into other elements, however the number of chemical substances that are elements can be more than 120, because some elements can occur as more than a single chemical substance (allotropes), for instance oxygen as oxygen and ozone. The majority of elements are classified as metals. These are elements with a characteristic lustre such as iron, copper, and gold. Metals typically conduct electricity and heat well, and they are malleable and ductile.[6] Around a dozen elements[7] such as carbon, nitrogen, and oxygen are classified as non-metals. Non-metals lack the metallic properties described above, they also have a high electronegativity and a tendency to form negative ions called anions. Certain elements such as silicon sometimes resemble metals and sometimes resemble non-metals, and are known as metalloids.
Chemical compounds
A pure chemical compound is a chemical substance that is composed of a particular set of molecules or ions. Two or more elements combined into one substance, through a chemical reaction, form what is called a chemical compound. A chemical compound can be either atoms bonded together in molecules or crystals in which atoms, molecules or ions form a crystalline lattice. Compounds based primarily on carbon and hydrogen atoms are called organic compounds, and all others are called inorganic compounds. Compounds containing bonds between carbon and a metal are called organometallic compounds.
Compounds in which components share electrons are known as covalent compounds. Compounds consisting of oppositely charged ions are known as ionic compounds, or salts.
In organic chemistry, there can be more than one chemical compound with the same composition and molecular weight. Generally, these are called isomers. Isomers usually have substantially different chemical properties, may be isolated and do not spontaneously convert to each other. A common example is glucose vs. fructose. The former is an aldehyde, the latter is a ketone. Their interconversion requires either enzymatic or acid-base catalysis. However, there are also tautomers, where isomerization occurs spontaneously, such that a pure substance cannot be isolated into its tautomers. A common example is glucose, which has open-chain and ring forms. One cannot manufacture pure open-chain glucose because glucose spontaneously cyclizes to the hemiacetal form.
Substances versus mixtures
All matter consists of various elements and chemical compounds, but these are often intimately mixed together. Mixtures contain more than one chemical substance, and they do not have a fixed composition. In principle, they can be separated into the component substances by purely mechanical processes. Butter, soil and wood are common examples of mixtures.
Grey iron metal and yellow sulfur are both chemical elements, and they can be mixed together in any ratio to form a yellow-grey mixture. No chemical process occurs, and the material can be identified as a mixture by the fact that the sulfur and the iron can be separated by a mechanical process, such as using a magnet to attract the iron away from the sulfur.
In contrast, if iron and sulfur are heated together in a certain ratio (56 grams (1 mol) of iron to 32 grams (1 mol) of sulfur), a chemical reaction takes place and a new substance is formed, the compound iron(II) sulfide, with chemical formula FeS. The resulting compound has all the properties of a chemical substance and is not a mixture. Iron(II) sulfide has its own distinct properties such as melting point and solubility, and the two elements cannot be separated using normal mechanical processes; a magnet will be unable to recover the iron, since there is no metallic iron present in the compound.
Chemicals versus Chemical substances
While the term chemical substance is a somewhat technical term used most often by professional chemists, the word chemical[8] is more widely used in the pharmaceutical industry, government and society in general. Thus the word chemical includes a much wider class of substances that includes many mixtures of chemical substances that often find application in many vocations [9]; and is most commonly used only for artificial or processed substances, such as the products of the chemical industry.
Naming and Indexing
Every chemical substance has one or more systematic names, usually named according to the IUPAC rules for naming. An alternative system is used by the Chemical Abstracts Service (CAS)
Many compounds are also known by their more common, simpler names, many of which predate the systematic name. For example, the long-known sugar glucose is now systematically named 6-(hydroxymethyl)oxane-2,3,4,5-tetrol. Natural products and pharmaceuticals are also given simpler names, for example the mild pain-killer Naproxen is the more common name for the chemical compound (S)-6-methoxy-α-methyl-2-naphthaleneacetic acid.
Chemists frequently refer to chemical compounds using chemical formulae or molecular structure of the compound. There has been a phenomenal growth in the number of chemical compounds being synthesized (or isolated), and then reported in the scientific literature by professional chemists around the world [10]. An enormous number of chemical compounds are possible through the chemical combination of the known chemical elements. At the last count, about thirty million chemical compounds are known.[11]. The names of many of these compounds are often nontrivial and hence not very easy to remember or cite accurately. Also it is difficult to keep the track of them in the literature. Several international organizations like the IUPAC and the Chemical Abstract Service (CAS) have intiated steps to make such tasks easier. CAS that provides the abstracting services of the chemical literature, provides a numerical identifier, known as CAS registry number to each chemical substance that has been reported in the chemical literature (such as chemistry journals and patents). This information is compiled as a database and is popularly known as the Chemical substances index. Other computer-friendly systems that have been developed for substance information, are: SMILES and the International Chemical Identifier or InChI.
| Common name | Systematic name | Chemical formula | Chemical structure | CAS registry number | InChI |
|---|---|---|---|---|---|
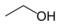
| alcohol, or ethyl alcohol |
ethanol | C2H5OH | [64-17-5] | 1/C2H6O/c1-2-3/h3H,2H2,1H3 |
Isolation, purification, characterisation and identification
Often a pure substance needs to be isolated from a mixture, for example from a natural source (where a sample often contains numerous chemical substances) or after a chemical reaction (which often give mixtures of chemical substances).
References and notes
- ↑ Hill, J. W.; Petrucci, R. H.; McCreary, T. W.; Perry, S. S. General Chemistry, 4th ed., p37, Pearson Prentice Hall, Upper Saddle River, New Jersey, 2005.
- ↑ http://dbhs.wvusd.k12.ca.us/webdocs/AtomicStructure/LawofDefiniteProportion.html
- ↑ Hill, J. W.; Petrucci, R. H.; McCreary, T. W.; Perry, S. S. General Chemistry, 4th ed., p5, Pearson Prentice Hall, Upper Saddle River, New Jersey, 2005
- ↑ http://www.diracdelta.co.uk/science/source/p/u/pure%20substance/source.html
- ↑ http://www.cas.org/ASSETS/58D34DD3892142D18F5C3B0A004D3A0C/indexguideapp.pdf
- ↑ Hill, J. W.; Petrucci, R. H.; McCreary, T. W.; Perry, S. S. General Chemistry, 4th ed., pp45-46, Pearson Prentice Hall, Upper Saddle River, New Jersey, 2005.
- ↑ The boundary between metalloids and non-metals is imprecise, as explained in the previous reference.
- ↑ What is a chemical
- ↑ http://www.bfr.bund.de/cd/569
- ↑ http://www.rz.uni-karlsruhe.de/~ed01/Jslit/eduquim.htm
- ↑ Chemical Abstracts substance count
ar:مادة كيميائية cs:Látka de:Stoff (Chemie) et:Keemiline aine el:Χημική ουσία eo:Kemiajxo id:Zat kimia he:כימיקלים lv:Ķīmiskā viela mk:Супстанција nl:Stof (chemie) no:Kjemisk substans sr:Супстанца su:Bahan kimia fi:Kemikaali sv:Kemikalie tl:Sustansyang kimikal th:สารเคมี yi:כעמיקאלע סובסטאנצן