Toxic multinodular goiter pathophysiology
|
Toxic multinodular goiter Microchapters |
|
Differentiating Toxic multinodular goiter from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Toxic multinodular goiter pathophysiology On the Web |
|
American Roentgen Ray Society Images of Toxic multinodular goiter pathophysiology |
|
Risk calculators and risk factors for Toxic multinodular goiter pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sunny Kumar MD [2]
Overview
The progression to Toxic multinodular goiter usually involves the somatic gain-of-function mutations in the TSH receptor in autonomously functioning thyroid nodules.
Pathophysiology
Thyroid physiology:
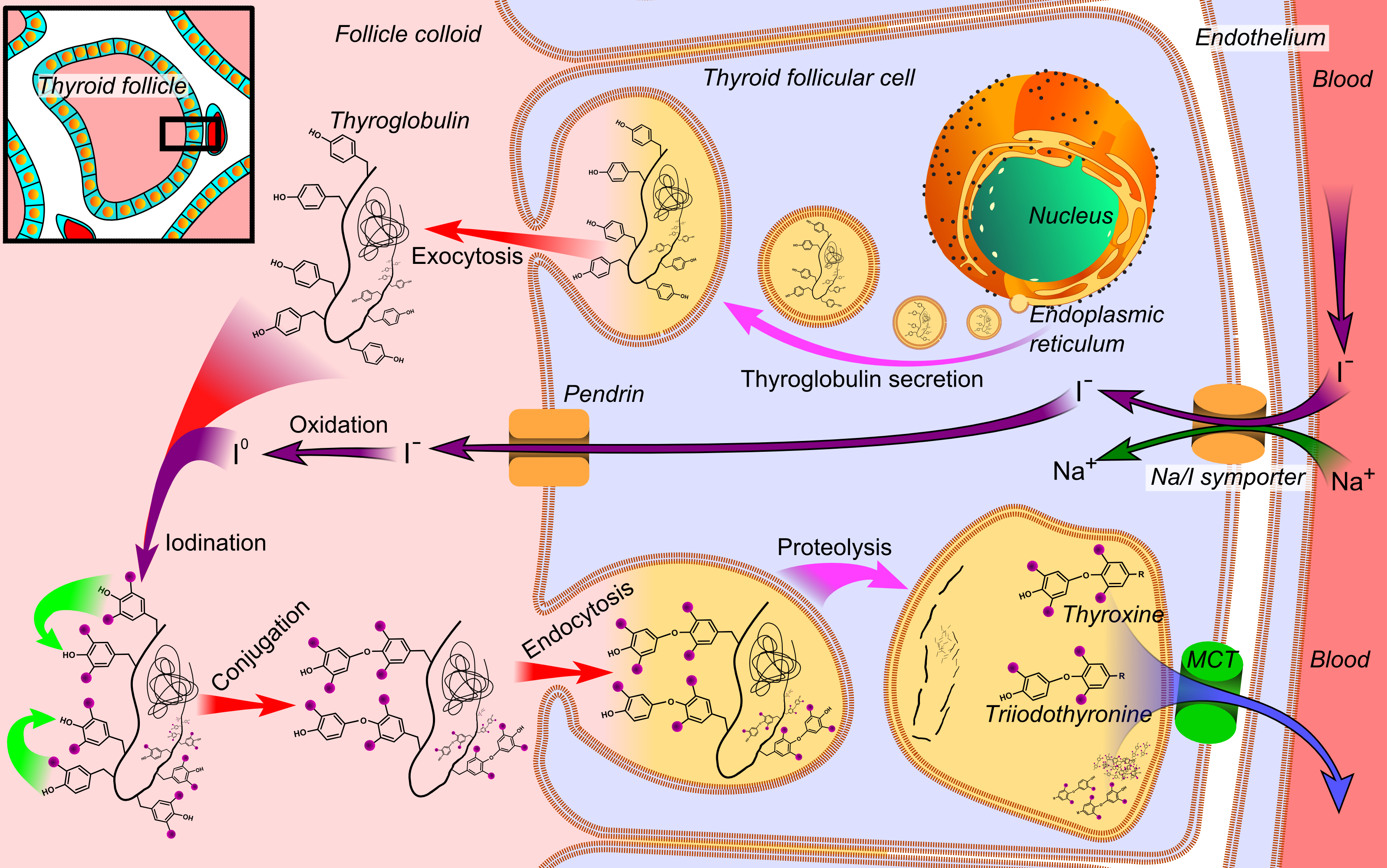
- The tissue of the thyroid gland is composed mostly of thyroid follicles.
- The follicles are made up of a central cavity filled with a sticky fluid called colloid surrounded by a wall of epithelial follicle cells, the colloid is the center of thyroid hormone production, and that production is dependent on the hormones’ essential and unique component: iodine.
- Thyroid is an endocrine gland which synthesizes and secretes thyroid hormones into the bloodstream directly.
- The thyroid gland is regulated by hypothalamus and pituitary gland.
- Thyroid hormones are of two biochemical structures. Triiodothyronine (T3), which is true and potent form and its pro-hormone, thyroxine (T4) majorly is secretory form later converted to T3 in peripheral tissues by deiodinase enzyme.
- Thyroid hormone has negative feedback on thyroid receptors located on hypothalamus and pituitary gland.
- Thyroid hormones majorly affects every part of body and maintains metabolic rate by acting on thyroid receptors which are nuclear receptors mediating gene expression.
- Functional unit of thyroid gland is thyroid follicles, which are aliened in continuous circular form forming hallow cavity between them called thyroid cavity. On basal side of thyroid follicle is connective tissue containing blood vessels for transport of thyroid hormone and blood cells and iodine. Apical side of thyroid follicle faces toward thyroid cavity where it has TPO enzymes located, which help in conversion of iodide to iodine. Iodine is organified to tyrosine residue of thyroglobin, which is synthesized and stored in thyroid follicle cavity. It forms mono-idodo or di-iodo thyroglobin and then they combine to form tri-iodo or trata-iodo thyroglobin. On demand of body thyroglobin goes in proteolysis and release T3,T4 in blood stream across thyroid follicle.
- At high magnification the cell surface lining the follicle is rich in microvilli that project into the follicular lumen where the colloid is secreted; hormones are secreted into the blood at the opposite basal cell pole adjoining the rich capillary net.
- TSH is heterodimeric cystine-knot glycoproteins consisting a unique β-subunit, which provides biological specificity to TSH receptor and secreted by the basophilic thyrotropes in anteroir pitutary gland.
- TSH receptor (TSH-R) which is found on basolateral surface of thyroid follicular cells, belongs to a rhodopsin/β-adrenergic receptor family in the seven-transmembrane domain, GPCR superfamily.[1]
- TSH-R is composed of 764 amino acids, plays important role in thyroid gland function and growth.[2]
- TSH-R gene is found on chromosome 14q31[3]
- TSH-R can be found on other parts of body such as lymphocytes, adipocytes, retroocular fibroblasts, neuronal cells, and astrocytes[4]
- TSH-R dependent interleukin-2 activation of cytotoxic lymphocytes has been associated with pathogenesis of many autoimmune thyroid disease.[5]
- This leads to activation of cAMP and formation of PIP2 and Ca ++.
- Iodide uptake, TPO, and TG synthesis occurs via the TSH-induced cAMP signal
- TSH-induced PIP2 cascade is playing role in iodide eflux, hydrogen peroxide generation, and iodination[6]
- TSH-R is divided in two parts. extracellular long hydrophillic end followed small cytoplasmic by seven loops of hydrophobic end which has a phosphrylantion site.
- Deletion of the signal peptide in the TSHR prevents its in situ processing and glycosylation.[7]

Pathogenesis
- The progression to Toxic multinodular goiter usually involves the somatic gain-of-function mutations in the TSH receptor.
- More than 30 different activating mutations causing nonautoimmune hyperthyroidism have been found which includes mutation in toxic nodule and multi nodular toxic goiter.
- “Two-State” Model of TSH-R explains that there are three states of TSH-R, [8]
- A: “closed” (inactive) TSHR conformation
- B: “opened” (unliganded) TSHR conformation.
- C: “opened” (hormone-activated) TSHR conformation.
- Activating mutations in the TSH-R genes may lead to increase of basal and TSH-induced activation of cAMP or inositol phosphate (IP) cascades which keep TSH-R[9] in opened unliganded or hormone-activated state. it is called somatic gain-of-function mutations.[10]
- Studies have shown that the solitary toxic nodule may have same pathogenesis in multi nodular toxic goiter.[11]
- Different nodules in multi-nodular goiter may have many hyperfunctioning nodules with different gain-in -function mutations which may be due different clonal origin.[12]
- There are studies which have shown that two toxic nodules in same multi nodular goiter may have same I630L mutations as well.[13]
- Follicles of tissue surrounding autonomous hyper functioning nodule lacks mutation so they go in superseded supply of iodine and TSH stimulation hence leads to reduced radio-iodine uptake on radio logic studies.
- Such wild type mutations are called constitutively active mutant receptor “CAMs”
- CAMs show higher activation of adenyl cyclase pathway as normal TSH concentration by Gs activation and higher activation of PIP2/ Ca2+ pathway at lower than median concentration of TSH by Gq activation.
- The positions and structural fragments of the wild type receptor plays a role in physiological coupling to both Gs and Gq.[14]
- Patients with multi-nodular goiter can have both hyperfunctioning and non-functioning nodules.
- CAMs are found on follicles of only hyper-functioning nodules which confirms the role of mutation in multi-nodular goiter.[15]
- In multi-nodular goiter two sets of hyper-functioning tissue populations can be found.
- There are two hyperfunctioning hyperplastic nodules both of which may have same genetic mutations.[16]
- In patients with Toxic multinodular goiter TSH is decreased and T3 and T4 is found in increased levels in serum.
Genetics
- The development of multi-nodular goiter is the result of multiple genetic base substitution mutations on exon 10 of chromosome 14q31
- Methionine replacing an isoleucine (I486M) in the first extracellular loop of TSH-R G-protien[17]
- Threonine in position 632 (T632I) sixth transmembrane segment of TSH-R G-protien[18]
- Less common mutations are given below:
| TSHr gene codon | Base substitution | Amino acid change |
|---|---|---|
| 486 | ATC/ATG | lle/Met |
| 619 | GAT/GGT | Asp/Gly |
| 623 | GCC/GTC | Ala/Val |
| 629 | TTG/TTT | Leu/Phe |
| 630 | ATC/CTC | Ile/Leu |
| 632 | ACC/ATC | Thr/Ile |
| 633 | GAC/GAG | Asp/Glu |
| 639 | CCA/TCA | Pro/Ser |
Associated Conditions
Gross Pathology
Some of the gross pathology analysis characteristic of multinodular goiter include:
- Enlarged thyroid gland
- Multiple nodules with fibrosis
- Hemorrhages and dystrophic calcification.
Microscopic Pathology
Some of the microscopic histopathological analysis characteristic of multinodular goiter include:
- Cubical or cylindrical epithelium
- Resorption vesicles in the colloid
- Discrete fibrous capsule
- Secondary nodules
- Co-existing encapsulated adenomatous nodules with degenerative changes of fibrosis
- Calcification and hemorrhage
References
- ↑ Kohn LD, Shimura H, Shimura Y, Hidaka A, Giuliani C, Napolitano G; et al. (1995). "The thyrotropin receptor". Vitam Horm. 50: 287–384. PMID 7709602.
- ↑ Graves PN, Davies TF (2000). "New insights into the thyroid-stimulating hormone receptor. The major antigen of Graves' disease". Endocrinol Metab Clin North Am. 29 (2): 267–86, vi. PMID 10874529.
- ↑ Rousseau-Merck MF, Misrahi M, Loosfelt H, Atger M, Milgrom E, Berger R (1990). "Assignment of the human thyroid stimulating hormone receptor (TSHR) gene to chromosome 14q31". Genomics. 8 (2): 233–6. PMID 2249847.
- ↑ Paschke R, Geenen V (1995). "Messenger RNA expression for a TSH receptor variant in the thymus of a two-year-old child". J Mol Med (Berl). 73 (11): 577–80. PMID 8751142.
- ↑ Abe E, Marians RC, Yu W, Wu XB, Ando T, Li Y; et al. (2003). "TSH is a negative regulator of skeletal remodeling". Cell. 115 (2): 151–62. PMID 14567913.
- ↑ Giuliani C, Cerrone D, Harii N, Thornton M, Kohn LD, Dagia NM; et al. (2012). "A TSHr-LH/CGr chimera that measures functional TSAb in Graves' disease". J Clin Endocrinol Metab. 97 (7): E1106–15. doi:10.1210/jc.2011-2893. PMID 22496495.
- ↑ Akamizu T, Kosugi S, Kohn LD (1990). "Thyrotropin receptor processing and interaction with thyrotropin". Biochem Biophys Res Commun. 169 (3): 947–52. PMID 2114112.
- ↑ Duprez L, Parma J, Costagliola S, Hermans J, Van Sande J, Dumont JE; et al. (1997). "Constitutive activation of the TSH receptor by spontaneous mutations affecting the N-terminal extracellular domain". FEBS Lett. 409 (3): 469–74. PMID 9224711.
- ↑ Tonacchera M, Van Sande J, Cetani F, Swillens S, Schvartz C, Winiszewski P; et al. (1996). "Functional characteristics of three new germline mutations of the thyrotropin receptor gene causing autosomal dominant toxic thyroid hyperplasia". J Clin Endocrinol Metab. 81 (2): 547–54. doi:10.1210/jcem.81.2.8636266. PMID 8636266.
- ↑ Kleinau G, Neumann S, Grüters A, Krude H, Biebermann H (2013). "Novel insights on thyroid-stimulating hormone receptor signal transduction". Endocr Rev. 34 (5): 691–724. doi:10.1210/er.2012-1072. PMC 3785642. PMID 23645907.
- ↑ Duprez L, Hermans J, Van Sande J, Dumont JE, Vassart G, Parma J (1997). "Two autonomous nodules of a patient with multinodular goiter harbor different activating mutations of the thyrotropin receptor gene". J Clin Endocrinol Metab. 82 (1): 306–8. doi:10.1210/jcem.82.1.3691. PMID 8989278.
- ↑ Duprez L, Hermans J, Van Sande J, Dumont JE, Vassart G, Parma J (1997). "Two autonomous nodules of a patient with multinodular goiter harbor different activating mutations of the thyrotropin receptor gene". J Clin Endocrinol Metab. 82 (1): 306–8. doi:10.1210/jcem.82.1.3691. PMID 8989278.
- ↑ Holzapfel HP, Führer D, Wonerow P, Weinland G, Scherbaum WA, Paschke R (1997). "Identification of constitutively activating somatic thyrotropin receptor mutations in a subset of toxic multinodular goiters". J Clin Endocrinol Metab. 82 (12): 4229–33. doi:10.1210/jcem.82.12.4441. PMID 9398745.
- ↑ Parma J, Van Sande J, Swillens S, Tonacchera M, Dumont J, Vassart G (1995). "Somatic mutations causing constitutive activity of the thyrotropin receptor are the major cause of hyperfunctioning thyroid adenomas: identification of additional mutations activating both the cyclic adenosine 3',5'-monophosphate and inositol phosphate-Ca2+ cascades". Mol Endocrinol. 9 (6): 725–33. doi:10.1210/mend.9.6.8592518. PMID 8592518.
- ↑ Tonacchera M, Agretti P, Chiovato L, Rosellini V, Ceccarini G, Perri A; et al. (2000). "Activating thyrotropin receptor mutations are present in nonadenomatous hyperfunctioning nodules of toxic or autonomous multinodular goiter". J Clin Endocrinol Metab. 85 (6): 2270–4. doi:10.1210/jcem.85.6.6634. PMID 10852462.
- ↑ Tonacchera M, Vitti P, Agretti P, Giulianetti B, Mazzi B, Cavaliere R; et al. (1998). "Activating thyrotropin receptor mutations in histologically heterogeneous hyperfunctioning nodules of multinodular goiter". Thyroid. 8 (7): 559–64. doi:10.1089/thy.1998.8.559. PMID 9709907.
- ↑ Tonacchera M, Agretti P, Chiovato L, Rosellini V, Ceccarini G, Perri A; et al. (2000). "Activating thyrotropin receptor mutations are present in nonadenomatous hyperfunctioning nodules of toxic or autonomous multinodular goiter". J Clin Endocrinol Metab. 85 (6): 2270–4. doi:10.1210/jcem.85.6.6634. PMID 10852462.
- ↑ Tonacchera M, Agretti P, Chiovato L, Rosellini V, Ceccarini G, Perri A; et al. (2000). "Activating thyrotropin receptor mutations are present in nonadenomatous hyperfunctioning nodules of toxic or autonomous multinodular goiter". J Clin Endocrinol Metab. 85 (6): 2270–4. doi:10.1210/jcem.85.6.6634. PMID 10852462.