Primary carnitine deficiency
| Primary carnitine deficiency | |
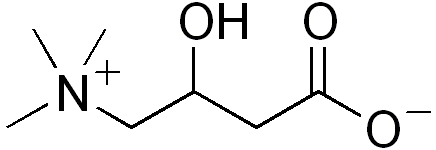 | |
|---|---|
| Carnitine | |
| ICD-10 | E71.3 |
| ICD-9 | 277.81 |
| OMIM | 212140 |
| DiseasesDB | 31103 |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Primary carnitine deficiency is a condition that prevents the body from using fats for energy, particularly during periods without food. Carnitine, a natural substance acquired mostly through diet, is used by cells to process fats and produce energy. People with primary carnitine deficiency have defective proteins called carnitine transporters, which bring carnitine into cells and prevent its escape from the body.
Historical Perspective
The current understanding of primary carnitine deficiency has been greatly influenced by the research of Doctors Susan C. Winter and Neil Buist. Dr. Winter was one of the first doctors in the United States to begin treating inborn errors of metabolism with intravenous carnitine.
Pathophysiology
There is a decrease in the transport function as a result carnitine is lost from the body. Cells are not supplied with an adequate amount of carnitine. Without carnitine, fats cannot be processed correctly and are not converted into energy, which can lead to characteristic signs and symptoms of this disorder.
Genetics
Mutations in the SLC22A5 gene lead to the production of defective carnitine transporters. This condition is inherited in an autosomal recessive pattern.
Differentiating Primary Carnitine Deficiency from other Diseases
This condition is sometimes mistaken for Reye syndrome, a severe disorder that develops in children while they appear to be recovering from viral infections such as chickenpox or flu. Most cases of Reye syndrome are associated with the use of aspirin during these viral infections.
Epidemiology and Demographics
On the average, primary carnitine deficiency affects 1 in every 40,000 live births in Japan and 1 in every 37,000 to 100,000 newborns in Australia. The incidence of this condition in other populations is unknown, but is probably similar to that reported for Japan.
Risk Factors
- Infections
- Fasting
- Malnutrition
- Strenuous physical work
Natural History, Complications and Prognosis
Serious complications such as heart failure, liver problems, coma, and sudden unexpected death can occur. Acute illness due to primary carnitine deficiency can be triggered by periods of fasting or illnesses such as viral infections, particularly when eating is reduced.
Diagnosis
Symptoms
Physical Examination
Typically, initial signs of this disorder occur during infancy or early childhood and often include brain function abnormalities
Heart
Neuroloogic
Laboratory Findings
Electrolyte and Biomarker Studies
Treatment
Primary carnitine deficiency is sometimes diagnosed in adults and is then thought to be less severe both in symptoms and life expectation. Treatment is usually done by supplementation of L-carnitine after assessing the severity of the deficiency after a muscular biopsy.
Contraindicated medications
Carnitine deficiency is considered an absolute contraindication to the use of the following medications:
References
This article incorporates public domain text from The U.S. National Library of Medicine
Template:Endocrine, nutritional and metabolic pathology