Dapagliflozin / saxagliptin: Difference between revisions
No edit summary |
No edit summary |
||
| Line 317: | Line 317: | ||
|packLabel=[[image:Dapagliflozin-saxagliptin_Package_Label.jpeg|none|thumb|400px|This image is provided by the National Library of Medicine.]] | |packLabel=[[image:Dapagliflozin-saxagliptin_Package_Label.jpeg|none|thumb|400px|This image is provided by the National Library of Medicine.]] | ||
|fdaPatientInfo=( | |fdaPatientInfo======Pancreatitis===== | ||
*Inform patients that acute pancreatitis has been reported during postmarketing use of saxagliptin. Inform patients that persistent severe abdominal pain, sometimes radiating to the back, which may or may not be accompanied by vomiting, is the hallmark symptom of acute pancreatitis. | |||
*Instruct patients to promptly discontinue QTERN and contact their healthcare provider if persistent severe abdominal pain occurs. | |||
=====Heart Failure===== | |||
*Inform patients of the signs and symptoms of heart failure. Instruct patients to contact their healthcare provider as soon as possible if they experience symptoms of heart failure, including increasing shortness of breath, rapid increase in weight or swelling of the feet. | |||
=====Hypotension===== | |||
*Inform patients that symptomatic hypotension may occur with QTERN and advise them to contact their healthcare provider if they experience such symptoms. Inform patients that dehydration may increase the risk for hypotension, and to have adequate fluid intake. | |||
=====Ketoacidosis===== | |||
*Inform patients that ketoacidosis is a serious life-threatening condition. Cases of ketoacidosis have been reported during use of dapagliflozin. Instruct patients to check ketones (when possible) if symptoms consistent with ketoacidosis occur even if blood glucose is not elevated. If symptoms of ketoacidosis (including nausea, vomiting, abdominal pain, tiredness and labored breathing) occur, instruct patients to discontinue QTERN and seek medical advice immediately. | |||
=====Acute Kidney Injury===== | |||
*Inform patients that acute kidney injury has been reported during use of dapagliflozin. Advise patients to seek medical advice immediately if they have reduced oral intake (due to acute illness or fasting) or increased fluid losses (due to vomiting, diarrhea, or excessive heat exposure), as it may be appropriate to temporarily discontinue QTERN use in those settings. | |||
=====Serious Urinary Tract Infections===== | |||
*Inform patients of the potential for urinary tract infections, which may be serious. Inform them of the symptoms of urinary tract infections and advise them to seek medical advice if symptoms occur. | |||
=====Hypersensitivity Reactions===== | |||
*Inform patients that serious hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, and exfoliative skin conditions) have been reported with dapagliflozin and saxagliptin, components of QTERN. Symptoms of these allergic reactions include: rash, skin flaking or peeling, urticaria, swelling of the skin, or swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing. | |||
*Advise patients to immediately report any signs or symptoms suggesting allergic reaction, angioedema or exfoliative skin conditions, and stop taking QTERN and seek medical advice promptly. | |||
=====Genital Mycotic Infections in Females (e.g., Vulvovaginitis)===== | |||
*Inform female patients that vaginal yeast infections may occur and provide them with information on the signs and symptoms of vaginal yeast infections. Advise them of treatment options and when to seek medical advice. | |||
=====Genital Mycotic Infections (e.g., Balanitis)===== | |||
*Inform male patients that yeast infections of the penis (e.g., balanitis or balanoposthitis) may occur, especially in patients with prior history. Provide them with information on the signs and symptoms of balanitis and balanoposthitis (rash or redness of the glans or foreskin of the penis). Advise them of treatment options and when to seek medical advice. | |||
=====Bladder Cancer===== | |||
*Inform patients to promptly report any signs of macroscopic hematuria or other symptoms potentially related to bladder cancer. | |||
=====Severe and Disabling Arthralgia===== | |||
*Inform patients that severe and disabling joint pain may occur with this class of drugs. The time to onset of symptoms can range from one day to years. Instruct patients to seek medical advice if severe joint pain occurs. | |||
=====Bullous Pemphigoid===== | |||
*Inform patients that bullous pemphigoid may occur with QTERN. Instruct patients to seek medical advice if blisters or erosions occur. | |||
=====Pregnancy and Lactating Mothers===== | |||
*Advise pregnant patients of the potential risk to a fetus with treatment with QTERN. Instruct patients to immediately inform their healthcare provider if pregnant or planning to become pregnant. Advise patients that use of QTERN is not recommended while breastfeeding. | |||
=====Laboratory Tests===== | |||
*Inform patients that due to its mechanism of action, patients taking QTERN will test positive for glucose in their urine. | |||
=====Missed Dose===== | |||
*Patients should be informed that if they miss a dose of QTERN they should take the next dose as prescribed, unless otherwise instructed by their healthcare provider. Patients should be instructed not to take an extra dose the next day. | |||
|nlmPatientInfo=(Link to patient information page) | |nlmPatientInfo=(Link to patient information page) | ||
|lookAlike= | |lookAlike= | ||
Revision as of 13:58, 13 July 2018
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Yashasvi Aryaputra[2];
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Warning Title
See full prescribing information for complete Boxed Warning.
Condition Name: (Content)
|
Overview
Dapagliflozin / saxagliptin is a Acetylcholine release inhibitor, Adrenergic receptor agonist that is FDA approved for the (type of indication of drug) of a list of indications, separated by commas.. There is a Black Box Warning for this drug as shown here. Common adverse reactions include a list of adverse reactions, separated by commas..
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Contraindications
CONTRAINDICATIONS
Warnings
|
Warning Title
See full prescribing information for complete Boxed Warning.
Condition Name: (Content)
|
Conidition 1
(Description)
Conidition 2
(Description)
Conidition 3
(Description)
Adverse Reactions
Clinical Trials Experience
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Condition 2
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Postmarketing Experience
(Description)
Drug Interactions
- Drug 1
- Drug 2
- Drug 3
- Drug 4
- Drug 5
Drug 1
(Description)
Drug 2
(Description)
Drug 3
(Description)
Drug 4
(Description)
Drug 5
(Description)
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
(Description)
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Dapagliflozin / saxagliptin in women who are pregnant.
Labor and Delivery
(Description)
Nursing Mothers
(Description)g
Pediatric Use
(Description)
Geriatic Use
(Description)
Gender
(Description)
Race
(Description)
Renal Impairment
(Description)
Hepatic Impairment
(Description)
Females of Reproductive Potential and Males
(Description)
Immunocompromised Patients
(Description)
Others
(Description)
Administration and Monitoring
Administration
(Oral/Intravenous/etc)
Monitoring
Condition 1
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section)
IV Compatibility
There is limited information regarding the compatibility of Dapagliflozin / saxagliptin and IV administrations.
Overdosage
Acute Overdose
Signs and Symptoms
(Description)
Management
(Description)
Chronic Overdose
Signs and Symptoms
(Description)
Management
(Description)
Pharmacology
Dapagliflozin / saxagliptin
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
(Description)
Structure
(Description with picture)
Pharmacodynamics
(Description)
Pharmacokinetics
(Description)
Nonclinical Toxicology
(Description)
Clinical Studies
Condition 1
(Description)
Condition 2
(Description)
Condition 3
(Description)
How Supplied
(Description)
Storage
(Description)
Images
Drug Images
{{#ask: Page Name::Dapagliflozin / saxagliptin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
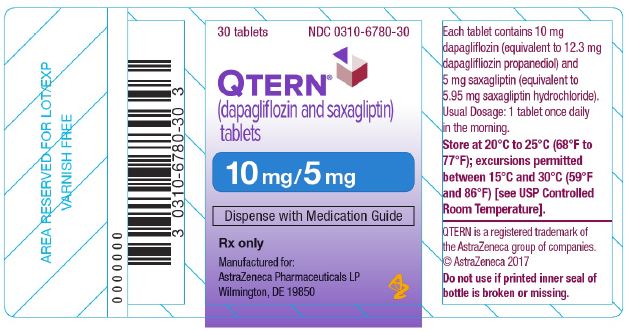
{{#ask: Label Page::Dapagliflozin / saxagliptin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Pancreatitis
- Inform patients that acute pancreatitis has been reported during postmarketing use of saxagliptin. Inform patients that persistent severe abdominal pain, sometimes radiating to the back, which may or may not be accompanied by vomiting, is the hallmark symptom of acute pancreatitis.
- Instruct patients to promptly discontinue QTERN and contact their healthcare provider if persistent severe abdominal pain occurs.
Heart Failure
- Inform patients of the signs and symptoms of heart failure. Instruct patients to contact their healthcare provider as soon as possible if they experience symptoms of heart failure, including increasing shortness of breath, rapid increase in weight or swelling of the feet.
Hypotension
- Inform patients that symptomatic hypotension may occur with QTERN and advise them to contact their healthcare provider if they experience such symptoms. Inform patients that dehydration may increase the risk for hypotension, and to have adequate fluid intake.
Ketoacidosis
- Inform patients that ketoacidosis is a serious life-threatening condition. Cases of ketoacidosis have been reported during use of dapagliflozin. Instruct patients to check ketones (when possible) if symptoms consistent with ketoacidosis occur even if blood glucose is not elevated. If symptoms of ketoacidosis (including nausea, vomiting, abdominal pain, tiredness and labored breathing) occur, instruct patients to discontinue QTERN and seek medical advice immediately.
Acute Kidney Injury
- Inform patients that acute kidney injury has been reported during use of dapagliflozin. Advise patients to seek medical advice immediately if they have reduced oral intake (due to acute illness or fasting) or increased fluid losses (due to vomiting, diarrhea, or excessive heat exposure), as it may be appropriate to temporarily discontinue QTERN use in those settings.
Serious Urinary Tract Infections
- Inform patients of the potential for urinary tract infections, which may be serious. Inform them of the symptoms of urinary tract infections and advise them to seek medical advice if symptoms occur.
Hypersensitivity Reactions
- Inform patients that serious hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, and exfoliative skin conditions) have been reported with dapagliflozin and saxagliptin, components of QTERN. Symptoms of these allergic reactions include: rash, skin flaking or peeling, urticaria, swelling of the skin, or swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing.
- Advise patients to immediately report any signs or symptoms suggesting allergic reaction, angioedema or exfoliative skin conditions, and stop taking QTERN and seek medical advice promptly.
Genital Mycotic Infections in Females (e.g., Vulvovaginitis)
- Inform female patients that vaginal yeast infections may occur and provide them with information on the signs and symptoms of vaginal yeast infections. Advise them of treatment options and when to seek medical advice.
Genital Mycotic Infections (e.g., Balanitis)
- Inform male patients that yeast infections of the penis (e.g., balanitis or balanoposthitis) may occur, especially in patients with prior history. Provide them with information on the signs and symptoms of balanitis and balanoposthitis (rash or redness of the glans or foreskin of the penis). Advise them of treatment options and when to seek medical advice.
Bladder Cancer
- Inform patients to promptly report any signs of macroscopic hematuria or other symptoms potentially related to bladder cancer.
Severe and Disabling Arthralgia
- Inform patients that severe and disabling joint pain may occur with this class of drugs. The time to onset of symptoms can range from one day to years. Instruct patients to seek medical advice if severe joint pain occurs.
Bullous Pemphigoid
- Inform patients that bullous pemphigoid may occur with QTERN. Instruct patients to seek medical advice if blisters or erosions occur.
Pregnancy and Lactating Mothers
- Advise pregnant patients of the potential risk to a fetus with treatment with QTERN. Instruct patients to immediately inform their healthcare provider if pregnant or planning to become pregnant. Advise patients that use of QTERN is not recommended while breastfeeding.
Laboratory Tests
- Inform patients that due to its mechanism of action, patients taking QTERN will test positive for glucose in their urine.
Missed Dose
- Patients should be informed that if they miss a dose of QTERN they should take the next dose as prescribed, unless otherwise instructed by their healthcare provider. Patients should be instructed not to take an extra dose the next day.
Precautions with Alcohol
Alcohol-Dapagliflozin / saxagliptin interaction has not been established. Talk to your doctor regarding the effects of taking alcohol with this medication.
Brand Names
- Qtern
Look-Alike Drug Names
There is limited information regarding Dapagliflozin / saxagliptin Look-Alike Drug Names in the drug label.
Drug Shortage Status
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.