Treprostinil (extended-release tablet)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Turky Alkathery, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Treprostinil (extended-release tablet) is a prostacyclin vasodilator that is FDA approved for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to improve exercise capacity. Common adverse reactions include headache, nausea, and diarrhea.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
Pulmonary Arterial Hypertension
- Treprostinil is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to improve exercise capacity. The study that established effectiveness included predominately patients with WHO functional class II-III symptoms and etiologies of idiopathic or heritable PAH (75%) or PAH associated with connective tissue disease (19%).
- When used as the sole vasodilator, the effect of treprostinil on exercise is about 10% of the deficit, and the effect, if any, on a background of another vasodilator is probably less than this. Treprostinil is probably most useful to replace subcutaneous, intravenous, or inhaled treprostinil, but this use has not been studied.
Dosage
Recommended Dosing
- Individualize dosing of treprostinil according to clinical response.
- Take treprostinil with food. Swallow treprostinil tablets whole; do not crush, split, or chew.
- The recommended starting dose of treprostinil is 0.25 mg twice daily (BID) with food, taken approximately 12 hours apart or 0.125 mg three times daily (TID) with food, taken approximately 8 hours apart. Increase the dose as tolerated to achieve optimal clinical response. The recommended increment is 0.25 or 0.5 mg BID or 0.125 mg TID every 3-4 days. If dose increments are not tolerated consider titrating slower.
- The maximum dose is determined by tolerability. The mean dose in a controlled clinical trial at 12 weeks was 3.4 mg BID. Maximum doses studied were 12 mg BID in the 12-week blinded study and up to 21 mg BID in an open-label long-term study.
- If intolerable pharmacologic effects occur, decrease the dose in increments of 0.25 mg. Avoid abrupt discontinuation.
Hepatic Impairment
- In patients with mild hepatic impairment (Child Pugh Class A) start at 0.125 mg BID with 0.125 mg BID dose increments every 3 to 4 days. Avoid use of treprostinil in patients with moderate hepatic impairment (Child Pugh Class B). Treprostinil is contraindicated in patients with severe hepatic impairment (Child Pugh Class C).
Concomitant Administration with CYP2C8 Inhibitors
- When co-administered with strong CYP2C8 inhibitors (e.g., gemfibrozil) the initial dose is 0.125 mg BID with 0.125 mg BID dose increments every 3 to 4 days.
Interruptions and Discontinuation
- If a dose of medication is missed, the patient should take the missed dose as soon as possible, with food. If a patient misses two or more doses, restart at a lower dose and re-titrate.
- In the event of a planned short-term treatment interruption for patients unable to take oral medications, consider a temporary infusion of subcutaneous or intravenous treprostinil. To calculate the total daily dose (mg) of treprostinil for the parenteral route divide the oral total daily dose by 5.
- When discontinuing treprostinil, reduce the dose in steps of 0.5 to 1 mg per day.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Treprostinil (extended-release tablet) in adult patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Treprostinil (extended-release tablet) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Safety and effectiveness in pediatric patients have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Treprostinil (extended-release tablet) in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Treprostinil (extended-release tablet) in pediatric patients.
Contraindications
- Severe hepatic impairment (Child Pugh Class C).
Warnings
Worsening PAH Symptoms upon Abrupt Withdrawal
- Abrupt discontinuation or sudden large reductions in dosage of treprostinil may result in worsening of PAH symptoms.
Risk of Bleeding
- Treprostinil inhibits platelet aggregation and increases the risk of bleeding.
Increased Exposure with Alcohol
- Do not take treprostinil with alcohol as release of treprostinil from the tablet may occur at a faster rate than intended.
Use in Patients with Blind-end Pouches
- The tablet shell does not dissolve. In patients with diverticulosis, treprostinil tablets can lodge in a diverticulum.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
- In a 12-week placebo-controlled monotherapy study (Study 1; WHO Group 1; functional class II-III), the most commonly reported adverse reactions that occurred in patients receiving treprostinil included: headache, nausea, and diarrhea. Table 1 lists the adverse reactions that occurred at a rate on treprostinil at least 5% higher than on placebo.
- Treprostinil patients in Table 1 for Study 1 (N = 151) had access to 0.25 mg tablets at randomization. Approximately 91% of such patients experienced an adverse reaction, but only 4% discontinued therapy for an adverse reaction (compared to 3% receiving placebo). The overall discontinuation rate for any reason was 17% for active and 14% for placebo.
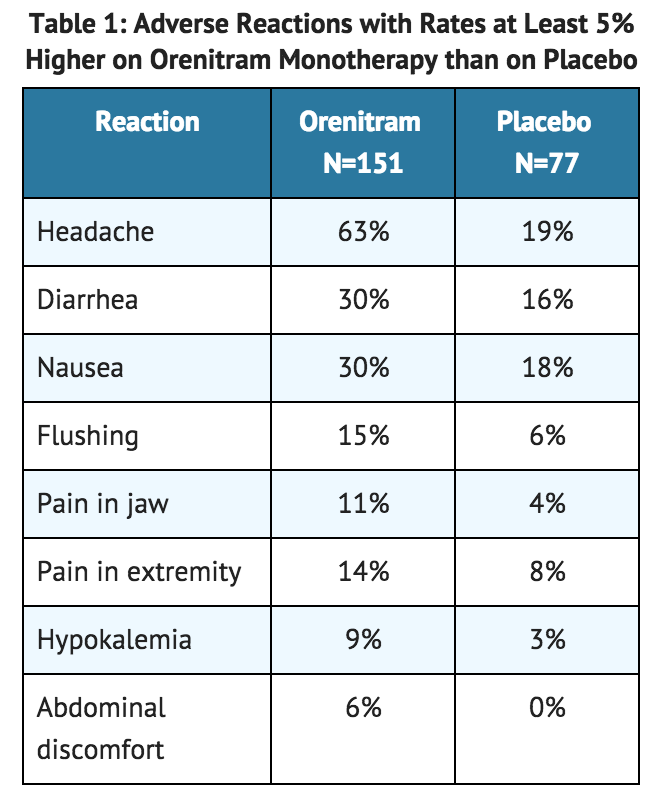
- Treprostinil was studied in a long-term, open-label extension study in which 824 patients were dosed for a mean duration of approximately 2 years. About 70% of patients continued treatment with treprostinil for at least a year. The mean dose was 4.2 mg BID at one year. The adverse reactions were similar to those observed in the placebo-controlled trials.
Postmarketing Experience
There is limited information regarding Treprostinil (extended-release tablet) Postmarketing Experience in the drug label.
Drug Interactions
Antihypertensive Agents or Other Vasodilators
- Concomitant administration of treprostinil with diuretics, antihypertensive agents or other vasodilators increases the risk of symptomatic hypotension.
Anticoagulants
- Treprostinil inhibits platelet aggregation; there is increased risk of bleeding, particularly among patients receiving anticoagulants.
Effect of CYP2C8 Inhibitors
- Co-administration of treprostinil and the CYP2C8 enzyme inhibitor gemfibrozil in healthy adult volunteers increases exposure to treprostinil. Reduce the starting dose of treprostinil to 0.125 mg BID and use 0.125 mg BID increments every 3 to 4 days.
Use in Specific Populations
Pregnancy
- Animal reproductive studies with treprostinil diolamine have shown an adverse effect on the fetus. There are no adequate and well-controlled studies in humans.
- In rats, treatment with treprostinil diolamine had no effect on reproductive performance or sperm motility at doses up to 10 mg/kg/day. The exposures at this dose level are about 10- (male) to 18- (female) fold the usual human exposure at the mean dose of 3.4 mg BID.
- In pregnant rats, reversible, dose-dependent decreases in body weight gain and food consumption were observed during the first four days of dosing in animals administered 10, 20 and 30 mg/kg/day treprostinil diolamine. In a dose range-finding study, there was a 17% decrease in the pregnancy rate in the animals administered 20 and 30 mg/kg/day. One dam in each of the 20 and 30 mg/kg/day had litters with no viable fetuses. In the definitive study (0, 5, 10 and 20 mg/kg/day), there were four treatment-related deaths, and a 32% decrease in the pregnancy rate for rats administered 20 mg/kg/day. There was an 8% decrease in the pregnancy rate in the animals administered 10 mg/kg/day. Across both studies, an increase in post-implantation loss was observed in animals administered 10 to 30 mg/kg/day, and a significant decrease in the mean number of live births was seen at dose levels ≥10 mg/kg/day. The no observed adverse effect level was 5 mg/kg/day (maternal, fetal viability and growth), and 20 mg/kg/day (teratogenicity), the highest dose tested in the definitive study. The exposures at 5 and 20 mg/kg/day doses represent 13 and 55 times, respectively, the human exposure.
- For F1 progeny, a decreased copulation index was observed at the 5 and 10 mg/kg/day treprostinil diolamine dose levels in rats. The no observed effect levels for physical development, reflex development, exploratory behavior, learning and memory, and sexual maturation was 10 mg/kg/day. The no observed effect level for F1 progeny general development (based on body weight) was 10 mg/kg/day for females and ≤ 2.5 mg/kg/day for males; the no observed effect level for F1 reproductive performance was 2.5 mg/kg/day or 6 times the human exposure.
- In pregnant rabbits, the primary maternal adverse effects were gastrointestinal disturbance; dose-dependent decreases in mean body weight, body weight gain, and food consumption were observed. During the post-dose phase, the effect was reversed. In a dose range-finding study, there was a 17% decrease in the pregnancy rate for animals administered 4 mg/kg/day. A dose-dependent increase in post-implantation loss was observed. Two dams administered 4 mg/kg/day had litters with no viable fetuses; the mean fetal weight was slightly decreased in animals administered 4 mg/kg/day. In the definitive study, mean fetal weights were significantly decreased in animals administered 0.5 to 3 mg/kg/day of treprostinil diolamine. At doses of 1.5 and 3 mg/kg/day, external fetal and soft tissue malformations were observed in a few fetuses, and the total fetal skeletal malformations were significantly increased. The no observed adverse effect level was less than 0.5 mg/kg/day (maternal), 1.5 mg/kg/day (fetal viability and growth), and 0.5 mg/kg/day (teratogenicity). The 0.5 mg/kg/day dose represents about 5 times the human exposure.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Treprostinil (extended-release tablet) in women who are pregnant.
Labor and Delivery
- The effect of treprostinil on labor and delivery in humans is unknown. No treprostinil treatment-related effects on labor and delivery were seen in animal studies.
Nursing Mothers
- It is not known whether treprostinil is excreted in human milk or absorbed systemically after ingestion. Because many drugs are excreted in human milk, choose treprostinil or breastfeeding.
Pediatric Use
- Safety and effectiveness in pediatric patients have not been established.
Geriatic Use
- Clinical studies of treprostinil did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently from younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic or cardiac function, and of concomitant disease or other drug therapy.
Gender
There is no FDA guidance on the use of Treprostinil (extended-release tablet) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Treprostinil (extended-release tablet) with respect to specific racial populations.
Renal Impairment
- No dose adjustments are required in patients with renal impairment. Treprostinil is not removed by dialysis.
Hepatic Impairment
- There is a marked increase in the systemic exposure to treprostinil in hepatically impaired patients.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Treprostinil (extended-release tablet) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Treprostinil (extended-release tablet) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral.
Monitoring
There is limited information regarding Treprostinil (extended-release tablet) Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Treprostinil (extended-release tablet) and IV administrations.
Overdosage
- Signs and symptoms of overdose with treprostinil during clinical trials reflect its dose-limiting pharmacologic effects and include severe headache, nausea, vomiting, diarrhea, and hypotension. Treat supportively.
Pharmacology
Mechanism of Action
- The major pharmacologic actions of treprostinil are direct vasodilation of pulmonary and systemic arterial vascular beds, inhibition of platelet aggregation, and inhibition of smooth muscle cell proliferation.
Structure
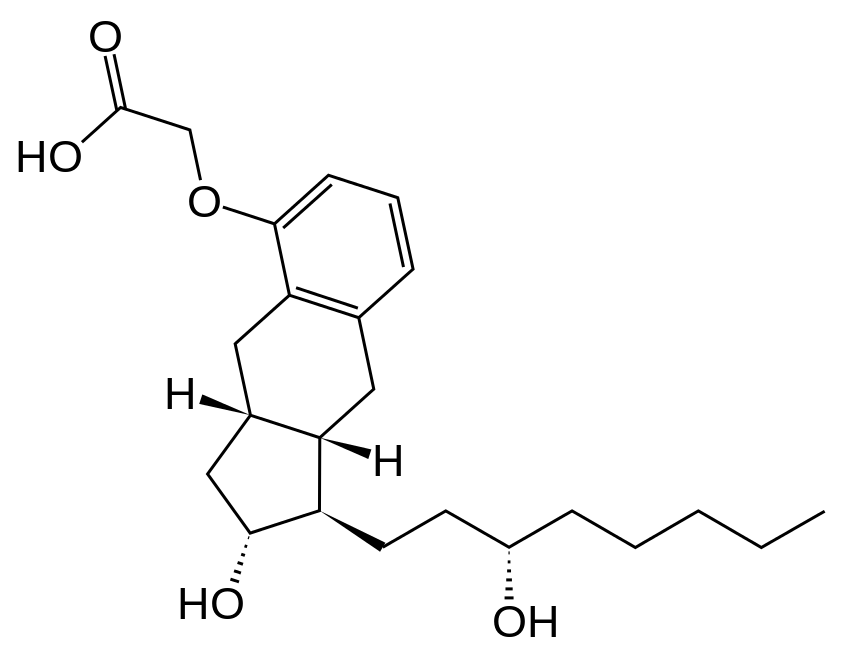
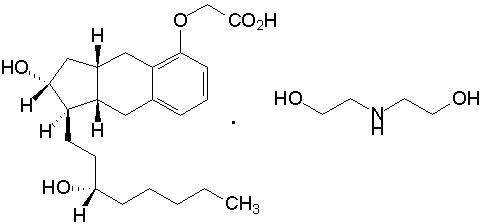
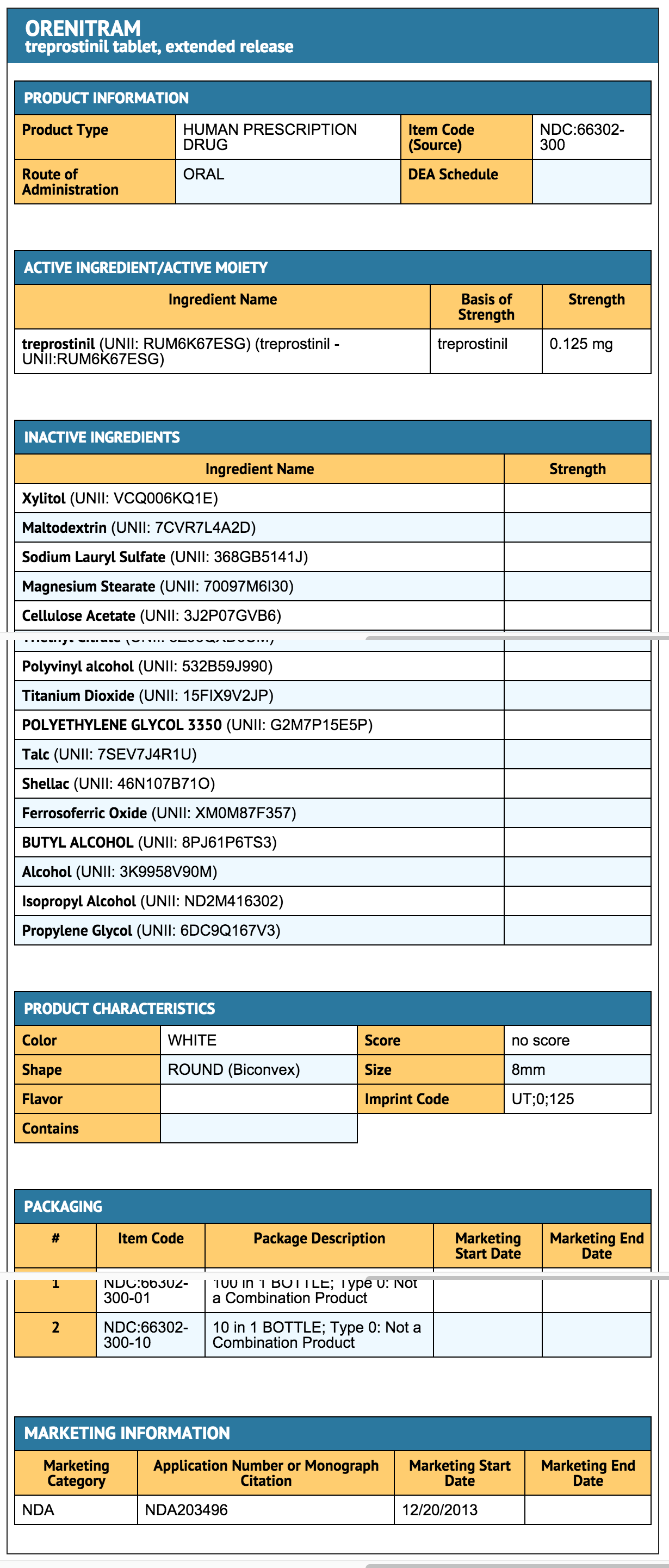
- Treprostinil is an extended release osmotic tablet for oral administration. Treprostinil is formulated as the diolamine salt of treprostinil, a tricyclic benzindene analogue of prostacyclin. The chemical name is Acetic acid, 2-(1R,2R,3aS,9aS)-2,3,3a,4,9,9a-hexahydro-2-hydroxy-1-[(3S)-3-hydroxyoctyl]-1H-benz[f]inden-5-yl]oxy]-, complexed with 2,2'-iminobis[ethanol] (1:1). The molecular formula is C23H34O5.C4H11NO2, the molecular weight is 495.65, and it has the following structural formula:
- Treprostinil tablets are formulated in four strengths, which contain 0.125 mg of treprostinil (equivalent to 0.159 mg treprostinil diolamine), 0.25 mg of treprostinil (equivalent to 0.317 mg treprostinil diolamine), 1 mg of treprostinil (equivalent to 1.27 mg treprostinil diolamine), or 2.5 mg of treprostinil (equivalent to 3.17 mg treprostinil diolamine). The formulations also contain xylitol, maltodextrin, sodium lauryl sulfate, magnesium stearate, cellulose acetate, triethyl citrate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, and talc. In addition tablets may contain colorants FD&C Blue #2, iron oxide yellow, and iron oxide red. The imprinting ink contains shellac glaze, ethanol, isopropyl alcohol USP, iron oxide black, n-butyl alcohol, propylene glycol, and ammonium hydroxide.
- Treprostinil is designed to release treprostinil at a near zero-order rate using an osmotic tablet technology. The tablet core is coated with a semi-permeable membrane and has a laser-drilled aperture through the membrane. Upon contact with water (e.g., after ingestion), the core tablet absorbs water through the semi-permeable membrane. The water dissolves the water-soluble treprostinil diolamine and the water-soluble osmotic excipients, which creates hydrostatic pressure within the membrane, eventually forcing the drug out through the tablet at a controlled rate.
Pharmacodynamics
- In a clinical trial of 240 healthy adult volunteers, single doses of inhaled treprostinil 54 µg (the target clinical dose) and 84 µg (supratherapeutic inhalation dose) prolonged the corrected QTc interval by approximately 10 msec. The QTc effect dissipated rapidly as the concentration of treprostinil decreased. Treprostinil has not been evaluated in a thorough QTc study.
Pharmacokinetics
- In patients with PAH, pharmacokinetics of treprostinil is dose-proportional for systemic exposure (AUC0-t) over the dose range of 0.5 and 15 mg BID. Upon repeat administration with a BID regimen, the accumulation in the systemic exposures to treprostinil is minimal and results in a peak-to-trough ratio of approximately 7. However, a TID regimen will reduce the peak-to-trough fluctuations to approximately 2.5 for the same total daily dose.
Absorption
- The absolute oral bioavailability of treprostinil is approximately 17%. Maximum treprostinil concentrations occur between approximately 4 and 6 hours following treprostinil administration.
- The absorption of treprostinil is affected by food. The AUCinf of treprostinil was increased by 49% and the Cmax was increased by an average of 13% when treprostinil was administered following a high-fat, high-calorie meal compared to fasting conditions in healthy volunteers. The relative bioavailability of treprostinil following oral administration of treprostinil 1 mg is not significantly altered by meal types ranging from 250 to 500 calories in healthy volunteers.
Distribution
- The treprostinil component of treprostinil is highly bound to human plasma proteins, approximately 96% over a treprostinil concentration range of 0.01-10 µg/mL.
Metabolism and Excretion
- In a study conducted in healthy volunteers using [14C] treprostinil, treprostinil was extensively metabolized on the side chain of the molecule via oxidation, oxidative cleavage, dehydration, and glucuronic acid conjugation. Treprostinil is primarily metabolized by CYP2C8 and to a lesser extent by CYP2C9. No new metabolites are found upon oral administration compared to parenteral administration of treprostinil. Only 1.13% and 0.19% is excreted as unchanged parent drug in the feces and urine, respectively. Based on in vitro studies treprostinil does not inhibit or induce major CYP enzymes.
Special Populations
- hepatic impairment: In subjects with mild (n=8) hepatic impairment, administration of a single 1 mg dose of treprostinil resulted in a mean Cmax and an AUC0-inf that were 1.6- and 2.1-fold, respectively values seen in healthy subjects. With moderate impairment (n=8), the corresponding ratios were 4.0- and 4.8-fold, and with severe impairment (n=6), they were 4.8- and 7.6-fold.
- Renal Impairment: In patients with severe renal impairment requiring dialysis (n=8), administration of a single 1 mg dose of treprostinil pre- and post-dialysis resulted in an AUC 0-inf that was not significantly altered compared to healthy subjects.
Drug Interactions
- Results of drug interaction studies are shown in Figure 1. Only for the strong CYP2C8 inhibitor does the interaction affect dosing.
Figure 1: Impact of Co-Administered Drugs on the Systemic Exposure of Treprostinil 1 mg Compared to treprostinil Administered Alone
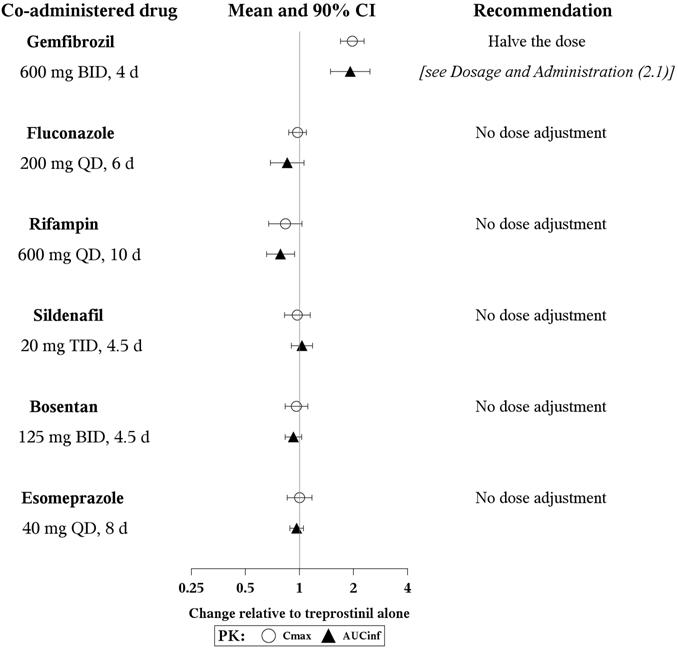
- Warfarin: A drug interaction study was carried out with Remodulin co-administered with warfarin (25 mg/day) in healthy volunteers. There was no clinically significant effect of either medication on the pharmacokinetics of treprostinil. Additionally, treprostinil did not affect the pharmacokinetics or pharmacodynamics of warfarin. The pharmacokinetics of R- and S- warfarin and the international normalized ratio (INR) in healthy subjects given a single 25 mg dose of warfarin were unaffected by continuous subcutaneous infusion of treprostinil at an infusion rate of 10 ng/kg/min.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Oral administration of treprostinil diolamine to Tg.rasH2 mice at 0, 5, 10 and 20 mg/kg/day in males and 0, 3, 7.5 and 15 mg/kg/day in females daily for 26 weeks did not significantly increase the incidence of tumors. The exposures obtained at the highest dose levels used in males and females are about 8- and 17-fold, respectively, the human exposure at the mean dose of 3.4 mg BID.
- In vitro genotoxicity studies with high doses of treprostinil did not demonstrate any mutagenic or clastogenic effects. Treprostinil diolamine was tested in vivo in a rat micronucleus assay and did not induce an increased incidence of micronucleated polychromatic erythrocytes.
- No adverse effect doses for fertility, fetal viability / growth, fetal development (teratogenicity), and postnatal development were determined in rats. In pregnant rabbits, external fetal and soft tissue malformations and fetal skeletal malformation occurred with the no observed adverse effect level for these adverse effects of 0.5 mg/kg/day (5 times the human exposure).
Clinical Studies
Clinical Trials in Pulmonary Arterial Hypertension (PAH)
- Three multi-center, randomized, double-blind studies were conducted and compared treprostinil to placebo in a total of 349 (Study 1), 350 (Study 2), and 310 (Study 3) patients with PAH.
- Study 1 (effect seen with no background vasodilator)
- Study 1 was a 12-week, randomized (2:1 treprostinil to placebo), double-blind, placebo-controlled, international efficacy and safety study of treprostinil in patients with WHO Group 1 PAH not currently receiving PAH therapy. The primary efficacy endpoint was placebo-corrected change in six-minute walk distance (6MWD) from Baseline to Week 12. Study drug dose was titrated to a maximum of 12 mg BID based on clinical response and study drug tolerability. Study 1 enrolled 349 patients (overall analysis population) who were not receiving any PAH medication. At the beginning of the study, subjects were dosed with only the 1 mg tablets with 0.5 and 0.25 mg tablets introduced at sequentially later dates during the study. The primary analysis population consisted of the 228 patients who had access to the 0.25 mg tablet at the time of randomization. Patients were administered treprostinil or placebo twice daily, with the doses titrated to effect over the course of the 12-week trial. Patients were in WHO functional class II (~33%) and class III (~66%) with either idiopathic or heritable PAH (~75%), collagen vascular disease associated PAH (~19%), or PAH associated with HIV (1%) or congenital heart defect (5%) or other conditions (~6%). The patients' mean baseline 6MWD was approximately 330 meters. In the primary analysis population, 17% of patients discontinued treprostinil compared to 14% of patients on placebo.
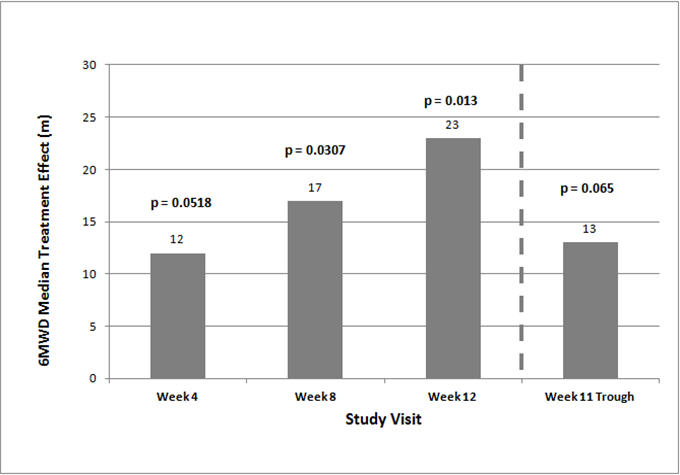
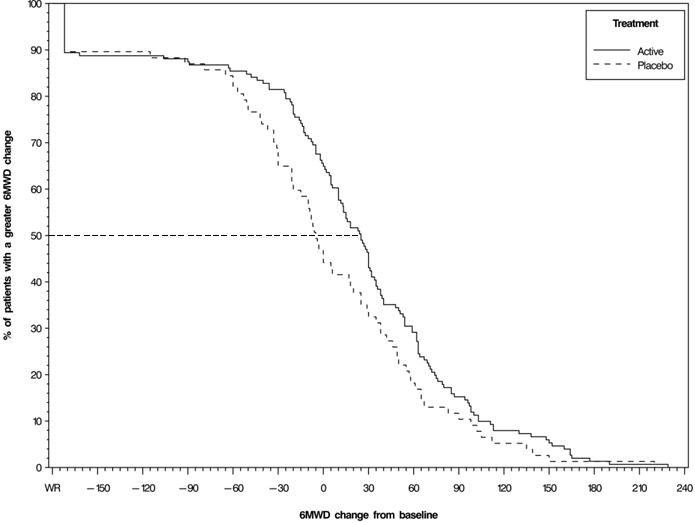
- The primary efficacy endpoint of the trial was the change in 6MWD at 12 weeks for the primary analysis population. Analysis of Study 1 results demonstrated that those patients receiving treprostinil compared to patients receiving placebo improved their median 6MWD by approximately +23 meters (Hodges-Lehmann estimate; p=0.013, non-parametric analysis of covariance in accordance with the pre-specified statistical analysis plan) as compared to patients receiving placebo as demonstrated in (Figure 2). The within group median change from baseline was +25 meters for treprostinil and -5 meters for placebo at week 12 (N=228). Mean dose (±SD) in the treprostinil group was 2.3 ± 1.3, 3.2 ± 1.9, and 3.4 ± 1.9 mg BID at Weeks 4, 8, and 12, respectively. The distribution of the 6MWD change from baseline at Week 12 was also plotted across the range of observed values (Figure 3).
- Figure 2: Hodges-Lehmann Estimate of Treatment Effect by Visit for the Primary Analysis Population (Study 1)
- Figure 3: Plot of the Distribution of Peak 6MWD Changes at Week 12 for the Primary Analysis Population (Study 1)
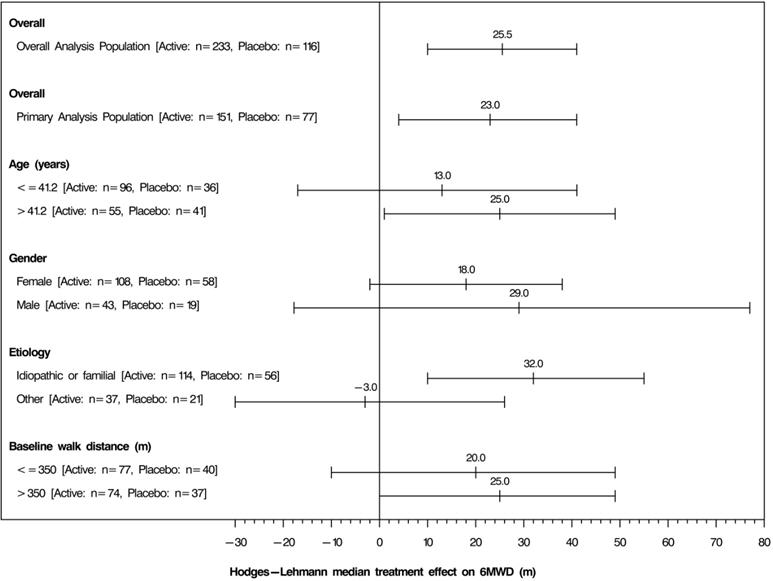
- The placebo-corrected median treatment effect on 6MWD was estimated (using the Hodges Lehmann estimator) within various subpopulations defined by age, gender, disease etiology, and baseline 6MWD (Figure 4).
Figure 4: Placebo Corrected Median Treatment Effect (Hodges-Lehmann estimate with 95% CI) on 6MWD Change from Baseline at Week 12 for Various Subgroups in the Primary Analysis Population (Study 1)
Studies 2 and 3 (no effect on a background of ERA, PDE5 inhibitor, or both)
- Studies 2 (N=350) and 3 (N=310) were 16-week, randomized, double-blind, placebo-controlled, international efficacy and safety studies of treprostinil in patients with WHO Group 1 PAH. The primary efficacy endpoint was placebo-corrected change in 6MWD from Baseline to Week 16. Patients were in WHO functional class II (~23%) and class III (~77%) with either idiopathic or heritable PAH (~66%), collagen vascular disease associated PAH (~29%), or PAH associated with HIV (1%) or congenital heart defect (4%). The patients' mean baseline 6MWD was approximately 340 meters. Approximately 40% were receiving both an ERA and a PDE5 inhibitor. The results did not demonstrate a benefit in exercise testing with median 6MWD at Week 16 (11 meters [Hodges-Lehmann estimate; p=0.072] and 10 meters [Hodges-Lehmann estimate; p=0.089], respectively).
Long-Term Treatment of Pulmonary Hypertension
- Patients (N=824) from the placebo-controlled studies entered a long-term, uncontrolled, open-label extension study. The average exposure to treprostinil was approximately 2 years with a maximum exposure of approximately 6 years. The dose of treprostinil continued to increase over time with doses (mean ± SD) of 3.6 ± 2.7, 4.2 ± 3.1, and 5 ± 3.7 mg BID at 6 (n=649), 12 (n=433), and 24 months (n=238), respectively. Reasons for discontinuation from the study included adverse event (16%), progression of disease (15%), death (13%), and withdrawn consent (7%). In the 522 subjects that completed the 12-month efficacy assessment, their mean 6MWD improved by 24 meters compared to baseline (30 meters in monotherapy patients and 20 meters when treprostinil was used in combination with an ERA and/or a PDE-5 inhibitor). Of the patients that remained in the study, overall survival was 92%, 87%, and 82% at the end of 1, 2, and 3-years, respectively, with progression-free survival (progression defined as death, discontinuation or addition of a PAH therapy) of 74%, 61%, and 47%. Without a control group, these data must be interpreted cautiously.
How Supplied
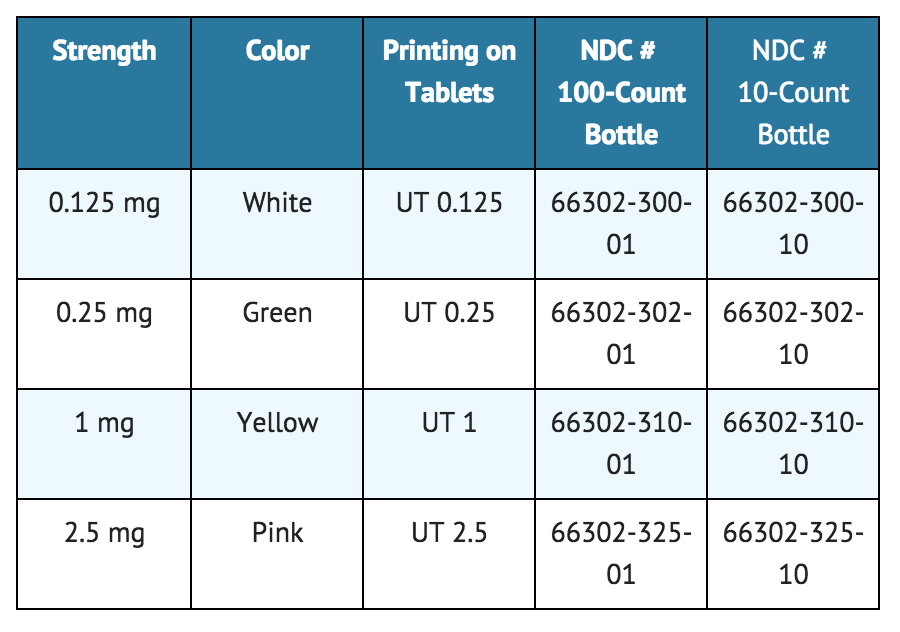
- Orenitram is an 8 mm round biconvex tablet with strength identifying color and printing and supplied as follows:
Storage
- Store at 25°C (77°F); excursions 15°C to 30°C (59°F to 86°F). Keep out of reach of children.
Images
Drug Images
{{#ask: Page Name::Treprostinil (extended-release tablet) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel




{{#ask: Label Page::Treprostinil (extended-release tablet) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Tell patients:
- Abrupt discontinuation of therapy could result in worsening of PAH symptoms.
- Take Orenitram with food.
- Swallow Orenitram tablets whole. Do not split, chew, crush, or break. Do not take a tablet that is :*damaged or broken.
- The biologically inert components of the tablet remain intact during gastrointestinal transit and are :*eliminated in the feces as an insoluble shell.
- Do not take Orenitram with alcohol.
Precautions with Alcohol
Increased Exposure with Alcohol
- Do not take Orenitram with alcohol as release of treprostinil from the tablet may occur at a faster rate than intended.
Brand Names
- ORENITRAM®[1]
Look-Alike Drug Names
- There is limited information regarding Look-Alike Drug Names.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.