Tioconazole: Difference between revisions
Adeel Jamil (talk | contribs) No edit summary |
m (Protected "Tioconazole": Bot: Protecting all pages from category Drug ([Edit=Allow only administrators] (indefinite) [Move=Allow only administrators] (indefinite))) |
||
| (2 intermediate revisions by one other user not shown) | |||
| Line 1: | Line 1: | ||
{{DrugProjectFormSinglePage | {{DrugProjectFormSinglePage | ||
|authorTag= | |authorTag={{AJ}} | ||
|OTC=Yes | |||
|genericName=tioconazole | |||
|aOrAn=a | |aOrAn=a | ||
| | |drugClass=antifungal , anti-Infective agent and imidazole | ||
|adverseReactions= | |indicationType=treatment | ||
|indication=[[Vaginal antifungal]] | |||
|adverseReactions=mild increase in vaginal burning, [[itching]] or irritation | |||
|blackBoxWarningTitle=<span style="color:#FF0000;">ConditionName: </span> | |blackBoxWarningTitle=<span style="color:#FF0000;">ConditionName: </span> | ||
|blackBoxWarningBody=<i><span style="color:#FF0000;">ConditionName: </span></i> | |blackBoxWarningBody=<i><span style="color:#FF0000;">ConditionName: </span></i> | ||
| Line 12: | Line 16: | ||
<!--FDA-Labeled Indications and Dosage (Adult)--> | <!--FDA-Labeled Indications and Dosage (Adult)--> | ||
|fdaLIADAdult= | |fdaLIADAdult=* Treats vaginal yeast infections | ||
====DIRECTIONS==== | |||
:* | * before using this product read the enclosed brochure and instructions on foil packet for complete directions and information | ||
* adults and children 12 years and over: | |||
* open the foil packet just before use and remove purple cap | |||
* insert entire contents of applicator into the vagina at bedtime. Throw applicator away after use. | |||
|offLabelAdultGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of {{PAGENAME}} in adult patients. | |||
|offLabelAdultGuideSupport= | |||
There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of {{PAGENAME}} in adult patients. | |||
<!--Non–Guideline-Supported Use (Adult)--> | <!--Non–Guideline-Supported Use (Adult)--> | ||
|offLabelAdultNoGuideSupport= | |offLabelAdultNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of {{PAGENAME}} in adult patients. | ||
There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of {{PAGENAME}} in adult patients. | |||
<!--Pediatric Indications and Dosage--> | <!--Pediatric Indications and Dosage--> | ||
<!--FDA-Labeled Indications and Dosage (Pediatric)--> | <!--FDA-Labeled Indications and Dosage (Pediatric)--> | ||
|fdaLIADPed= | |fdaLIADPed=* Children under 12 years of age: ask a doctor | ||
* | |||
: | |||
<!--Guideline-Supported Use (Pediatric)--> | <!--Guideline-Supported Use (Pediatric)--> | ||
|offLabelPedGuideSupport= | |offLabelPedGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of {{PAGENAME}} in pediatric patients. | ||
There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of {{PAGENAME}} in pediatric patients. | |||
<!--Non–Guideline-Supported Use (Pediatric)--> | <!--Non–Guideline-Supported Use (Pediatric)--> | ||
|offLabelPedNoGuideSupport= | |offLabelPedNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of {{PAGENAME}} in pediatric patients. | ||
* | <!--Contraindications--> | ||
|contraindications=There is limited information regarding <i>Contraindications</i> of {{PAGENAME}} in pediatric patients. | |||
|warnings=* For vaginal use only | |||
=====Do not use===== | |||
* if you have never had a vaginal yeast infection diagnosed by a doctor | |||
=====When using this product===== | |||
* do not use tampons, douches, spermicides, or other vaginal products. Condoms and diaphragms may be damaged and fail to prevent pregnancy or sexually transmitted disease (STDs). | |||
* do not have vaginal intercourse | |||
* mild increase in vaginal burning, itching or irritation may occur | |||
=====Stop use and ask a doctor if===== | |||
* symptoms do not get better after 3 days | |||
* symptoms last more than 7 days | |||
* you get a rash or hives, abdominal pain, fever, chills, nausea, vomiting, or foul-smelling vaginal discharge | |||
=====Keep out of reach of children===== | |||
* If swallowed, get medical help or contact a Poison Control Center right away. | |||
|clinicalTrials=There is limited information regarding <i>Clinical Trial Experience</i> of {{PAGENAME}} in the drug label. | |clinicalTrials=There is limited information regarding <i>Clinical Trial Experience</i> of {{PAGENAME}} in the drug label. | ||
|postmarketing=There is limited information regarding <i>Postmarketing Experience</i> of {{PAGENAME}} in the drug label. | |postmarketing=There is limited information regarding <i>Postmarketing Experience</i> of {{PAGENAME}} in the drug label. | ||
|drugInteractions=There is limited information regarding <i>Drug Interactions</i> of {{PAGENAME}} in the drug label. | |||
|useInPregnancyFDA=* ask a health professional before use. | |||
|useInPregnancyAUS=* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | |useInPregnancyAUS=* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | ||
| Line 255: | Line 74: | ||
|useInNursing=There is no FDA guidance on the use of {{PAGENAME}} with respect to nursing mothers. | |useInNursing=There is no FDA guidance on the use of {{PAGENAME}} with respect to nursing mothers. | ||
|useInPed=There is no FDA guidance on the use of {{PAGENAME}} with respect to pediatric patients. | |useInPed=There is no FDA guidance on the use of {{PAGENAME}} with respect to pediatric patients. | ||
|useInGeri= | |useInGeri=* If swallowed, get medical help or contact a Poison Control Center right away. | ||
|useInGender=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific gender populations. | |useInGender=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific gender populations. | ||
|useInRace=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific racial populations. | |useInRace=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific racial populations. | ||
| Line 264: | Line 84: | ||
<!--Administration and Monitoring--> | <!--Administration and Monitoring--> | ||
|administration=* | |administration=* Topical | ||
|monitoring=There is limited information regarding <i>Monitoring</i> of {{PAGENAME}} in the drug label. | |monitoring=There is limited information regarding <i>Monitoring</i> of {{PAGENAME}} in the drug label. | ||
| Line 275: | Line 93: | ||
<!--Overdosage--> | <!--Overdosage--> | ||
|overdose= | |overdose=There is limited information regarding <i>Chronic Overdose</i> of {{PAGENAME}} in the drug label. | ||
<!--Pharmacology--> | |||
<!--Drug box 2--> | |||
|drugBox={{one source|date=May 2014}} | |||
==== | {{Drugbox2 | ||
| Verifiedfields = changed | |||
| verifiedrevid = 415485693 | |||
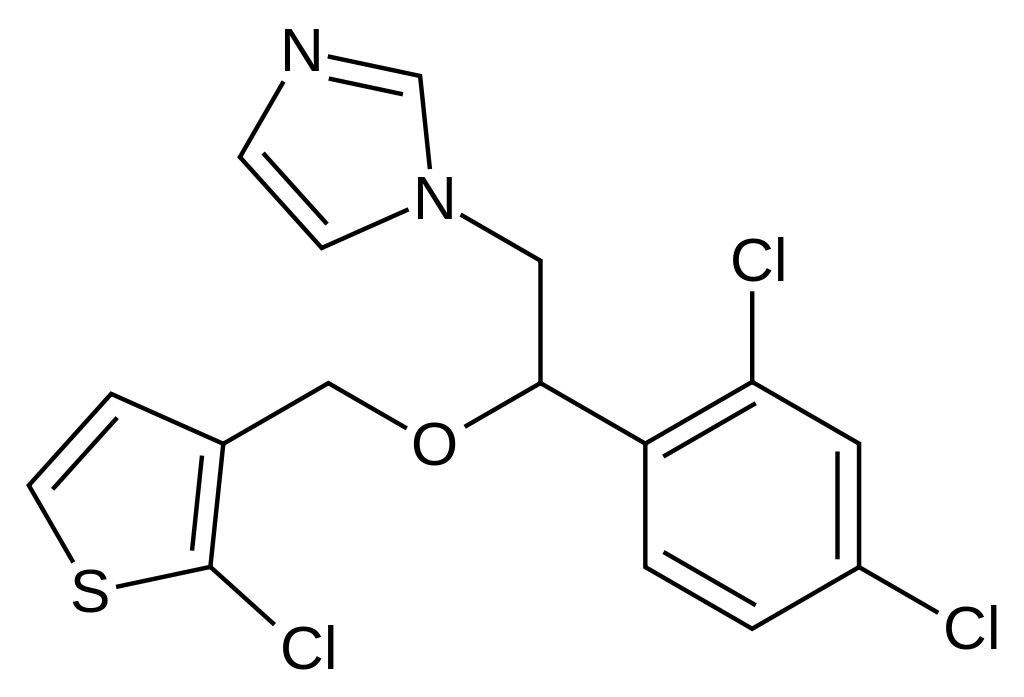
| IUPAC_name = (''RS'')-1-[2-[(2-Chloro-3-thienyl)methoxy]-2-(2,4-dichlorophenyl)ethyl]-1''H''-imidazole | |||
| image = Tioconazole.png | |||
| imagename = 1 : 1 mixture (racemate) | |||
| drug_name = Tioconazole | |||
=== | <!--Clinical data--> | ||
| tradename = Vagistat-1 | |||
| Drugs.com = {{drugs.com|monograph|tioconazole}} | |||
| pregnancy_category = | |||
| legal_US= OTC | |||
| routes_of_administration = Topical | |||
<!--Pharmacokinetic data--> | |||
| bioavailability = | |||
| protein_bound = | |||
| metabolism = | |||
| elimination_half-life = | |||
<!-- | <!--Identifiers--> | ||
| CASNo_Ref = {{cascite|correct|CAS}} | |||
| CAS_number_Ref = {{cascite|correct|??}} | |||
| CAS_number = 65899-73-2 | |||
| ATC_prefix = D01 | |||
| ATC_suffix = AC07 | |||
| ATC_supplemental = {{ATC|G01|AF08}} | |||
| PubChem = 5482 | |||
| DrugBank_Ref = {{drugbankcite|changed|drugbank}} | |||
| DrugBank = DB01007 | |||
| ChemSpiderID_Ref = {{chemspidercite|changed|chemspider}} | |||
| ChemSpiderID = 5282 | |||
| UNII_Ref = {{fdacite|changed|FDA}} | |||
| UNII = S57Y5X1117 | |||
| KEGG_Ref = {{keggcite|correct|kegg}} | |||
| KEGG = D00890 | |||
| ChEBI_Ref = {{ebicite|changed|EBI}} | |||
| ChEBI = 9604 | |||
| ChEMBL_Ref = {{ebicite|changed|EBI}} | |||
| ChEMBL = 1200438 | |||
<!-- | <!--Chemical data--> | ||
| | | C=16 | H=13 | Cl=3 | N=2 | O=1 | S=1 | ||
| molecular_weight = 387.711 g/mol | |||
| synonyms = Thioconazole | |||
}} | |||
|mechAction=* | |mechAction=* | ||
<!--Structure--> | <!--Structure--> | ||
|structure=* | |structure=* butylated hydroxyanisole, magnesium aluminum silicate, white petrolatum | ||
<!--Pharmacodynamics--> | <!--Pharmacodynamics--> | ||
| Line 313: | Line 165: | ||
<!--How Supplied--> | <!--How Supplied--> | ||
| | |storage=* Store at 20° - 25°C (68° - 77°F) | ||
|packLabel= | |packLabel======PRINCIPAL DISPLAY PANEL===== | ||
|fdaPatientInfo= | |||
Compare to 1-Day active ingredient | |||
tioconazole 1 | |||
tioconozole ointment 6.5% - vaginal antifungal | |||
1 dose vaginal treatment | |||
ready-to-use prefilled applicator | |||
cures most vaginal yeast infections | |||
1 dose treatment | |||
NET WT 4.6g (0.16 OZ) filled applicator | |||
[[File:TIOCONAZOLE drug lable01.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
[[File:TIOCONAZOLE drug lable02.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
|fdaPatientInfo=[[File:TIOCONAZOLE consumer info 01.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
[[File:TIOCONAZOLE consumer info 02.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
[[File:TIOCONAZOLE consumer info 03.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
[[File:TIOCONAZOLE consumer info 04.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
[[File:TIOCONAZOLE consumer info 05.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
[[File:TIOCONAZOLE consumer info 06.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
|alcohol=* Alcohol-{{PAGENAME}} interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | |alcohol=* Alcohol-{{PAGENAME}} interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | ||
<!--Brand Names--> | <!--Brand Names--> | ||
|brandNames=* ® | |brandNames=* ® | ||
|drugShortage= | |drugShortage= | ||
}} | }} | ||
Latest revision as of 17:17, 20 August 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Adeel Jamil, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
NOTE: Most over the counter (OTC) are not reviewed and approved by the FDA. However, they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Overview
Tioconazole is a antifungal , anti-Infective agent and imidazole that is FDA approved for the treatment of Vaginal antifungal. Common adverse reactions include mild increase in vaginal burning, itching or irritation.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Treats vaginal yeast infections
DIRECTIONS
- before using this product read the enclosed brochure and instructions on foil packet for complete directions and information
- adults and children 12 years and over:
- open the foil packet just before use and remove purple cap
- insert entire contents of applicator into the vagina at bedtime. Throw applicator away after use.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Tioconazole in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Tioconazole in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Children under 12 years of age: ask a doctor
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Tioconazole in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Tioconazole in pediatric patients.
Contraindications
There is limited information regarding Contraindications of Tioconazole in pediatric patients.
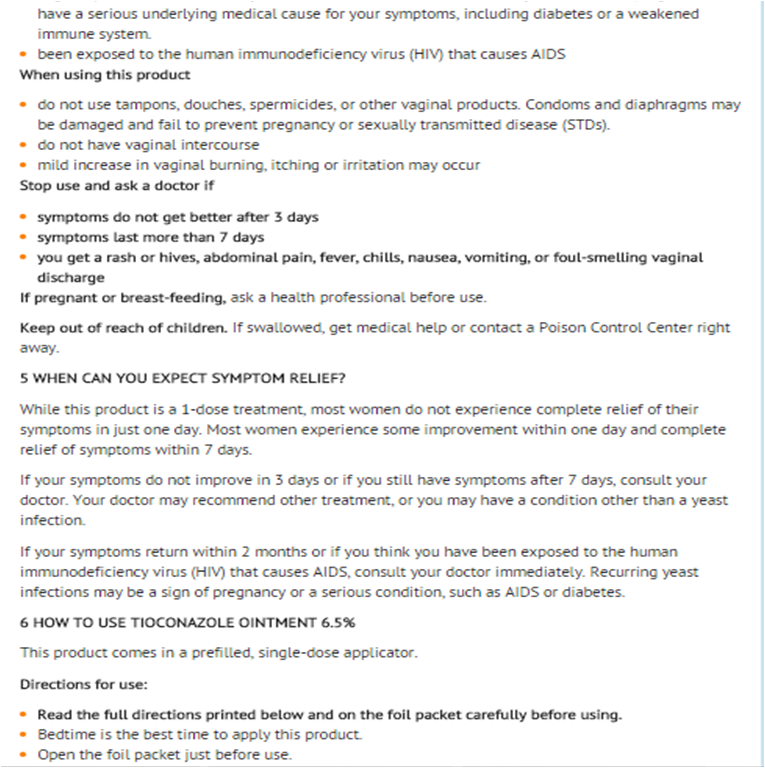
Warnings
- For vaginal use only
Do not use
- if you have never had a vaginal yeast infection diagnosed by a doctor
When using this product
- do not use tampons, douches, spermicides, or other vaginal products. Condoms and diaphragms may be damaged and fail to prevent pregnancy or sexually transmitted disease (STDs).
- do not have vaginal intercourse
- mild increase in vaginal burning, itching or irritation may occur
Stop use and ask a doctor if
- symptoms do not get better after 3 days
- symptoms last more than 7 days
- you get a rash or hives, abdominal pain, fever, chills, nausea, vomiting, or foul-smelling vaginal discharge
Keep out of reach of children
- If swallowed, get medical help or contact a Poison Control Center right away.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Tioconazole in the drug label.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Tioconazole in the drug label.
Drug Interactions
There is limited information regarding Drug Interactions of Tioconazole in the drug label.
Use in Specific Populations
Pregnancy
- ask a health professional before use.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Tioconazole in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Tioconazole during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Tioconazole with respect to nursing mothers.
Pediatric Use
There is no FDA guidance on the use of Tioconazole with respect to pediatric patients.
Geriatic Use
- If swallowed, get medical help or contact a Poison Control Center right away.
Gender
There is no FDA guidance on the use of Tioconazole with respect to specific gender populations.
Race
There is no FDA guidance on the use of Tioconazole with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Tioconazole in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Tioconazole in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Tioconazole in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Tioconazole in patients who are immunocompromised.
Administration and Monitoring
Administration
- Topical
Monitoring
There is limited information regarding Monitoring of Tioconazole in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Tioconazole in the drug label.
Overdosage
There is limited information regarding Chronic Overdose of Tioconazole in the drug label.
Pharmacology
This article relies largely or entirely on a single source. (May 2014) |
| |
1 : 1 mixture (racemate)Tioconazole
| |
| Systematic (IUPAC) name | |
| (RS)-1-[2-[(2-Chloro-3-thienyl)methoxy]-2-(2,4-dichlorophenyl)ethyl]-1H-imidazole | |
| Identifiers | |
| CAS number | |
| ATC code | D01 G01AF08 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 387.711 g/mol |
| Synonyms | Thioconazole |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | Topical |
Mechanism of Action
Structure
- butylated hydroxyanisole, magnesium aluminum silicate, white petrolatum
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Tioconazole in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Tioconazole in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Tioconazole in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Tioconazole in the drug label.
How Supplied
There is limited information regarding Tioconazole How Supplied in the drug label.
Storage
- Store at 20° - 25°C (68° - 77°F)
Images
Drug Images
{{#ask: Page Name::Tioconazole |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
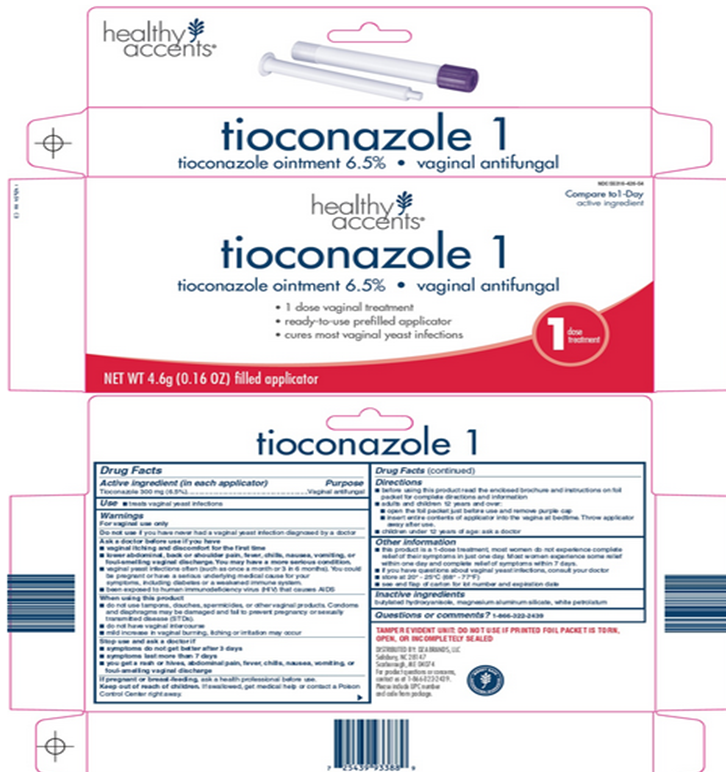
PRINCIPAL DISPLAY PANEL
Compare to 1-Day active ingredient
tioconazole 1
tioconozole ointment 6.5% - vaginal antifungal
1 dose vaginal treatment
ready-to-use prefilled applicator
cures most vaginal yeast infections
1 dose treatment
NET WT 4.6g (0.16 OZ) filled applicator

{{#ask: Label Page::Tioconazole |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information





Precautions with Alcohol
- Alcohol-Tioconazole interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- ®
Look-Alike Drug Names
There is limited information regarding Tioconazole Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Tioconazole
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Tioconazole |Label Name=Tioconazole11.png
}}
{{#subobject:
|Label Page=Tioconazole |Label Name=Tioconazole11.png
}}