Myocarditis endomyocardial biopsy: Difference between revisions
No edit summary |
|||
| Line 3: | Line 3: | ||
==Overview== | ==Overview== | ||
[[Endomyocardial biopsy]] remains the gold standard test to evaluate for the presence of and to subclassify the type of myocarditis. A small tissue sample of the [[endocardium]] and [[myocardium]] is obtained via right sided cardiac catheterization. The sample is then evaluated by a pathologist using [[immunochemistry]] and special staining techniques as necessary. Histopathological features include abundant [[edema]] in the myocardial [[interstitium]] and an inflammatory infiltrate which is rich in [[lymphocyte]]s and [[macrophage]]s. Focal destruction of [[myocytes]] as a result of the inflammatory process explains the myocardial [[pump failure]].<ref name="pmid11070105">{{cite journal| author=Feldman AM, McNamara D| title=Myocarditis. | journal=N Engl J Med | year= 2000 | volume= 343 | issue= 19 | pages= 1388-98 | pmid=11070105 | doi=10.1056/NEJM200011093431908 | pmc= | url= }} </ref> | |||
==Endomyocardial biopsy== | ==Endomyocardial biopsy== | ||
Revision as of 23:25, 5 September 2011
|
Myocarditis Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Myocarditis endomyocardial biopsy On the Web |
|
American Roentgen Ray Society Images of Myocarditis endomyocardial biopsy |
|
Risk calculators and risk factors for Myocarditis endomyocardial biopsy |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-In-Chief:Varun Kumar, M.B.B.S.
Overview
Endomyocardial biopsy remains the gold standard test to evaluate for the presence of and to subclassify the type of myocarditis. A small tissue sample of the endocardium and myocardium is obtained via right sided cardiac catheterization. The sample is then evaluated by a pathologist using immunochemistry and special staining techniques as necessary. Histopathological features include abundant edema in the myocardial interstitium and an inflammatory infiltrate which is rich in lymphocytes and macrophages. Focal destruction of myocytes as a result of the inflammatory process explains the myocardial pump failure.[1]
Endomyocardial biopsy
Heart Failure Society of America recommends that endomyocardial biopsy should be considered when the cardiac function acutely deteriorates with unknown etiology and is unresponsive to medical therapy (Strength of Evidence = B)[2].
Non-specific findings such as hypertrophy, cell loss and fibrosis may be noted on biopsy. However, these biopsy findings significantly affecting patient management has not been established conclusively[3]. For example, inflammatory changes in myocardium may be detected in viral myocarditis. Nevertheless, majority of the patients with biopsy proven myocarditis improve with just supportive care without any need for antiviral or anti-inflammatory treatment[4]. Endomyocardial biopsy has a low sensitivity and specificity which could be explained by the focal and transient nature of the inflammatory infiltrates[5][6].
Myocardial biopsy may be useful in:
- Confirming the clinically suspected cause of myocarditis.
- Confirming the presence of cardiac disorders such as amyloidosis which may weigh against the eligibility for transplantation.
- Giant cell myocarditis can be identified by biopsy. These patients generally progress rapidly to death and are unresponsive to treatment. Hence, myocardial biopsy helps in effective management of these patients by providing circulatory support or immediate heart transplantation[7].
Dallas criteria
Histologically, both active inflammatory cellular infiltrate within the myocardium and associated myocyte necrosis (the Dallas pathologic criteria)[8] may be noted in myocarditis. Dallas criteria despite its considerable limitations, has established uniform histologic criteria and has thus substantially reduced the variations in diagnosing this disease.
| “ |
|
” |
2009 ACC / AHA Guidelines for Endomyocardial Biopsy[9]
| “ |
Class IIa
Class III
|
” |
The AHA/ACCF/ESC Scientific Statement: The role of Endomyocardial Biopsy in fourteen clinical scenarios[10]
| “ |
Class I 1. New-onset heart failure of <2 weeks’ duration associated with a normal-sized or dilated left ventricle and hemodynamic compromise. (Level of Evidence: B) 2. New-onset heart failure of 2 weeks’ to 3 months’ duration associated with a dilated left ventricle and new ventricular arrhythmias, second- or third-degree heart block, or failure to respond to usual care within 1 to 2 weeks. (Level of Evidence: B) Class IIa 1. Heart failure of >3 months’ duration associated with a dilated left ventricle and new ventricular arrhythmias, second- or third-degree heart block, or failure to respond to usual care within 1 to 2 weeks. (Level of Evidence: C) 2. Heart failure associated with a DCM of any duration associated with suspected allergic reaction and/or eosinophilia. (Level of Evidence: C) 3. Heart failure associated with suspected anthracycline cardiomyopathy. (Level of Evidence: C) 4. Heart failure associated with unexplained restrictive cardiomyopathy. (Level of Evidence: C) 5. Suspected cardiac tumors. (Level of Evidence: C) 6. Unexplained cardiomyopathy in children. (Level of Evidence: C) Class IIb 1. New-onset heart failure of 2 weeks’ to 3 months’ duration associated with a dilated left ventricle, without new ventricular arrhythmias or second- or third-degree heart block, that responds to usual care within 1 to 2 weeks. (Level of Evidence: B) 2. Heart failure of >3 months’ duration associated with a dilated left ventricle, without new ventricular arrhythmias or second- or third-degree heart block, that responds to usual care within 1 to 2 weeks. (Level of Evidence: C) 3. Heart failure associated with unexplained HCM. (Level of Evidence: C) 4. Suspected ARVD/C. (Level of Evidence: C) 5. Unexplained ventricular arrhythmias. (Level of Evidence: C) Class III 1. Unexplained atrial fibrillation. (Level of Evidence: C) |
” |
Indications for endomyocardial biopsy[11][12]
- Subacute or acute symptoms of heart failure refractory to standard management
- Rapid worsening of ejection fraction despite standard pharmacological therapy
- Development of cardiac arrhythmias such as ventricular tachycardia and heart blocks
- Heart failure with concomitant rash, fever, or peripheral eosinophilia
- Cardiac dysfunction secondary to following causes as biopsy may change or modify the therapy:
Complications for endomyocardial biopsy[10][13]
Complications may be as high as 6% as observed in a series where 546 patients with cardiomyopathy underwent right ventricular endomyocardial biopsy[14]. Several other studies reported the incidence of complications ranging between 0.5-1.5%[13][15].
- Myocardial perforation leading to pericardial tamponade
- Heart block
- Pulmonary embolization
- Arrhythmias
- Pneumothorax
- Nerve injury
- Hematoma
- Tricuspid valve damage
- Arteriovenous fistula
- Deep venous thrombosis
Pathologic Findings
Autopsy Study
Clinical Summary
A 21-year-old male with sickle cell anemia had recurrent attacks of acute rheumatic fever beginning at age 14.
Mitral insufficiency and stenosis were present by age 16.
On prophylactic antibiotics, the patient had no evidence of recurrence until three weeks before his final admission, when an upper respiratory infection developed. A few weeks later he developed acute migratory polyarthritis. This was associated with rapid deterioration of cardiac function and death.
Autopsy Findings
At autopsy, the heart was enlarged (weighing 675 grams) especially the left atrium. Both the aortic and mitral valves showed fibrosis as well as the fresh, tiny verrucae characteristic of acute rheumatic fever.
Images
-
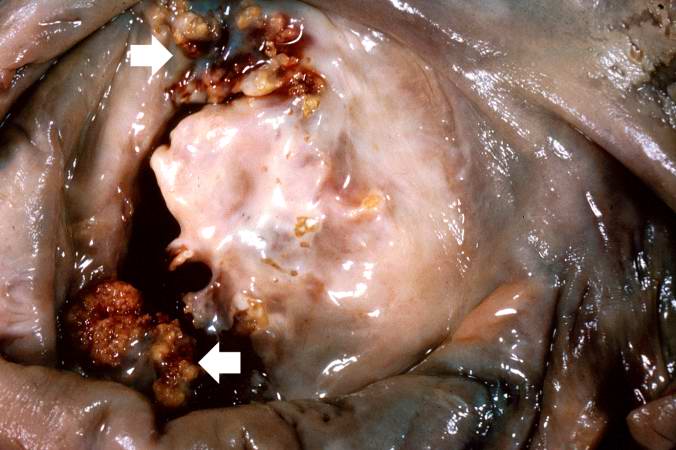
This is a gross photograph of mitral valve demonstrating marked thickening and fibrosis of the valve leaflet. There are also numerous foci of fibrinoid necrosis within the cusps and friable vegetations (verrucae) along the lines of closure (arrows). These irregular, warty projections are found at sites of erosion on the inflamed endocardial surface. The verrucae probably result from the precipitation of fibrin where the leaflets impinge on each other.
-
This is a low-power photomicrograph of heart tissue. Little can be seen at this magnification, except that the tissue looks relatively normal.
-
This is a higher-power photomicrograph of myocardium showing cellular accumulations--Aschoff bodies (arrows)--within the interstitium of the myocardium. These are found especially around blood vessels.
-
This is a higher-power photomicrograph of myocardium containing Aschoff bodies (arrows) within the interstitium.
-
This high-power photomicrograph of myocardium shows the cellular detail of an Aschoff body. Aschoff bodies are foci of fibrinoid necrosis surrounded by lymphocytes, macrophages, an occasional plasma cell, and plump “activated” histiocytes called Anitschkow cells or Aschoff cells (arrows). These distinctive cells have abundant amphophilic cytoplasm and central round-to-ovoid nuclei in which the chromatin is disposed in a central, slender, wavy ribbon resembling a caterpillar (hence the designation “caterpillar cells”).
-
This high-power photomicrograph of myocardium shows the cellular detail of another Aschoff body. In this case there appears to be a multinucleated Aschoff giant cell (arrow).
Histopathology
The Heart in Toxoplasma Gondii Myocarditis
<youtube v=2s9OuW9XlUw/>
The Heart in Coxsackie B2 Myocarditis
<youtube v=R_7AXF61QGg/>
References
- ↑ Feldman AM, McNamara D (2000). "Myocarditis". N Engl J Med. 343 (19): 1388–98. doi:10.1056/NEJM200011093431908. PMID 11070105.
- ↑ Heart Failure Society of America. Lindenfeld J, Albert NM, Boehmer JP, Collins SP, Ezekowitz JA; et al. (2010). "HFSA 2010 Comprehensive Heart Failure Practice Guideline". J Card Fail. 16 (6): e1–194. doi:10.1016/j.cardfail.2010.04.004. PMID 20610207.
- ↑ Chow LC, Dittrich HC, Shabetai R (1988). "Endomyocardial biopsy in patients with unexplained congestive heart failure". Ann Intern Med. 109 (7): 535–9. PMID 3421562.
- ↑ Mason JW, O'Connell JB, Herskowitz A, Rose NR, McManus BM, Billingham ME; et al. (1995). "A clinical trial of immunosuppressive therapy for myocarditis. The Myocarditis Treatment Trial Investigators". N Engl J Med. 333 (5): 269–75. doi:10.1056/NEJM199508033330501. PMID 7596370.
- ↑ Mahrholdt H, Goedecke C, Wagner A, Meinhardt G, Athanasiadis A, Vogelsberg H; et al. (2004). "Cardiovascular magnetic resonance assessment of human myocarditis: a comparison to histology and molecular pathology". Circulation. 109 (10): 1250–8. doi:10.1161/01.CIR.0000118493.13323.81. PMID 14993139.
- ↑ Hauck AJ, Kearney DL, Edwards WD (1989). "Evaluation of postmortem endomyocardial biopsy specimens from 38 patients with lymphocytic myocarditis: implications for role of sampling error". Mayo Clin Proc. 64 (10): 1235–45. PMID 2593714.
- ↑ Cooper LT, Berry GJ, Shabetai R (1997). "Idiopathic giant-cell myocarditis--natural history and treatment. Multicenter Giant Cell Myocarditis Study Group Investigators". N Engl J Med. 336 (26): 1860–6. doi:10.1056/NEJM199706263362603. PMID 9197214.
- ↑ Aretz HT, Billingham ME, Edwards WD, Factor SM, Fallon JT, Fenoglio JJ; et al. (1987). "Myocarditis. A histopathologic definition and classification". Am J Cardiovasc Pathol. 1 (1): 3–14. PMID 3455232.
- ↑ Hunt SA, Abraham WT, Chin MH, Feldman AM, Francis GS, Ganiats TG; et al. (2009). "2009 focused update incorporated into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the International Society for Heart and Lung Transplantation". Circulation. 119 (14): e391–479. doi:10.1161/CIRCULATIONAHA.109.192065. PMID 19324966.
- ↑ 10.0 10.1 10.2 10.3 Cooper LT, Baughman KL, Feldman AM, Frustaci A, Jessup M, Kuhl U; et al. (2007). "The role of endomyocardial biopsy in the management of cardiovascular disease: a scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology". Circulation. 116 (19): 2216–33. doi:10.1161/CIRCULATIONAHA.107.186093. PMID 17959655.
- ↑ Wu LA, Lapeyre AC, Cooper LT (2001). "Current role of endomyocardial biopsy in the management of dilated cardiomyopathy and myocarditis". Mayo Clin Proc. 76 (10): 1030–8. PMID 11605687.
- ↑ Magnani JW, Dec GW (2006). "Myocarditis: current trends in diagnosis and treatment". Circulation. 113 (6): 876–90. doi:10.1161/CIRCULATIONAHA.105.584532. PMID 16476862.
- ↑ 13.0 13.1 Yilmaz A, Kindermann I, Kindermann M, Mahfoud F, Ukena C, Athanasiadis A; et al. (2010). "Comparative evaluation of left and right ventricular endomyocardial biopsy: differences in complication rate and diagnostic performance". Circulation. 122 (9): 900–9. doi:10.1161/CIRCULATIONAHA.109.924167. PMID 20713901.
- ↑ Deckers JW, Hare JM, Baughman KL (1992). "Complications of transvenous right ventricular endomyocardial biopsy in adult patients with cardiomyopathy: a seven-year survey of 546 consecutive diagnostic procedures in a tertiary referral center". J Am Coll Cardiol. 19 (1): 43–7. PMID 1729344.
- ↑ Holzmann M, Nicko A, Kühl U, Noutsias M, Poller W, Hoffmann W; et al. (2008). "Complication rate of right ventricular endomyocardial biopsy via the femoral approach: a retrospective and prospective study analyzing 3048 diagnostic procedures over an 11-year period". Circulation. 118 (17): 1722–8. doi:10.1161/CIRCULATIONAHA.107.743427. PMID 18838566.