Fluconazole description
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ahmed Zaghw, M.D. [2]
Description
DIFLUCAN® (fluconazole), the first of a new subclass of synthetic triazole antifungal agents, is available as tablets for oral administration, as a powder for oral suspension.
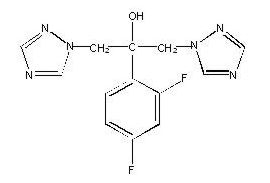
Fluconazole is designated chemically as 2,4-difluoro-α,α1-bis(1H-1,2,4-triazol-1-ylmethyl) benzyl alcohol with an empirical formula of C13H12F2N6O and molecular weight of 306.3. The structural formula is:
 |
Fluconazole is a white crystalline solid which is slightly soluble in water and saline.
DIFLUCAN Tablets contain 50, 100, 150, or 200 mg of fluconazole and the following inactive ingredients: microcrystalline cellulose, dibasic calcium phosphate anhydrous, povidone, croscarmellose sodium, FD&C Red No. 40 aluminum lake dye, and magnesium stearate.
DIFLUCAN for Oral Suspension contains 350 mg or 1400 mg of fluconazole and the following inactive ingredients: sucrose, sodium citrate dihydrate, citric acid anhydrous, sodium benzoate, titanium dioxide, colloidal silicon dioxide, xanthan gum, and natural orange flavor. After reconstitution with 24 mL of distilled water or Purified Water (USP), each mL of reconstituted suspension contains 10 mg or 40 mg of fluconazole. [1]
References
Adapted from the FDA Package Insert.