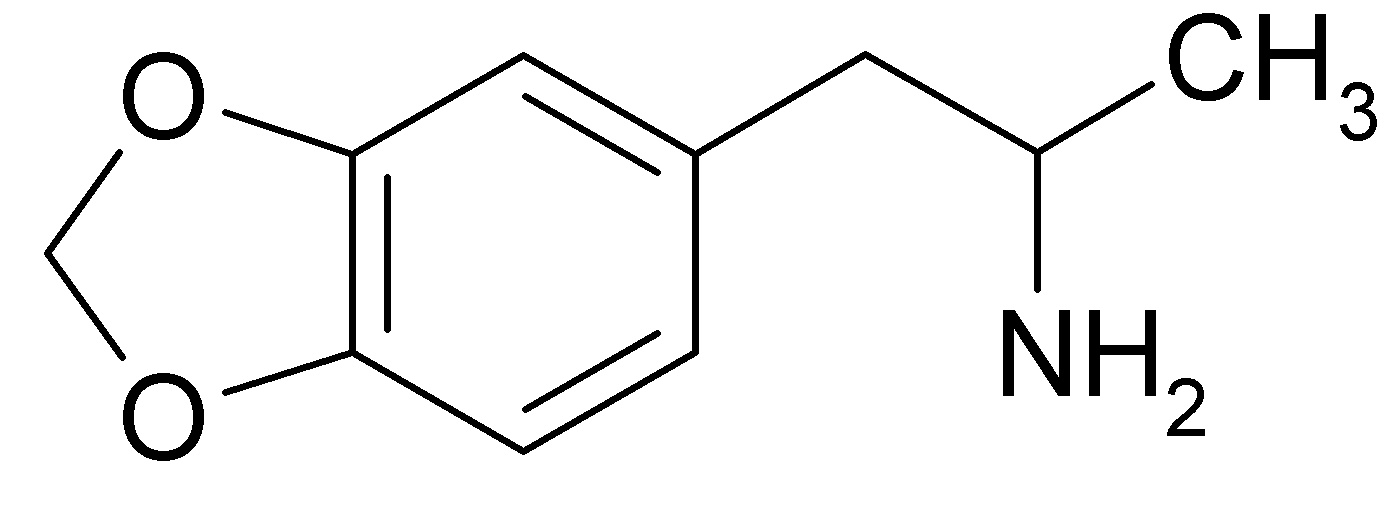
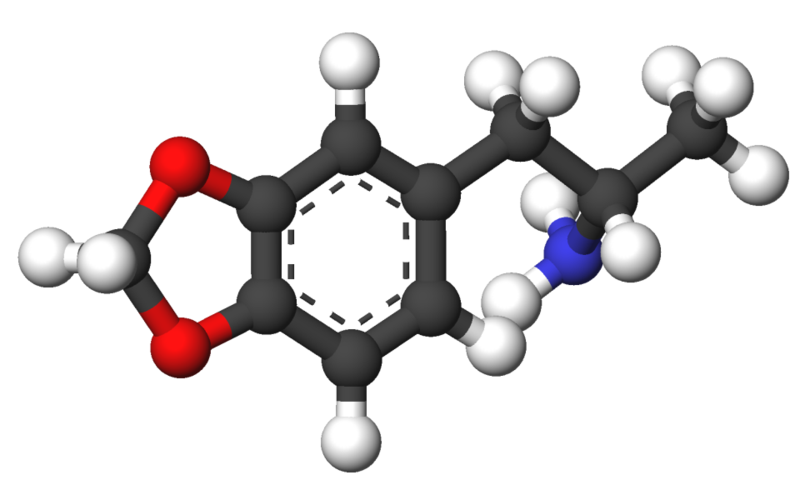
3,4-Methylenedioxyamphetamine
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Sublingual |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic, CYP extensively involved |
| Elimination half-life | dose dependent 6-10 hours |
| Excretion | Renal |
| Identifiers | |
| |
| PubChem CID | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C10H13NO2 |
| Molar mass | 179.22 g/mol |
| 3D model (JSmol) | |
| |
Overview
3,4-Methylenedioxyamphetamine (MDA or Tenamfetamine), is a psychedelic hallucinogenic drug and empathogen/entactogen of the phenethylamine family. It was first synthesized by G. Mannish and W. Jacobson in 1910. There are about 20 different synthetic routes described in the literature for its preparation.
Medical use
MDA was first used in animal tests in 1939, and human trials began in 1941 in the exploration of possible therapies for Parkinson's disease. From 1949 to 1957, more than 500 human subjects were given MDA in an investigation of its potential use as an antidepressant and/or anorectic by Smith, Kline, and French. The United States Army also experimented with the drug, code named EA-1298, while working to develop a truth drug or incapacitating agent. One human subject died in January 1953 after being intravenously injected with 500mg of the drug. MDA was patented as a cough suppressant by H. D. Brown in 1958, as an ataractic by Smith, Kline, and French in 1960, and as an anorectic under the trade name “Amphedoxamine” in 1961. Several researchers, including Claudio Naranjo, have explored MDA in the field of psychotherapy.
MDA is very similar to MDMA but it is often described as less euphoric than its cousin. However, it can cause immense euphoria as well, but in a way described to be different. MDA is said universally to have much more psychedelic activity than MDMA
Synthesis
One method of MDA synthesis, is to turn safrole into isosafrole via isomerization. The isosafrole is then oxidized, using a Wacker process or a peroxyacid, to produce MDP2P (methylenedioxyphenylacetone). Finally, it is converted to MDA via reductive amination of Ammonia. This synthesis is very similar to that of MDMA (Ecstasy) and of MDEA. Sometimes, because of the similar synthesis, a manufacturer who intends to make MDMA will accidentally have MDA as a result of a mistake in synthesis.
Recreational use
MDA began to appear on the recreational drug scene around 1963 to 1964. It was then inexpensive and readily available as a research chemical from several scientific supply houses. Although now illegal, MDA continues to be bought, sold, and used for recreational purposes, often in the form of tablets purporting to contain MDMA (Ecstasy).
Effects
A recreational dose of MDA is commonly between 80 and 160mg. The “R” optical isomer is more potent than the “S” optical isomer. Although there is some debate, the duration of the drug is now generally believed to be roughly 6 to 10 hours(In the late 90s, Alexander Shulgin changed his opinion of the duration to 3-6 hours). The effects of the drug are quite similar to those of MDMA (Ecstasy), including empathogen/entactogenic effects, though typically less intense than a similar dosage of MDMA. Because of these effects, MDA was called the “hug drug” and was alleged to stand for “Mellow Drug of America” in the 1960s. Some users feel that MDA has more psychedelic or hallucinogenic qualities than MDMA.
The toxicity of MDA is not fully known. The LD50 in mice has been reported as 92mg/kg by intraperitoneal injection. The website erowid.org lists the fatality rate at roughly 2 in 100,000 users.[1] MDA is considered to be more potentially neurotoxic than its methylated cousin MDMA. It is a direct metabolite of MDMA.
The experience is similar but has some distinct differences compared with MDMA. MDA causes the same kind of serotonin release as MDMA, leading to euphoria and its' empathogen/entactogen effects. However, MDA has a higher affinity for 5-HT2A receptors, thus causing more psychedelic-like effects, such as hallucinations.
Legality
In 1970, the Controlled Substances Act was enacted in the United States, placing MDA into Schedule I. It is similarly controlled in other nations. In Canada MDA is a Schedule III drug. Internationally, MDA is a Schedule I drug under the Convention on Psychotropic Substances. The US has also essentially banned any drug with (methylenedioxy) in its structure, this means that all unscheduled MDXX chemicals can be prosecuted under the Federal Analog Act [2]
See also
External links
References
- Lee, M.A. and Shlain, B., Acid Dreams: The CIA, LSD, and the Sixties Rebellion. Grove, 1985.
- Stafford, P. Psychedelics Encyclopedia. Ronin, 1992.
Template:Entactogens Template:Hallucinogenic phenethylamines Template:Amphetamines Template:PiHKAL
de:3,4-Methylendioxyamphetamin gl:Metilenedioxianfetamina nl:3,4-methyleendioxyamfetamine sv:MDA (preparat)
- Pages with script errors
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Articles containing unverified chemical infoboxes
- Amphetamines
- Entactogens and Empathogens
- Psychedelic phenethylamines