Alipogene tiparvovec
| Clinical data | |
|---|---|
| Trade names | Glybera |
| AHFS/Drugs.com | UK Drug Information |
| Routes of administration | Intramuscular injection |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| UNII | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Associate Editor(s)-in-Chief: Sheng Shi, M.D. [2]
Overview
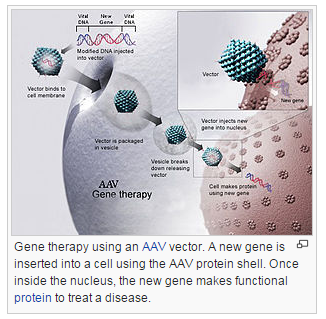
Alipogene tiparvovec (marketed under the trade name Glybera) is a gene therapy treatment that compensates for lipoprotein lipase deficiency (LPLD), which can cause severe pancreatitis.[1] In July 2012, the European Medicines Agency recommended it for approval, the first recommendation for a gene therapy treatment in either Europe or the United States. The recommendation was endorsed by the European Commission in November 2012[2][3] and commercial rollout is expected in late 2013.[4]
Mechanism
The adeno-associated virus serotype 1 (AAV1) viral vector delivers an intact copy of the human lipoprotein lipase (LPL) gene. Data from the clinical trials indicates that fat concentrations in blood were reduced between 3 and 12 weeks after injection, in nearly all patients. The advantages of AAV include apparent lack of pathogenicity, delivery to non-dividing cells, and non-integrating in contrast to retroviruses, which show random insertion with accompanying risk of cancer. AAV also presents very low immunogenicity, mainly restricted to generating neutralizing antibodies, and little well defined cytotoxic response.[5][6][7] The cloning capacity of the vector is limited to replacement of the virus's 4.8 kilobase genome.
Cost
Alipogene tiparvovec is expected to cost around $1.6 million for treatment[8] which will make it the most expensive medicine in the world.[9]
See Also
References
- ↑ European Agency Backs Approval of a Gene Therapy July 20, 2012
- ↑ Gallagher, James. (2012-11-02) BBC News – Gene therapy: Glybera approved by European Commission. Bbc.co.uk. Retrieved on 2012-12-15.
- ↑ Richards, Sabrina. "Gene Therapy Arrives in Europe". The Scientist. Retrieved 16 November 2012.
- ↑ Press Release. UniQure (2012-11-02). Retrieved on 2012-12-15.
- ↑ Chirmule N, Propert K, Magosin S, Qian Y, Qian R, Wilson J (September 1999). "Immune responses to adenovirus and adeno-associated virus in humans". Gene Therapy. 6 (9): 1574–83. doi:10.1038/sj.gt.3300994. PMID 10490767.
- ↑ Hernandez YJ, Wang J, Kearns WG, Loiler S, Poirier A, Flotte TR (1 October 1999). "Latent Adeno-Associated Virus Infection Elicits Humoral but Not Cell-Mediated Immune Responses in a Nonhuman Primate Model". Journal of Virology. 73 (10): 8549–58. PMC 112875. PMID 10482608.
- ↑ Ponnazhagan S, Mukherjee P, Yoder MC; et al. (April 1997). "Adeno-associated virus 2-mediated gene transfer in vivo: organ-tropism and expression of transduced sequences in mice". Gene. 190 (1): 203–10. doi:10.1016/S0378-1119(96)00576-8. PMID 9185868.
- ↑ Gene-Therapy Approval Marks Major Milestone
- ↑ Gene therapy approved in Europe for first time
- Pages with script errors
- CS1 maint: Multiple names: authors list
- CS1 maint: Explicit use of et al.
- Template:drugs.com link with non-standard subpage
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Infobox drug articles without a structure image
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles containing unverified chemical infoboxes
- Drug
- Applied genetics
- Gene delivery
- Cardiovascular Drugs
- Drugs