Tetracycline (topical): Difference between revisions
(Created page with "{{DrugProjectFormSinglePage |authorTag= <!--Overview--> |genericName= |aOrAn= a |drugClass= |indication= |hasBlackBoxWarning= Yes |adverseReactions= <!--Bla...") |
No edit summary |
||
| Line 1: | Line 1: | ||
{{DrugProjectFormSinglePage | {{DrugProjectFormSinglePage | ||
|authorTag= | |authorTag={{AV}} | ||
<!--Overview--> | <!--Overview--> | ||
|genericName= | |genericName=tetracycline hydrochloride ointment | ||
| Line 23: | Line 23: | ||
|hasBlackBoxWarning= | |hasBlackBoxWarning= | ||
|adverseReactions= | |adverseReactions= | ||
| Line 43: | Line 42: | ||
<!--FDA-Labeled Indications and Dosage (Adult)--> | <!--FDA-Labeled Indications and Dosage (Adult)--> | ||
|fdaLIADAdult= | |fdaLIADAdult=*first aid to help prevent infection in minor: | ||
:*cuts | |||
:*scrapes | |||
:*burns | |||
====Directions==== | |||
:*clean the affected area | |||
:*hold the tube gently, with swab handle facing upward, pinch tube firmly at dotted line. With other hand, grasp applicator handle at base. | |||
:*bend gently down until seal is broken, twist gently until handle separates from tube | |||
:*use EasySnap™ handle to pull swab from tube | |||
:*apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily | |||
:*may be covered with a sterile bandage | |||
:*discard swab applicator after use | |||
<!--Off-Label Use and Dosage (Adult)--> | <!--Off-Label Use and Dosage (Adult)--> | ||
| Line 96: | Line 104: | ||
|contraindications= | |contraindications= | ||
<!--Warnings--> | <!--Warnings--> | ||
| Line 102: | Line 110: | ||
|warnings= | |warnings= | ||
* | * For external use only | ||
*DO NOT USE | |||
:*if you are allergic to any of the ingredients | |||
:*Pin the eyes | |||
:*over large areas of the body | |||
*ASK A DOCTOR BEFORE USE IF YOU HAVE | |||
:*deep or puncture wounds | |||
:*animal bites | |||
:*serious burns | |||
* | *STOP USE AND ASK A DOCTOR IF | ||
:*you need to use longer than 1 week | |||
:*condition persists or gets worse | |||
:*rash or other allergic reaction develop | |||
*KEEP OUT OF REACH OF CHILDREN. | |||
:*If swallowed, get medical help or contact a Poison Control Center right away. | |||
<!--Adverse Reactions--> | <!--Adverse Reactions--> | ||
| Line 128: | Line 151: | ||
|drugInteractions= | |drugInteractions= | ||
<!--Use in Specific Populations--> | <!--Use in Specific Populations--> | ||
|useInPregnancyFDA= | |useInPregnancyFDA= | ||
|useInPregnancyAUS= | |useInPregnancyAUS= | ||
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of {{PAGENAME}} in women who are pregnant. | There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of {{PAGENAME}} in women who are pregnant. | ||
| Line 160: | Line 182: | ||
|useInRenalImpair= | |useInRenalImpair= | ||
There is no FDA guidance on the use of {{PAGENAME}} in patients with renal impairment. | There is no FDA guidance on the use of {{PAGENAME}} in patients with [[renal impairment]]. | ||
|useInHepaticImpair= | |useInHepaticImpair= | ||
There is no FDA guidance on the use of {{PAGENAME}} in patients with hepatic impairment. | There is no FDA guidance on the use of {{PAGENAME}} in patients with [[hepatic impairment]]. | ||
|useInReproPotential= | |useInReproPotential= | ||
| Line 169: | Line 191: | ||
|useInImmunocomp= | |useInImmunocomp= | ||
There is no FDA guidance one the use of {{PAGENAME}} in patients who are immunocompromised. | There is no FDA guidance one the use of {{PAGENAME}} in patients who are [[immunocompromised]]. | ||
<!--Administration and Monitoring--> | <!--Administration and Monitoring--> | ||
| Line 203: | Line 225: | ||
<!--Drug box 2--> | <!--Drug box 2--> | ||
|drugBox= | |drugBox={{drugbox2 | Watchedfields = changed | ||
| verifiedrevid = 470603713 | |||
| image = Tetracycline Structural Formula V.1.png | |||
<!--Clinical data--> | |||
| tradename = Sumycin | |||
| Drugs.com = {{drugs.com|monograph|tetracycline}} | |||
| MedlinePlus = a682098 | |||
| licence_US = Tetracycline | |||
| pregnancy_AU = D | |||
| pregnancy_US = D | |||
| legal_status = Rx-only | |||
| routes_of_administration = oral, [[topical]] (skin and eye), [[intramuscular]], [[intravenous]] | |||
<!--Pharmacokinetic data--> | |||
| bioavailability = 75% | |||
| metabolism = Not metabolised | |||
| elimination_half-life = 8-11 hours, 57-108 hours (kidney impairment) | |||
| excretion = Urine (>60%), feces | |||
<!--Identifiers--> | |||
| CASNo_Ref = {{cascite|correct|CAS}} | |||
| CAS_number_Ref = {{cascite|correct|??}} | |||
| CAS_number = 60-54-8 | |||
| CAS_supplemental = <br/>{{CAS|64-75-5}} (hydrochloride) <!-- Also CAS verified --> | |||
| ATC_prefix = A01 | |||
| ATC_suffix = AB13 | |||
| ATC_supplemental = {{ATC|D06|AA04}} {{ATC|J01|AA07}} {{ATC|S01|AA09}} {{ATC|S02|AA08}} {{ATC|S03|AA02}} {{ATCvet|G01|AA90}} {{ATCvet|G51|AA02}} {{ATCvet|J51|AA07}} | |||
| ChEBI_Ref = {{ebicite|correct|EBI}} | |||
| ChEBI = 27902 | |||
| PubChem = 643969 | |||
| DrugBank_Ref = {{drugbankcite|correct|drugbank}} | |||
| DrugBank = DB00759 | |||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} | |||
| ChemSpiderID = 10257122 | |||
| UNII_Ref = {{fdacite|correct|FDA}} | |||
| UNII = F8VB5M810T | |||
| KEGG_Ref = {{keggcite|correct|kegg}} | |||
| KEGG = D00201 | |||
| ChEMBL_Ref = {{ebicite|correct|EBI}} | |||
| ChEMBL = 1440 | |||
<!--Chemical data--> | |||
| C=22 | H=24 | N=2 | O=8 | |||
| molecular_weight = 444.435 g/mol | |||
| smiles = C[C@]1(c2cccc(c2C(=O)C3=C([C@]4([C@@H](C[C@@H]31)[C@@H](C(=C(C4=O)C(=O)N)O)N(C)C)O)O)O)O | |||
| InChI = 1/C22H24N2O8/c1-21(31)8-5-4-6-11(25)12(8)16(26)13-9(21)7-10-15(24(2)3)17(27)14(20(23)30)19(29)22(10,32)18(13)28/h4-6,9-10,15,25,27-28,31-32H,7H2,1-3H3,(H2,23,30)/t9-,10-,15-,21+,22-/m0/s1 | |||
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} | |||
| StdInChI = 1S/C22H24N2O8/c1-21(31)8-5-4-6-11(25)12(8)16(26)13-9(21)7-10-15(24(2)3)17(27)14(20(23)30)19(29)22(10,32)18(13)28/h4-6,9-10,15,25,27-28,31-32H,7H2,1-3H3,(H2,23,30)/t9-,10-,15-,21+,22-/m0/s1 | |||
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} | |||
| StdInChIKey = OFVLGDICTFRJMM-WESIUVDSSA-N | |||
}} | |||
| Line 249: | Line 322: | ||
|howSupplied= | |howSupplied= | ||
* | *tamper evident: safety sealed swabs. Do not use if seal has been broken. | ||
*store at room temperature 20-25°C (68-77°F) | |||
<!--Patient Counseling Information--> | <!--Patient Counseling Information--> | ||
Revision as of 15:13, 21 May 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Aparna Vuppala, M.B.B.S. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Tetracycline (topical) is a that is FDA approved for the {{{indicationType}}} of . Common adverse reactions include .
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- first aid to help prevent infection in minor:
- cuts
- scrapes
- burns
Directions
- clean the affected area
- hold the tube gently, with swab handle facing upward, pinch tube firmly at dotted line. With other hand, grasp applicator handle at base.
- bend gently down until seal is broken, twist gently until handle separates from tube
- use EasySnap™ handle to pull swab from tube
- apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
- discard swab applicator after use
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Tetracycline (topical) in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Tetracycline (topical) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Tetracycline (topical) in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Tetracycline (topical) in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Tetracycline (topical) in pediatric patients.
Contraindications
There is limited information regarding Tetracycline (topical) Contraindications in the drug label.
Warnings
- For external use only
- DO NOT USE
- if you are allergic to any of the ingredients
- Pin the eyes
- over large areas of the body
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- deep or puncture wounds
- animal bites
- serious burns
- STOP USE AND ASK A DOCTOR IF
- you need to use longer than 1 week
- condition persists or gets worse
- rash or other allergic reaction develop
- KEEP OUT OF REACH OF CHILDREN.
- If swallowed, get medical help or contact a Poison Control Center right away.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Tetracycline (topical) in the drug label.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Tetracycline (topical) in the drug label.
Drug Interactions
There is limited information regarding Tetracycline (topical) Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Tetracycline (topical) in women who are pregnant.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Tetracycline (topical) in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Tetracycline (topical) during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Tetracycline (topical) with respect to nursing mothers.
Pediatric Use
There is no FDA guidance on the use of Tetracycline (topical) with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of Tetracycline (topical) with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Tetracycline (topical) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Tetracycline (topical) with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Tetracycline (topical) in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Tetracycline (topical) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Tetracycline (topical) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Tetracycline (topical) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- Intravenous
Monitoring
There is limited information regarding Monitoring of Tetracycline (topical) in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Tetracycline (topical) in the drug label.
Overdosage
There is limited information regarding Chronic Overdose of Tetracycline (topical) in the drug label.
Pharmacology
Mechanism of Action
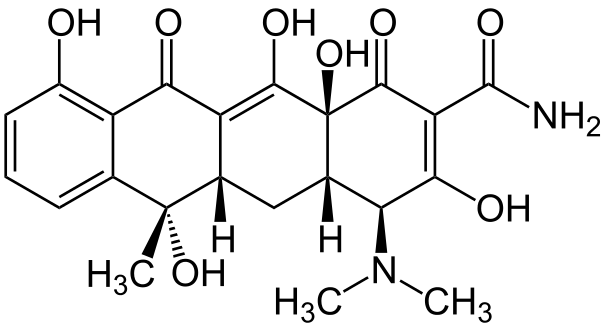
Structure
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Tetracycline (topical) in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Tetracycline (topical) in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Tetracycline (topical) in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Tetracycline (topical) in the drug label.
How Supplied
- tamper evident: safety sealed swabs. Do not use if seal has been broken.
- store at room temperature 20-25°C (68-77°F)
Storage
There is limited information regarding Tetracycline (topical) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Tetracycline (topical) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Tetracycline (topical) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Tetracycline (topical) in the drug label.
Precautions with Alcohol
- Alcohol-Tetracycline (topical) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- ®[1]
Look-Alike Drug Names
- A® — B®[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Empty citation (help)
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Label Page=Tetracycline (topical) |Label Name=Tetracycline (topical)11.png
}}
{{#subobject:
|Label Page=Tetracycline (topical) |Label Name=Tetracycline (topical)11.png
}}