Paliperidone (oral): Difference between revisions
(Blanked the page) |
No edit summary |
||
| Line 1: | Line 1: | ||
{{DrugProjectFormSinglePage | |||
|authorTag={{SS}} | |||
|genericName=Paliperidone | |||
|aOrAn=a | |||
|drugClass=Typical Antipsychotic | |||
|indicationType=treatment | |||
|indication=[[Schizophrenia]], [[Schizoaffective Disorder]] | |||
|hasBlackBoxWarning=Yes | |||
|adverseReactions=[[Tachycardia]], [[Hyperprolactinemia]],Weight gain, [[Constipation]], [[Indigestion]], [[Akathisia]],[[Dyskinesia]], [[Dystonia]],Extrapyramidal disease,[[Parkinsonism]],[[Somnolence]],[[Tremor]],[[Anxiety]],[[Nasopharyngitis]] | |||
|blackBoxWarningTitle=<span style="color:#FF0000;">WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS</span> | |||
|blackBoxWarningBody=<i><span style="color:#FF0000;">Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA® is not approved for use in patients with dementia-related psychosis. (5.1)</span></i> | |||
|fdaLIADAdult=<h4>[[Schizophrenia]]</h4> | |||
* Dosing information | |||
:* Recommended dosage: '''6 mg PO qd'''. | |||
::* Initial dose titration is not required. Although it has not been systematically established that doses above 6 mg have additional benefit, there was a general trend for greater effects with higher doses. This must be weighed against the dose-related increase in adverse reactions. Thus, some patients may benefit from higher doses, up to '''12 mg/day''', and for some patients, a lower dose of '''3 mg/day''' may be sufficient. Dose increases above 6 mg/day should be made only after clinical reassessment and generally should occur at intervals of more than 5 days. When dose increases are indicated, increments of 3 mg/day are recommended. The maximum recommended dose is 12 mg/day. | |||
:* In a longer-term study, INVEGA® has been shown to be effective in delaying time to relapse in patients with schizophrenia who were stabilized on INVEGA® for 6 weeks [see Clinical Studies (14)]. INVEGA® should be prescribed at the lowest effective dose for maintaining clinical stability and the physician should periodically reevaluate the long-term usefulness of the drug in individual patients. | |||
<h4>[[Schizoaffective Disorder]]</h4> | |||
* Dosing information | |||
:* Recommended dosage: '''6 mg PO qd'''. | |||
::* Initial dose titration is not required. | |||
::* Recommended dose range: '''3 to 12 mg PO qd''' | |||
:::* A general trend for greater effects was seen with higher doses. This trend must be weighed against dose-related increase in adverse reactions. Dosage adjustment, if indicated, should occur only after clinical reassessment. Dose increases, if indicated, generally should occur at intervals of more than 4 days. When dose increases are indicated, increments of 3 mg/day are recommended. | |||
::* The maximum recommended dose is 12 mg/day. | |||
|offLabelAdultGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Paliperidone in adult patients. | |||
|offLabelAdultNoGuideSupport=<h4>[[Bipolar I disorder]]</h4> | |||
* Dosing information | |||
:* Not applicable 20565430 | |||
|fdaLIADPed=<h4>[[Schizophrenia]] </h4> | |||
* Dosing information for Adolescents (12–17 years of age) | |||
:* Recommended starting dosage: '''3 mg PO qd'''. Initial dose titration is not required. | |||
::* Dose increases, if considered necessary, should be made only after clinical reassessment and should occur at increments of '''3 mg/day''' at intervals of more than '''5 days'''. Prescribers should be mindful that, in the adolescent schizophrenia study, there was no clear enhancement to efficacy at the higher doses, i.e., 6 mg for subjects weighing less than 51 kg and 12 mg for subjects weighing 51 kg or greater, while adverse events were dose-related. | |||
|offLabelPedGuideSupport=There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Paliperidone in pediatric patients. | |||
|offLabelPedNoGuideSupport=There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of Paliperidone in pediatric patients. | |||
|contraindications=[[Hypersensitivity reactions]], including [[anaphylactic reactions]] and [[angioedema]], have been observed in patients treated with [[risperidone]] and paliperidone. INVEGA® (paliperidone) is a metabolite of [[risperidone]] and is therefore contraindicated in patients with a known [[hypersensitivity]] to either paliperidone or [[risperidone]], or to any of the excipients in INVEGA®. | |||
|warnings=====Increased Mortality in Elderly Patients with Dementia-Related Psychosis==== | |||
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA® (paliperidone) is not approved for the treatment of dementia-related psychosis. | |||
====Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients With Dementia-Related Psychosis==== | |||
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly subjects with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated subjects. INVEGA® was not marketed at the time these studies were performed. INVEGA® is not approved for the treatment of patients with dementia-related psychosis [see also Boxed Warning and Warnings and Precautions (5.1)]. | |||
====Neuroleptic Malignant Syndrome==== | |||
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs, including paliperidone. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure. | |||
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases in which the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system pathology. | |||
The management of NMS should include: (1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy; (2) intensive symptomatic treatment and medical monitoring; and (3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS. | |||
If a patient appears to require antipsychotic drug treatment after recovery from NMS, reintroduction of drug therapy should be closely monitored, since recurrences of NMS have been reported. | |||
====QT Prolongation==== | |||
Paliperidone causes a modest increase in the corrected QT (QTc) interval. The use of paliperidone should be avoided in combination with other drugs that are known to prolong QTc including Class 1A (e.g., quinidine, procainamide) or Class III (e.g., amiodarone, sotalol) antiarrhythmic medications, antipsychotic medications (e.g., chlorpromazine, thioridazine), antibiotics (e.g., gatifloxacin, moxifloxacin), or any other class of medications known to prolong the QTc interval. Paliperidone should also be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias. | |||
Certain circumstances may increase the risk of the occurrence of torsade de pointes and/or sudden death in association with the use of drugs that prolong the QTc interval, including (1) bradycardia; (2) hypokalemia or hypomagnesemia; (3) concomitant use of other drugs that prolong the QTc interval; and (4) presence of congenital prolongation of the QT interval. | |||
The effects of paliperidone on the QT interval were evaluated in a double-blind, active-controlled (moxifloxacin 400 mg single dose), multicenter QT study in adults with schizophrenia and schizoaffective disorder, and in three placebo- and active-controlled 6-week, fixed-dose efficacy trials in adults with schizophrenia. | |||
In the QT study (n = 141), the 8 mg dose of immediate-release oral paliperidone (n=50) showed a mean placebo-subtracted increase from baseline in QTcLD of 12.3 msec (90% CI: 8.9; 15.6) on day 8 at 1.5 hours post-dose. The mean steady-state peak plasma concentration for this 8 mg dose of paliperidone immediate-release was more than twice the exposure observed with the maximum recommended 12 mg dose of INVEGA® (Cmax ss = 113 ng/mL and 45 ng/mL, respectively, when administered with a standard breakfast). In this same study, a 4 mg dose of the immediate-release oral formulation of paliperidone, for which Cmax ss = 35 ng/mL, showed an increased placebo-subtracted QTcLD of 6.8 msec (90% CI: 3.6; 10.1) on day 2 at 1.5 hours post-dose. None of the subjects had a change exceeding 60 msec or a QTcLD exceeding 500 msec at any time during this study. | |||
For the three fixed-dose efficacy studies in subjects with schizophrenia, electrocardiogram (ECG) measurements taken at various time points showed only one subject in the INVEGA® 12 mg group had a change exceeding 60 msec at one time-point on Day 6 (increase of 62 msec). No subject receiving INVEGA® had a QTcLD exceeding 500 msec at any time in any of these three studies. | |||
====Tardive Dyskinesia==== | |||
A syndrome of potentially irreversible, involuntary, dyskinetic movements may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown. | |||
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible appear to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase, but the syndrome can develop after relatively brief treatment periods at low doses, although this is uncommon. | |||
There is no known treatment for established tardive dyskinesia, although the syndrome may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment itself may suppress (or partially suppress) the signs and symptoms of the syndrome and may thus mask the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown. | |||
Given these considerations, INVEGA® should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that is known to respond to antipsychotic drugs. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically. | |||
If signs and symptoms of tardive dyskinesia appear in a patient treated with INVEGA®, drug discontinuation should be considered. However, some patients may require treatment with INVEGA® despite the presence of the syndrome. | |||
====Metabolic Changes==== | |||
Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and body weight gain. While all of the drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile. | |||
Hyperglycemia and Diabetes Mellitus | |||
Hyperglycemia and diabetes mellitus, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, have been reported in patients treated with all atypical antipsychotics. These cases were, for the most part, seen in post-marketing clinical use and epidemiologic studies, not in clinical trials, and there have been few reports of hyperglycemia or diabetes in trial subjects treated with INVEGA®. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population. Given these confounders, the relationship between atypical antipsychotic use and hyperglycemia-related adverse events is not completely understood. However, epidemiological studies suggest an increased risk of treatment-emergent hyperglycemia-related adverse events in patients treated with the atypical antipsychotics. Because INVEGA® was not marketed at the time these studies were performed, it is not known if INVEGA® is associated with this increased risk. | |||
Patients with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics should be monitored regularly for worsening of glucose control. Patients with risk factors for diabetes mellitus (e.g., obesity, family history of diabetes) who are starting treatment with atypical antipsychotics should undergo fasting blood glucose testing at the beginning of treatment and periodically during treatment. Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Patients who develop symptoms of hyperglycemia during treatment with atypical antipsychotics should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients required continuation of anti-diabetic treatment despite discontinuation of the suspect drug. | |||
Pooled data from the three placebo-controlled, 6-week, fixed-dose studies in adult subjects with schizophrenia are presented in Table 1a. | |||
[[File:Paliperidone_warning_01.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
In the uncontrolled, longer-term open-label extension studies, INVEGA® was associated with a mean change in glucose of +3.3 mg/dL at Week 24 (n=570) and +4.6 mg/dL at Week 52 (n=314). | |||
Data from the placebo-controlled 6-week study in adolescent subjects (12–17 years of age) with schizophrenia are presented in Table 1b. | |||
[[File:Paliperidone_warning_02.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
<i>Dyslipidemia</i> | |||
Undesirable alterations in lipids have been observed in patients treated with atypical antipsychotics. | |||
Pooled data from the three placebo-controlled, 6-week, fixed-dose studies in adult subjects with schizophrenia are presented in Table 2a. | |||
[[File:Paliperidone_warning_03.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
In the uncontrolled, longer-term open-label extension studies, INVEGA® was associated with a mean change in (a) total cholesterol of -1.5 mg/dL at Week 24 (n=573) and -1.5 mg/dL at Week 52 (n=317), (b) triglycerides of -6.4 mg/dL at Week 24 (n=573) and -10.5 mg/dL at Week 52 (n=317); (c) LDL of -1.9 mg/dL at Week 24 (n=557) and -2.7 mg/dL at Week 52 (n=297); and (d) HDL of +2.2 mg/dL at Week 24 (n=568) and +3.6 mg/dL at Week 52 (n=302). | |||
Data from the placebo-controlled 6-week study in adolescent subjects (12–17 years of age) with schizophrenia are presented in Table 2b. | |||
[[File:Paliperidone_warning_04.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
<i>Weight Gain</i> | |||
Weight gain has been observed with atypical antipsychotic use. Clinical monitoring of weight is recommended. | |||
Schizophrenia Trials | |||
Data on mean changes in body weight and the proportion of subjects meeting a weight gain criterion of ≥ 7% of body weight from the three placebo-controlled, 6-week, fixed-dose studies in adult subjects are presented in Table 3a. | |||
[[File:Paliperidone_warning_05.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
In the uncontrolled, longer-term open-label extension studies, INVEGA® was associated with a mean change in weight of +1.4 kg at Week 24 (n=63) and +2.6 kg at Week 52 (n=302). | |||
Weight gain in adolescent subjects with schizophrenia was assessed in a 6-week, double-blind, placebo-controlled study and an open-label extension with a median duration of exposure to INVEGA® of 182 days. Data on mean changes in body weight and the proportion of subjects meeting a weight gain criterion of ≥ 7% of body weight [see Clinical Studies (14.1)] from the placebo-controlled 6-week study in adolescent subjects (12–17 years of age) are presented in Table 3b. | |||
[[File:Paliperidone_warning_06.png|thumb|none|400px|This image is provided by the National Library of Medicine.]] | |||
In the open-label long-term study the proportion of total subjects treated with INVEGA® with an increase in body weight of ≥ 7% from baseline was 33%. When treating adolescent patients with INVEGA®, weight gain should be assessed against that expected with normal growth. When taking into consideration the median duration of exposure to INVEGA® in the open-label study (182 days) along with expected normal growth in this population based on age and gender, an assessment of standardized scores relative to normative data provides a more clinically relevant measure of changes in weight. The mean change from open-label baseline to endpoint in standardized score for weight was 0.1 (4% above the median for normative data). Based on comparison to the normative data, these changes are not considered to be clinically significant. | |||
Schizoaffective Disorder Trials | |||
In the pooled data from the two placebo-controlled, 6-week studies in adult subjects with schizoaffective disorder, a higher percentage of INVEGA®-treated subjects (5%) had an increase in body weight of ≥ 7% compared with placebo-treated subjects (1%). In the study that examined high- and low-dose groups, the increase in body weight of ≥ 7% was 3% in the low-dose group, 7% in the high-dose group, and 1% in the placebo group. | |||
====[[Hyperprolactinemia]]==== | |||
Like other drugs that antagonize dopamine D2 receptors, paliperidone elevates prolactin levels and the elevation persists during chronic administration. Paliperidone has a prolactin-elevating effect similar to that seen with risperidone, a drug that is associated with higher levels of prolactin than other antipsychotic drugs. | |||
Hyperprolactinemia, regardless of etiology, may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotrophin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported in patients receiving prolactin-elevating compounds. Long-standing hyperprolactinemia when associated with hypogonadism may lead to decreased bone density in both female and male subjects. | |||
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin dependent in vitro, a factor of potential importance if the prescription of these drugs is considered in a patient with previously detected breast cancer. An increase in the incidence of pituitary gland, mammary gland, and pancreatic islet cell neoplasia (mammary adenocarcinomas, pituitary and pancreatic adenomas) was observed in the risperidone carcinogenicity studies conducted in mice and rats [see Nonclinical Toxicology (13.1)]. Neither clinical studies nor epidemiologic studies conducted to date have shown an association between chronic administration of this class of drugs and tumorigenesis in humans, but the available evidence is too limited to be conclusive. | |||
====Potential for [[Gastrointestinal Obstruction]]==== | |||
Because the INVEGA® tablet is non-deformable and does not appreciably change in shape in the gastrointestinal tract, INVEGA® should ordinarily not be administered to patients with pre-existing severe gastrointestinal narrowing (pathologic or iatrogenic, for example: esophageal motility disorders, small bowel inflammatory disease, "short gut" syndrome due to adhesions or decreased transit time, past history of peritonitis, cystic fibrosis, chronic intestinal pseudoobstruction, or Meckel's diverticulum). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of drugs in non-deformable controlled-release formulations. Because of the controlled-release design of the tablet, INVEGA® should only be used in patients who are able to swallow the tablet whole. | |||
A decrease in transit time, e.g., as seen with diarrhea, would be expected to decrease bioavailability and an increase in transit time, e.g., as seen with gastrointestinal neuropathy, diabetic gastroparesis, or other causes, would be expected to increase bioavailability. These changes in bioavailability are more likely when the changes in transit time occur in the upper GI tract. | |||
====[[Orthostatic Hypotension]] and [[Syncope]]==== | |||
Paliperidone can induce orthostatic hypotension and syncope in some patients because of its alpha-blocking activity. In pooled results of the three placebo-controlled, 6-week, fixed-dose trials in subjects with schizophrenia, syncope was reported in 0.8% (7/850) of subjects treated with INVEGA® (3 mg, 6 mg, 9 mg, 12 mg) compared to 0.3% (1/355) of subjects treated with placebo. INVEGA® should be used with caution in patients with known cardiovascular disease (e.g., heart failure, history of myocardial infarction or ischemia, conduction abnormalities), cerebrovascular disease, or conditions that predispose the patient to hypotension (e.g., dehydration, hypovolemia, and treatment with antihypertensive medications). Monitoring of orthostatic vital signs should be considered in patients who are vulnerable to hypotension. | |||
====[[Leukopenia]], [[Neutropenia]], and [[Agranulocytosis]]==== | |||
Class Effect: In clinical trial and/or postmarketing experience, events of leukopenia/neutropenia have been reported temporally related to antipsychotic agents, including INVEGA®. Agranulocytosis has also been reported. | |||
Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC) and history of drug-induced leukopenia/neutropenia. Patients with a history of a clinically significant low WBC or a drug-induced leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and discontinuation of INVEGA® should be considered at the first sign of a clinically significant decline in WBC in the absence of other causative factors. | |||
Patients with clinically significant neutropenia should be carefully monitored for fever or other symptoms or signs of infection and treated promptly if such symptoms or signs occur. Patients with severe neutropenia (absolute neutrophil count <1000/mm3) should discontinue INVEGA® and have their WBC followed until recovery. | |||
====Potential for Cognitive and Motor Impairment==== | |||
Somnolence was reported in subjects treated with INVEGA® [see Adverse Reactions (6.1, 6.2)]. Antipsychotics, including INVEGA®, have the potential to impair judgment, thinking, or motor skills. Patients should be cautioned about performing activities requiring mental alertness, such as operating hazardous machinery or operating a motor vehicle, until they are reasonably certain that paliperidone therapy does not adversely affect them. | |||
====Seizures==== | |||
During premarketing clinical trials in subjects with schizophrenia (the three placebo-controlled, 6-week, fixed-dose studies and a study conducted in elderly schizophrenic subjects), seizures occurred in 0.22% of subjects treated with INVEGA® (3 mg, 6 mg, 9 mg, 12 mg) and 0.25% of subjects treated with placebo. Like other antipsychotic drugs, INVEGA® should be used cautiously in patients with a history of seizures or other conditions that potentially lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in patients 65 years or older. | |||
====Dysphagia==== | |||
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Aspiration pneumonia is a common cause of morbidity and mortality in patients with advanced Alzheimer's dementia. INVEGA® and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia. | |||
====Suicide==== | |||
The possibility of suicide attempt is inherent in psychotic illnesses, and close supervision of high-risk patients should accompany drug therapy. Prescriptions for INVEGA® should be written for the smallest quantity of tablets consistent with good patient management in order to reduce the risk of overdose. | |||
====Priapism==== | |||
Drugs with alpha-adrenergic blocking effects have been reported to induce priapism. Priapism has been reported with INVEGA® during postmarketing surveillance. Severe priapism may require surgical intervention. | |||
====Thrombotic Thrombocytopenic Purpura (TTP)==== | |||
No cases of TTP were observed during clinical studies with paliperidone. Although cases of TTP have been reported in association with risperidone administration, the relationship to risperidone therapy is unknown. | |||
====Body Temperature Regulation==== | |||
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing INVEGA® to patients who will be experiencing conditions which may contribute to an elevation in core body temperature, e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration. | |||
====Antiemetic Effect==== | |||
An antiemetic effect was observed in preclinical studies with paliperidone. This effect, if it occurs in humans, may mask the signs and symptoms of overdosage with certain drugs or of conditions such as intestinal obstruction, Reye's syndrome, and brain tumor. | |||
====Use in Patients with Concomitant Illness==== | |||
Clinical experience with INVEGA® in patients with certain concomitant illnesses is limited [see Clinical Pharmacology (12.3)]. | |||
Patients with Parkinson's Disease or Dementia with Lewy Bodies are reported to have an increased sensitivity to antipsychotic medication. Manifestations of this increased sensitivity include confusion, obtundation, postural instability with frequent falls, extrapyramidal symptoms, and clinical features consistent with the neuroleptic malignant syndrome. | |||
INVEGA® has not been evaluated or used to any appreciable extent in patients with a recent history of myocardial infarction or unstable heart disease. Patients with these diagnoses were excluded from premarketing clinical trials. Because of the risk of orthostatic hypotension with INVEGA®, caution should be observed in patients with known cardiovascular disease [see Warnings and Precautions (5.9)]. | |||
====Monitoring: Laboratory Tests==== | |||
No specific laboratory tests are recommended. | |||
|alcohol=Alcohol-Paliperidone interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | |||
}} | |||
Revision as of 15:23, 22 August 2014
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sheng Shi, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS
See full prescribing information for complete Boxed Warning.
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA® is not approved for use in patients with dementia-related psychosis. (5.1)
|
Overview
Paliperidone (oral) is a Typical Antipsychotic that is FDA approved for the treatment of Schizophrenia, Schizoaffective Disorder. There is a Black Box Warning for this drug as shown here. Common adverse reactions include Tachycardia, Hyperprolactinemia,Weight gain, Constipation, Indigestion, Akathisia,Dyskinesia, Dystonia,Extrapyramidal disease,Parkinsonism,Somnolence,Tremor,Anxiety,Nasopharyngitis.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Schizophrenia
- Dosing information
- Recommended dosage: 6 mg PO qd.
- Initial dose titration is not required. Although it has not been systematically established that doses above 6 mg have additional benefit, there was a general trend for greater effects with higher doses. This must be weighed against the dose-related increase in adverse reactions. Thus, some patients may benefit from higher doses, up to 12 mg/day, and for some patients, a lower dose of 3 mg/day may be sufficient. Dose increases above 6 mg/day should be made only after clinical reassessment and generally should occur at intervals of more than 5 days. When dose increases are indicated, increments of 3 mg/day are recommended. The maximum recommended dose is 12 mg/day.
- In a longer-term study, INVEGA® has been shown to be effective in delaying time to relapse in patients with schizophrenia who were stabilized on INVEGA® for 6 weeks [see Clinical Studies (14)]. INVEGA® should be prescribed at the lowest effective dose for maintaining clinical stability and the physician should periodically reevaluate the long-term usefulness of the drug in individual patients.
Schizoaffective Disorder
- Dosing information
- Recommended dosage: 6 mg PO qd.
- Initial dose titration is not required.
- Recommended dose range: 3 to 12 mg PO qd
- A general trend for greater effects was seen with higher doses. This trend must be weighed against dose-related increase in adverse reactions. Dosage adjustment, if indicated, should occur only after clinical reassessment. Dose increases, if indicated, generally should occur at intervals of more than 4 days. When dose increases are indicated, increments of 3 mg/day are recommended.
- The maximum recommended dose is 12 mg/day.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Paliperidone in adult patients.
Non–Guideline-Supported Use
Bipolar I disorder
- Dosing information
- Not applicable 20565430
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Schizophrenia
- Dosing information for Adolescents (12–17 years of age)
- Recommended starting dosage: 3 mg PO qd. Initial dose titration is not required.
- Dose increases, if considered necessary, should be made only after clinical reassessment and should occur at increments of 3 mg/day at intervals of more than 5 days. Prescribers should be mindful that, in the adolescent schizophrenia study, there was no clear enhancement to efficacy at the higher doses, i.e., 6 mg for subjects weighing less than 51 kg and 12 mg for subjects weighing 51 kg or greater, while adverse events were dose-related.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Paliperidone in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Paliperidone in pediatric patients.
Contraindications
Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been observed in patients treated with risperidone and paliperidone. INVEGA® (paliperidone) is a metabolite of risperidone and is therefore contraindicated in patients with a known hypersensitivity to either paliperidone or risperidone, or to any of the excipients in INVEGA®.
Warnings
|
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS
See full prescribing information for complete Boxed Warning.
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA® is not approved for use in patients with dementia-related psychosis. (5.1)
|
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA® (paliperidone) is not approved for the treatment of dementia-related psychosis.
Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients With Dementia-Related Psychosis
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly subjects with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated subjects. INVEGA® was not marketed at the time these studies were performed. INVEGA® is not approved for the treatment of patients with dementia-related psychosis [see also Boxed Warning and Warnings and Precautions (5.1)].
Neuroleptic Malignant Syndrome
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs, including paliperidone. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure. The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases in which the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system pathology. The management of NMS should include: (1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy; (2) intensive symptomatic treatment and medical monitoring; and (3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS. If a patient appears to require antipsychotic drug treatment after recovery from NMS, reintroduction of drug therapy should be closely monitored, since recurrences of NMS have been reported.
QT Prolongation
Paliperidone causes a modest increase in the corrected QT (QTc) interval. The use of paliperidone should be avoided in combination with other drugs that are known to prolong QTc including Class 1A (e.g., quinidine, procainamide) or Class III (e.g., amiodarone, sotalol) antiarrhythmic medications, antipsychotic medications (e.g., chlorpromazine, thioridazine), antibiotics (e.g., gatifloxacin, moxifloxacin), or any other class of medications known to prolong the QTc interval. Paliperidone should also be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias. Certain circumstances may increase the risk of the occurrence of torsade de pointes and/or sudden death in association with the use of drugs that prolong the QTc interval, including (1) bradycardia; (2) hypokalemia or hypomagnesemia; (3) concomitant use of other drugs that prolong the QTc interval; and (4) presence of congenital prolongation of the QT interval. The effects of paliperidone on the QT interval were evaluated in a double-blind, active-controlled (moxifloxacin 400 mg single dose), multicenter QT study in adults with schizophrenia and schizoaffective disorder, and in three placebo- and active-controlled 6-week, fixed-dose efficacy trials in adults with schizophrenia. In the QT study (n = 141), the 8 mg dose of immediate-release oral paliperidone (n=50) showed a mean placebo-subtracted increase from baseline in QTcLD of 12.3 msec (90% CI: 8.9; 15.6) on day 8 at 1.5 hours post-dose. The mean steady-state peak plasma concentration for this 8 mg dose of paliperidone immediate-release was more than twice the exposure observed with the maximum recommended 12 mg dose of INVEGA® (Cmax ss = 113 ng/mL and 45 ng/mL, respectively, when administered with a standard breakfast). In this same study, a 4 mg dose of the immediate-release oral formulation of paliperidone, for which Cmax ss = 35 ng/mL, showed an increased placebo-subtracted QTcLD of 6.8 msec (90% CI: 3.6; 10.1) on day 2 at 1.5 hours post-dose. None of the subjects had a change exceeding 60 msec or a QTcLD exceeding 500 msec at any time during this study. For the three fixed-dose efficacy studies in subjects with schizophrenia, electrocardiogram (ECG) measurements taken at various time points showed only one subject in the INVEGA® 12 mg group had a change exceeding 60 msec at one time-point on Day 6 (increase of 62 msec). No subject receiving INVEGA® had a QTcLD exceeding 500 msec at any time in any of these three studies.
Tardive Dyskinesia
A syndrome of potentially irreversible, involuntary, dyskinetic movements may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown. The risk of developing tardive dyskinesia and the likelihood that it will become irreversible appear to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase, but the syndrome can develop after relatively brief treatment periods at low doses, although this is uncommon. There is no known treatment for established tardive dyskinesia, although the syndrome may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment itself may suppress (or partially suppress) the signs and symptoms of the syndrome and may thus mask the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown. Given these considerations, INVEGA® should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that is known to respond to antipsychotic drugs. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically. If signs and symptoms of tardive dyskinesia appear in a patient treated with INVEGA®, drug discontinuation should be considered. However, some patients may require treatment with INVEGA® despite the presence of the syndrome.
Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and body weight gain. While all of the drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile. Hyperglycemia and Diabetes Mellitus Hyperglycemia and diabetes mellitus, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, have been reported in patients treated with all atypical antipsychotics. These cases were, for the most part, seen in post-marketing clinical use and epidemiologic studies, not in clinical trials, and there have been few reports of hyperglycemia or diabetes in trial subjects treated with INVEGA®. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population. Given these confounders, the relationship between atypical antipsychotic use and hyperglycemia-related adverse events is not completely understood. However, epidemiological studies suggest an increased risk of treatment-emergent hyperglycemia-related adverse events in patients treated with the atypical antipsychotics. Because INVEGA® was not marketed at the time these studies were performed, it is not known if INVEGA® is associated with this increased risk. Patients with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics should be monitored regularly for worsening of glucose control. Patients with risk factors for diabetes mellitus (e.g., obesity, family history of diabetes) who are starting treatment with atypical antipsychotics should undergo fasting blood glucose testing at the beginning of treatment and periodically during treatment. Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Patients who develop symptoms of hyperglycemia during treatment with atypical antipsychotics should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients required continuation of anti-diabetic treatment despite discontinuation of the suspect drug. Pooled data from the three placebo-controlled, 6-week, fixed-dose studies in adult subjects with schizophrenia are presented in Table 1a.
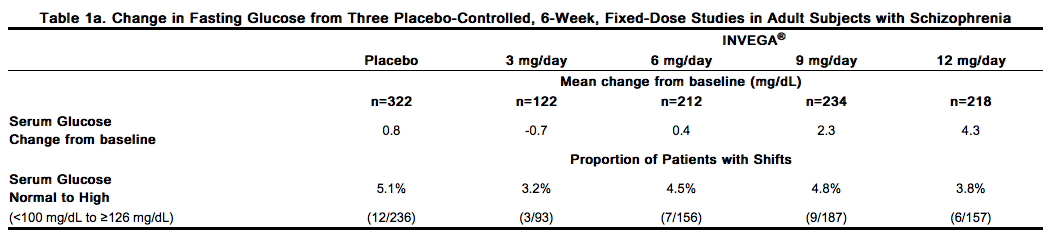
In the uncontrolled, longer-term open-label extension studies, INVEGA® was associated with a mean change in glucose of +3.3 mg/dL at Week 24 (n=570) and +4.6 mg/dL at Week 52 (n=314). Data from the placebo-controlled 6-week study in adolescent subjects (12–17 years of age) with schizophrenia are presented in Table 1b.
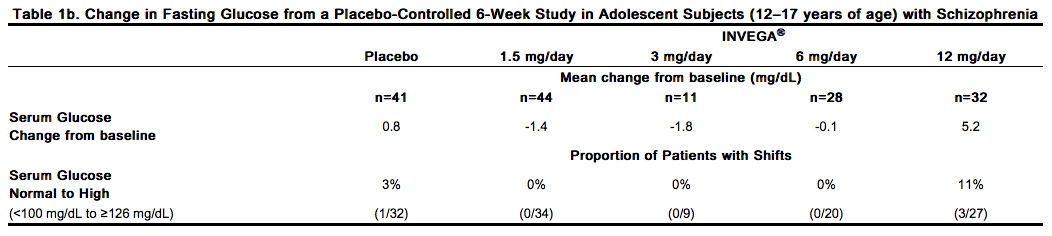
Dyslipidemia
Undesirable alterations in lipids have been observed in patients treated with atypical antipsychotics. Pooled data from the three placebo-controlled, 6-week, fixed-dose studies in adult subjects with schizophrenia are presented in Table 2a.

In the uncontrolled, longer-term open-label extension studies, INVEGA® was associated with a mean change in (a) total cholesterol of -1.5 mg/dL at Week 24 (n=573) and -1.5 mg/dL at Week 52 (n=317), (b) triglycerides of -6.4 mg/dL at Week 24 (n=573) and -10.5 mg/dL at Week 52 (n=317); (c) LDL of -1.9 mg/dL at Week 24 (n=557) and -2.7 mg/dL at Week 52 (n=297); and (d) HDL of +2.2 mg/dL at Week 24 (n=568) and +3.6 mg/dL at Week 52 (n=302). Data from the placebo-controlled 6-week study in adolescent subjects (12–17 years of age) with schizophrenia are presented in Table 2b.
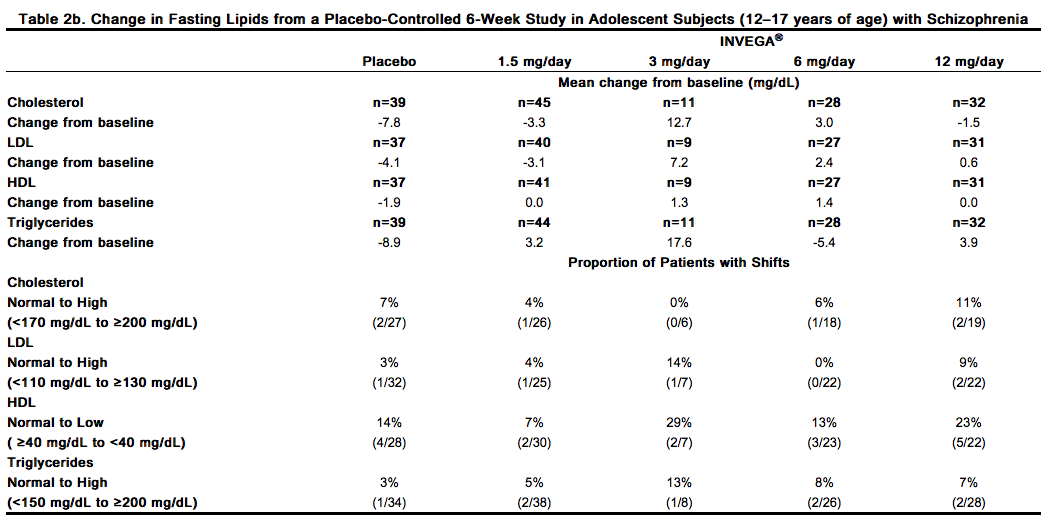
Weight Gain
Weight gain has been observed with atypical antipsychotic use. Clinical monitoring of weight is recommended. Schizophrenia Trials Data on mean changes in body weight and the proportion of subjects meeting a weight gain criterion of ≥ 7% of body weight from the three placebo-controlled, 6-week, fixed-dose studies in adult subjects are presented in Table 3a.
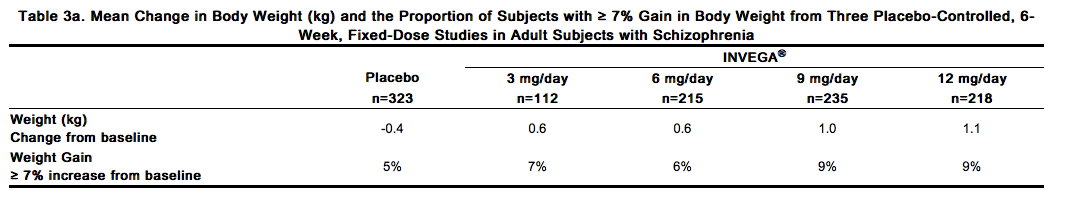
In the uncontrolled, longer-term open-label extension studies, INVEGA® was associated with a mean change in weight of +1.4 kg at Week 24 (n=63) and +2.6 kg at Week 52 (n=302). Weight gain in adolescent subjects with schizophrenia was assessed in a 6-week, double-blind, placebo-controlled study and an open-label extension with a median duration of exposure to INVEGA® of 182 days. Data on mean changes in body weight and the proportion of subjects meeting a weight gain criterion of ≥ 7% of body weight [see Clinical Studies (14.1)] from the placebo-controlled 6-week study in adolescent subjects (12–17 years of age) are presented in Table 3b.
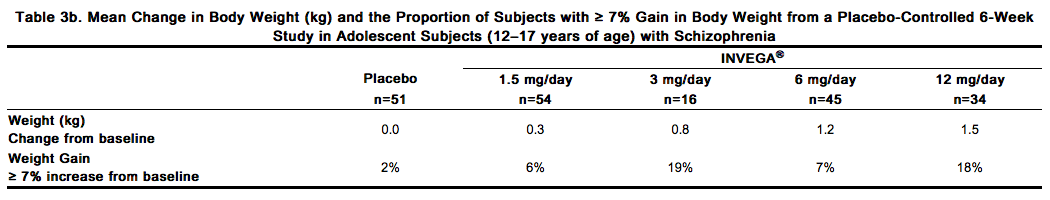
In the open-label long-term study the proportion of total subjects treated with INVEGA® with an increase in body weight of ≥ 7% from baseline was 33%. When treating adolescent patients with INVEGA®, weight gain should be assessed against that expected with normal growth. When taking into consideration the median duration of exposure to INVEGA® in the open-label study (182 days) along with expected normal growth in this population based on age and gender, an assessment of standardized scores relative to normative data provides a more clinically relevant measure of changes in weight. The mean change from open-label baseline to endpoint in standardized score for weight was 0.1 (4% above the median for normative data). Based on comparison to the normative data, these changes are not considered to be clinically significant. Schizoaffective Disorder Trials In the pooled data from the two placebo-controlled, 6-week studies in adult subjects with schizoaffective disorder, a higher percentage of INVEGA®-treated subjects (5%) had an increase in body weight of ≥ 7% compared with placebo-treated subjects (1%). In the study that examined high- and low-dose groups, the increase in body weight of ≥ 7% was 3% in the low-dose group, 7% in the high-dose group, and 1% in the placebo group.
Hyperprolactinemia
Like other drugs that antagonize dopamine D2 receptors, paliperidone elevates prolactin levels and the elevation persists during chronic administration. Paliperidone has a prolactin-elevating effect similar to that seen with risperidone, a drug that is associated with higher levels of prolactin than other antipsychotic drugs. Hyperprolactinemia, regardless of etiology, may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotrophin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported in patients receiving prolactin-elevating compounds. Long-standing hyperprolactinemia when associated with hypogonadism may lead to decreased bone density in both female and male subjects. Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin dependent in vitro, a factor of potential importance if the prescription of these drugs is considered in a patient with previously detected breast cancer. An increase in the incidence of pituitary gland, mammary gland, and pancreatic islet cell neoplasia (mammary adenocarcinomas, pituitary and pancreatic adenomas) was observed in the risperidone carcinogenicity studies conducted in mice and rats [see Nonclinical Toxicology (13.1)]. Neither clinical studies nor epidemiologic studies conducted to date have shown an association between chronic administration of this class of drugs and tumorigenesis in humans, but the available evidence is too limited to be conclusive.
Potential for Gastrointestinal Obstruction
Because the INVEGA® tablet is non-deformable and does not appreciably change in shape in the gastrointestinal tract, INVEGA® should ordinarily not be administered to patients with pre-existing severe gastrointestinal narrowing (pathologic or iatrogenic, for example: esophageal motility disorders, small bowel inflammatory disease, "short gut" syndrome due to adhesions or decreased transit time, past history of peritonitis, cystic fibrosis, chronic intestinal pseudoobstruction, or Meckel's diverticulum). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of drugs in non-deformable controlled-release formulations. Because of the controlled-release design of the tablet, INVEGA® should only be used in patients who are able to swallow the tablet whole. A decrease in transit time, e.g., as seen with diarrhea, would be expected to decrease bioavailability and an increase in transit time, e.g., as seen with gastrointestinal neuropathy, diabetic gastroparesis, or other causes, would be expected to increase bioavailability. These changes in bioavailability are more likely when the changes in transit time occur in the upper GI tract.
Orthostatic Hypotension and Syncope
Paliperidone can induce orthostatic hypotension and syncope in some patients because of its alpha-blocking activity. In pooled results of the three placebo-controlled, 6-week, fixed-dose trials in subjects with schizophrenia, syncope was reported in 0.8% (7/850) of subjects treated with INVEGA® (3 mg, 6 mg, 9 mg, 12 mg) compared to 0.3% (1/355) of subjects treated with placebo. INVEGA® should be used with caution in patients with known cardiovascular disease (e.g., heart failure, history of myocardial infarction or ischemia, conduction abnormalities), cerebrovascular disease, or conditions that predispose the patient to hypotension (e.g., dehydration, hypovolemia, and treatment with antihypertensive medications). Monitoring of orthostatic vital signs should be considered in patients who are vulnerable to hypotension.
Leukopenia, Neutropenia, and Agranulocytosis
Class Effect: In clinical trial and/or postmarketing experience, events of leukopenia/neutropenia have been reported temporally related to antipsychotic agents, including INVEGA®. Agranulocytosis has also been reported. Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC) and history of drug-induced leukopenia/neutropenia. Patients with a history of a clinically significant low WBC or a drug-induced leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and discontinuation of INVEGA® should be considered at the first sign of a clinically significant decline in WBC in the absence of other causative factors. Patients with clinically significant neutropenia should be carefully monitored for fever or other symptoms or signs of infection and treated promptly if such symptoms or signs occur. Patients with severe neutropenia (absolute neutrophil count <1000/mm3) should discontinue INVEGA® and have their WBC followed until recovery.
Potential for Cognitive and Motor Impairment
Somnolence was reported in subjects treated with INVEGA® [see Adverse Reactions (6.1, 6.2)]. Antipsychotics, including INVEGA®, have the potential to impair judgment, thinking, or motor skills. Patients should be cautioned about performing activities requiring mental alertness, such as operating hazardous machinery or operating a motor vehicle, until they are reasonably certain that paliperidone therapy does not adversely affect them.
Seizures
During premarketing clinical trials in subjects with schizophrenia (the three placebo-controlled, 6-week, fixed-dose studies and a study conducted in elderly schizophrenic subjects), seizures occurred in 0.22% of subjects treated with INVEGA® (3 mg, 6 mg, 9 mg, 12 mg) and 0.25% of subjects treated with placebo. Like other antipsychotic drugs, INVEGA® should be used cautiously in patients with a history of seizures or other conditions that potentially lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in patients 65 years or older.
Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Aspiration pneumonia is a common cause of morbidity and mortality in patients with advanced Alzheimer's dementia. INVEGA® and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia.
Suicide
The possibility of suicide attempt is inherent in psychotic illnesses, and close supervision of high-risk patients should accompany drug therapy. Prescriptions for INVEGA® should be written for the smallest quantity of tablets consistent with good patient management in order to reduce the risk of overdose.
Priapism
Drugs with alpha-adrenergic blocking effects have been reported to induce priapism. Priapism has been reported with INVEGA® during postmarketing surveillance. Severe priapism may require surgical intervention.
Thrombotic Thrombocytopenic Purpura (TTP)
No cases of TTP were observed during clinical studies with paliperidone. Although cases of TTP have been reported in association with risperidone administration, the relationship to risperidone therapy is unknown.
Body Temperature Regulation
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing INVEGA® to patients who will be experiencing conditions which may contribute to an elevation in core body temperature, e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration.
Antiemetic Effect
An antiemetic effect was observed in preclinical studies with paliperidone. This effect, if it occurs in humans, may mask the signs and symptoms of overdosage with certain drugs or of conditions such as intestinal obstruction, Reye's syndrome, and brain tumor.
Use in Patients with Concomitant Illness
Clinical experience with INVEGA® in patients with certain concomitant illnesses is limited [see Clinical Pharmacology (12.3)]. Patients with Parkinson's Disease or Dementia with Lewy Bodies are reported to have an increased sensitivity to antipsychotic medication. Manifestations of this increased sensitivity include confusion, obtundation, postural instability with frequent falls, extrapyramidal symptoms, and clinical features consistent with the neuroleptic malignant syndrome. INVEGA® has not been evaluated or used to any appreciable extent in patients with a recent history of myocardial infarction or unstable heart disease. Patients with these diagnoses were excluded from premarketing clinical trials. Because of the risk of orthostatic hypotension with INVEGA®, caution should be observed in patients with known cardiovascular disease [see Warnings and Precautions (5.9)].
Monitoring: Laboratory Tests
No specific laboratory tests are recommended.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Paliperidone (oral) Clinical Trials Experience in the drug label.
Postmarketing Experience
There is limited information regarding Paliperidone (oral) Postmarketing Experience in the drug label.
Drug Interactions
There is limited information regarding Paliperidone (oral) Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Paliperidone (oral) in women who are pregnant.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Paliperidone (oral) in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Paliperidone (oral) during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Paliperidone (oral) in women who are nursing.
Pediatric Use
There is no FDA guidance on the use of Paliperidone (oral) in pediatric settings.
Geriatic Use
There is no FDA guidance on the use of Paliperidone (oral) in geriatric settings.
Gender
There is no FDA guidance on the use of Paliperidone (oral) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Paliperidone (oral) with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Paliperidone (oral) in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Paliperidone (oral) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Paliperidone (oral) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Paliperidone (oral) in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Paliperidone (oral) Administration in the drug label.
Monitoring
There is limited information regarding Paliperidone (oral) Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Paliperidone (oral) and IV administrations.
Overdosage
There is limited information regarding Paliperidone (oral) overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology
There is limited information regarding Paliperidone (oral) Pharmacology in the drug label.
Mechanism of Action
There is limited information regarding Paliperidone (oral) Mechanism of Action in the drug label.
Structure
There is limited information regarding Paliperidone (oral) Structure in the drug label.
Pharmacodynamics
There is limited information regarding Paliperidone (oral) Pharmacodynamics in the drug label.
Pharmacokinetics
There is limited information regarding Paliperidone (oral) Pharmacokinetics in the drug label.
Nonclinical Toxicology
There is limited information regarding Paliperidone (oral) Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Paliperidone (oral) Clinical Studies in the drug label.
How Supplied
There is limited information regarding Paliperidone (oral) How Supplied in the drug label.
Storage
There is limited information regarding Paliperidone (oral) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Paliperidone (oral) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Paliperidone (oral) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Paliperidone (oral) Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Paliperidone interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Paliperidone (oral) Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Paliperidone (oral) Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.