Carbonic anhydrase
| Template:Chembox header| Carbonic anhydrase | |
|---|---|
| Systematic name | carbonate hydrolyase |
| Other names | carbonate dehydratase; carbonate anhydrase; carbonic acid anhydrase |
| EC number | EC 4.2.1.1 |
| CAS number | 9001-03-0 |
| Template:Chembox header | Disclaimer and references | |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Carbonic anhydrase (carbonate dehydratase) is a family of metalloenzymes (enzymes that contain one or more metal atoms as a functional component of the enzyme) that catalyze the rapid conversion of carbon dioxide to bicarbonate and protons, a reaction that occurs rather slowly in the absence of a catalyst.[1] Carbonic anhydrase greatly increases the rate of the reaction, with typical catalytic rates of the different forms of this enzyme ranging between 104 and 106 reactions per second.[2] The active site of most carbonic anhydrases contains a zinc ion.
Structure and function of CA
Several forms of carbonic anhydrase occur in nature. In the best-studied α-carbonic anhydrase form present in animals, the zinc ion is coordinated by the imidazole rings of 3 histidine residues, His94, His96 and His119. The primary function of the enzyme in animals is to interconvert carbon dioxide and bicarbonate to maintain acid-base balance in blood and other tissues, and to help transport carbon dioxide out of tissues. Plants contain a different form called β-carbonic anhydrase, which, from an evolutionary standpoint, is a distinct enzyme, but participates in the same reaction and also uses a zinc ion in its active site. In plants, carbonic anhydrase helps raise the concentration of CO2 within the chloroplast in order to increase the carboxylation rate of the enzyme Rubisco. This is the reaction that integrates CO2 into organic carbon sugars during photosynthesis, and can use only the CO2 form of carbon, not carbonic acid or bicarbonate.
In 2000, a cadmium-containing carbonic anhydrase was found to be expressed in marine diatoms during zinc limitation. In the open ocean, zinc is often in such low concentrations that it can limit the growth of phytoplankton like diatoms; thus a carbonic anhydrase using a different metal ion would be beneficial in these environments. Before this discovery, cadmium has generally been thought of as a very toxic heavy metal without biological function. As of 2005, this peculiar carbonic anhydrase form hosts the only known beneficial cadmium-dependent biological reaction.
Reaction catalyzed by carbonic anhydrase:
- <math>\rm CO_2 + H_2O \rightarrow^{Carbonic\ anhydrase} HCO_3^- + H^+</math>[3] (in tissues - high CO2 concentration)
The reaction rate of carbonic anhydrase is one of the fastest of all enzymes, and its rate is typically limited by the diffusion rate of its substrates.
The reverse reaction is also relatively slow (kinetics in the 15-second range), which is why a carbonated drink does not instantly degas when opening the container, but will rapidly degas in one's mouth when carbonic anhydrase is added with saliva.
- <math>\rm HCO_3^- + H^+ \rightarrow H_2CO_3 \rightarrow CO_2 + H_2O</math> (in lungs and nephrons of the kidney - low CO2 concentration, in plant cells)
Mechanism
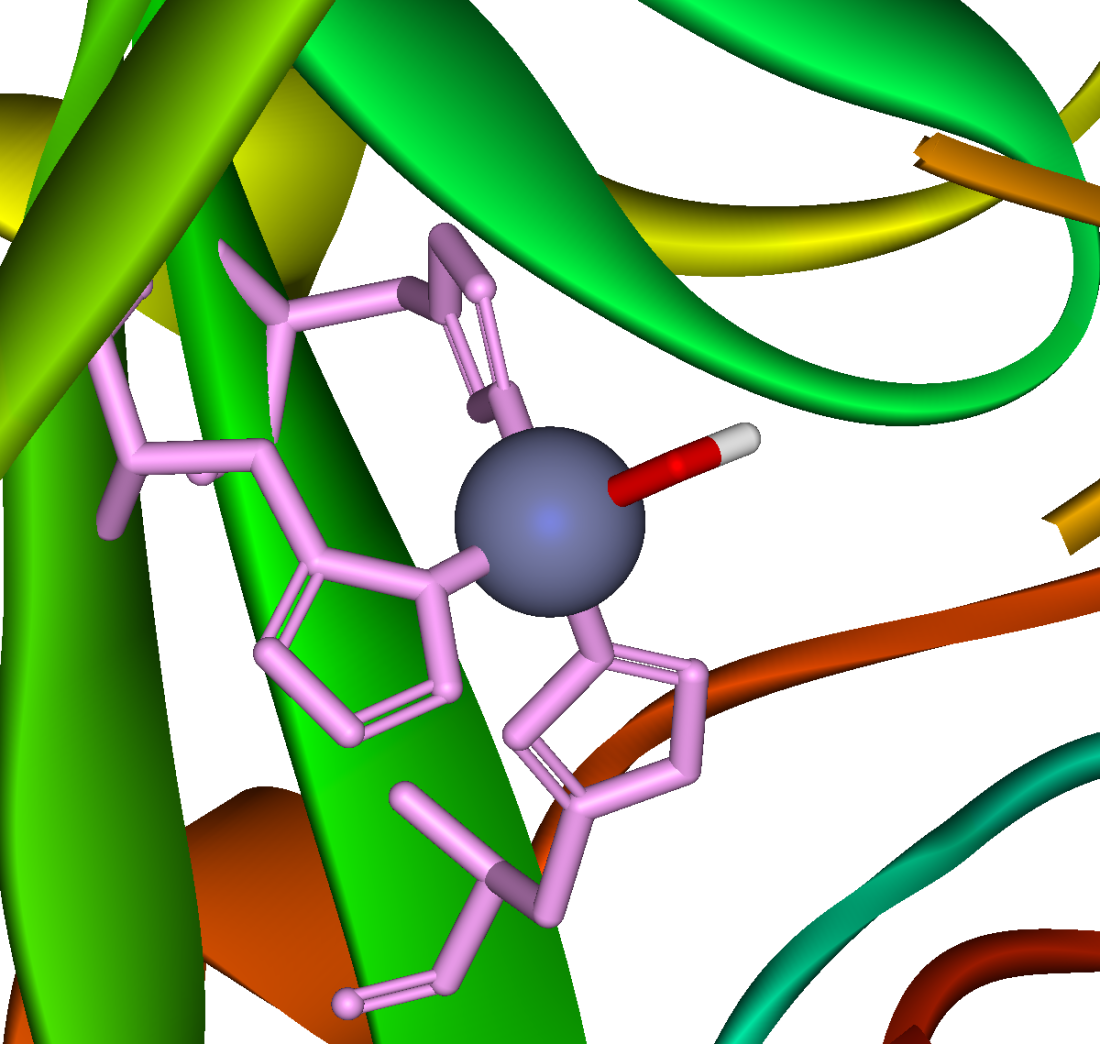
A zinc prosthetic group in the enzyme is coordinated in three positions by histidine side chains. The fourth coordination position is occupied by water. This causes polarisation of the hydrogen-oxygen bond, making the oxygen slightly more negative, thereby weakening it.
A fourth histidine is placed close to the substrate of water and accepts a proton, in an example of general acid-general base catalysis. This leaves a hydroxide attached to the zinc.
The active site also contains specificity pocket for carbon dioxide, bringing it close to the hydroxide group. This allows the electron rich hydroxide to attack the carbon dioxide, forming bicarbonate.
CA families

There are at least five distinct CA families (α, β, γ, δ and ε). These families have no significant amino acid sequence similarity and in most cases are thought to be an example of convergent evolution. The α-CAs are found in humans.
α-CA
The CA enzymes found in mammals are divided into four broad subgroups[4], which, in turn consist of several isoforms:
- the cytosolic CAs (CA-I, CA-II, CA-III, CA-VII and CA XIII) (Template:Gene2, Template:Gene2, Template:Gene2, Template:Gene2, Template:Gene2)
- mitochondrial CAs (CA-VA and CA-VB) (CA5A, CA5B)
- secreted CAs (CA-VI) (Template:Gene2)
- membrane-associated CAs (CA-IV, CA-IX, CA-XII, CA-XIV and CA-XV) (Template:Gene2, Template:Gene2, Template:Gene2, Template:Gene2)
There are three additional "acatalytic" CA isoforms (CA-VIII, CA-X, and CA-XI) (Template:Gene2, Template:Gene2, Template:Gene2) whose functions remain unclear.[5]
| Isoform | Gene | Molecular mass [6] | Location (cell) | Location (tissue) [6] | Relative activity [6] | Sensitivity to sulfonamides [6] | |
|---|---|---|---|---|---|---|---|
| CA-I | Template:Gene2 | 29 kDa | cytosol | RBCs and GI tract | 15% | high | |
| CA-II | Template:Gene2 | 29 kDa | cytosol | almost ubiquitous | 100% | high | |
| CA-III | Template:Gene2 | 29 kDa | cytosol | 8% of soluble protein in Type I muscle | 1% | low | |
| CA-IV | Template:Gene2 | 35 kDa | extracellularily GPI-linked | Widely distributed, e.g. acid-transporting epithelium | ~100% | moderate | |
| CA-VA | CA5A | mitochondria | |||||
| CA-VB | CA5B | mitochondria | secreting cells | ||||
| CA-VI | Template:Gene2 | ||||||
| CA-VII | Template:Gene2 | cytosol | |||||
| CA-IX | Template:Gene2 | cell membrane-associated | |||||
| CA-XII | Template:Gene2 | 44 kDa | extracellularily located active site | certain cancers | ~30% | ||
| CA XIII | Template:Gene2 | cytosol | |||||
| CA-XIV | Template:Gene2 | 54 kDa | extracellularily located active site | kidney, heart, skeletal muscle, brain | |||
| and CA-XV |
β-CA
Most prokaryotic and plant chloroplast CAs belong to the beta family. Two signature patterns for this family have been identified:
- C-[SA]-D-S-R-[LIVM]-x-[AP]
- [EQ]-[YF]-A-[LIVM]-x(2)-[LIVM]-x(4)-[LIVMF](3)-x-G-H-x(2)-C-G
γ-CA
The gamma class of CAs come from methane-producing bacteria that grow in hot springs.
δ-CA
The delta class of CAs has been described in diatoms. The distinction of this class of CA has recently[7] come into question, however.
ε-CA
The epsilon class of CAs occurs exclusively in bacteria in a few chemolithotrophs and marine cyanobacteria that contain cso-carboxysomes.[8] Recent 3-dimensional analyses[7] suggest that ε-CA bears some structural resemblance to β-CA, particularly near the metal ion site. Thus, the two forms may be distantly related, even though the underlying amino acid sequence has since diverged considerably.
Pharmacological agents affecting CA
External links
- PDB Molecule of the Month Carbonic anhydrase
References
- ↑ Badger MR, Price GD (1994). "The role of carbonic anhydrase in photosynthesis". Annu. Rev. Plant Physiol. Plant Mol. Bio. 45: 369–392. doi:10.1146/annurev.pp.45.060194.002101.
- ↑ Lindskog S (1997). "Structure and mechanism of carbonic anhydrase". Pharmacol. Ther. 74 (1): 1–20. doi:10.1016/S0163-7258(96)00198-2. PMID 9336012.
- ↑ Carbonic acid has a pKa of around 6.36 (the exact value depends on the medium) so at pH 7 a small percentage of the bicarbonate is protonated. See carbonic acid for details concerning the equilibria HCO3- + H+ <math>\rightleftharpoons</math> H2CO3 and H2CO3 <math>\rightleftharpoons</math> CO2 + H2O
- ↑ Breton S (2001). "The cellular physiology of carbonic anhydrases". JOP. 2 (4 Suppl): 159–64. PMID 11875253.
- ↑ Lovejoy DA, Hewett-Emmett D, Porter CA, Cepoi D, Sheffield A, Vale WW, Tashian RE (1998). "Evolutionarily conserved, "acatalytic" carbonic anhydrase-related protein XI contains a sequence motif present in the neuropeptide sauvagine: the human CA-RP XI gene (CA11) is embedded between the secretor gene cluster and the DBP gene at 19q13.3". Genomics. 54 (3): 484–93. doi:10.1006/geno.1998.5585. PMID 9878252.
- ↑ 6.0 6.1 6.2 6.3 Unless else specified: Walter F., PhD. Boron. Medical Physiology: A Cellular And Molecular Approaoch. Elsevier/Saunders. ISBN 1-4160-2328-3. Page 638
- ↑ 7.0 7.1 Sawaya MR, Cannon GC, Heinhorst S, Tanaka S, Williams EB, Yeates TO, Kerfeld CA (2006). "The structure of beta-carbonic anhydrase from the carboxysomal shell reveals a distinct subclass with one active site for the price of two". J. Biol. Chem. 281 (11): 7546–55. doi:10.1074/jbc.M510464200. PMID 16407248.
- ↑ So AK, Espie GS, Williams EB, Shively JM, Heinhorst S, Cannon GC (2004). "A novel evolutionary lineage of carbonic anhydrase (epsilon class) is a component of the carboxysome shell". J. Bacteriol. 186 (3): 623–30. doi:10.1128/JB.186.3.623-630.2004. PMID 14729686.
de:Carboanhydratase it:Carbonato deidratasi fi:Karboanhydraasi sv:Karboanhydras
