Burosumab: Difference between revisions
No edit summary |
No edit summary |
||
| Line 225: | Line 225: | ||
(Description regarding monitoring, from ''Warnings'' section) | (Description regarding monitoring, from ''Warnings'' section) | ||
|overdose= | |overdose=*There have been no reports of overdose with CRYSVITA. CRYSVITA has been administered in pediatric clinical trials without dose limiting toxicity using doses up to 2 mg/kg body weight with a maximal dose of 90 mg, administered every two weeks. In adult clinical trials, no dose limiting toxicity has been observed using doses up to 1 mg/kg or a maximal total dose of 128 mg every 4 weeks. In non-XLH rabbits and cynomolgus monkeys, ectopic mineralization in multiple tissues and organs was observed at doses of burosumab-twza that resulted in supra-physiologic serum phosphate levels. Adverse effects on bone including reductions in bone mineral density, bone mineralization and bone strength were also observed at exposure greater than human exposure. | ||
*In case of overdose, it is recommended that serum phosphorus levels, serum calcium levels and renal function be measured immediately and monitored periodically until resolution to normal/baseline levels. In case of hyperphosphatemia, withhold CRYSVITA and initiate appropriate medical treatment. | |||
|drugBox= | |drugBox= | ||
Revision as of 14:37, 6 July 2018
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1];
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Warning Title
See full prescribing information for complete Boxed Warning.
Condition Name: (Content)
|
Overview
Burosumab is a Acetylcholine release inhibitor, Adrenergic receptor agonist that is FDA approved for the (type of indication of drug) of a list of indications, separated by commas.. There is a Black Box Warning for this drug as shown here. Common adverse reactions include a list of adverse reactions, separated by commas..
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Contraindications
CONTRAINDICATIONS
Warnings
|
Warning Title
See full prescribing information for complete Boxed Warning.
Condition Name: (Content)
|
Conidition 1
(Description)
Conidition 2
(Description)
Conidition 3
(Description)
Adverse Reactions
Clinical Trials Experience
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Condition 2
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Postmarketing Experience
(Description)
Drug Interactions
- Drug 1
- Drug 2
- Drug 3
- Drug 4
- Drug 5
Drug 1
(Description)
Drug 2
(Description)
Drug 3
(Description)
Drug 4
(Description)
Drug 5
(Description)
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
(Description)
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Burosumab in women who are pregnant.
Labor and Delivery
(Description)
Nursing Mothers
(Description)g
Pediatric Use
(Description)
Geriatic Use
(Description)
Gender
(Description)
Race
(Description)
Renal Impairment
(Description)
Hepatic Impairment
(Description)
Females of Reproductive Potential and Males
(Description)
Immunocompromised Patients
(Description)
Others
(Description)
Administration and Monitoring
Administration
(Oral/Intravenous/etc)
Monitoring
Condition 1
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section)
IV Compatibility
There is limited information regarding the compatibility of Burosumab and IV administrations.
Overdosage
- There have been no reports of overdose with CRYSVITA. CRYSVITA has been administered in pediatric clinical trials without dose limiting toxicity using doses up to 2 mg/kg body weight with a maximal dose of 90 mg, administered every two weeks. In adult clinical trials, no dose limiting toxicity has been observed using doses up to 1 mg/kg or a maximal total dose of 128 mg every 4 weeks. In non-XLH rabbits and cynomolgus monkeys, ectopic mineralization in multiple tissues and organs was observed at doses of burosumab-twza that resulted in supra-physiologic serum phosphate levels. Adverse effects on bone including reductions in bone mineral density, bone mineralization and bone strength were also observed at exposure greater than human exposure.
- In case of overdose, it is recommended that serum phosphorus levels, serum calcium levels and renal function be measured immediately and monitored periodically until resolution to normal/baseline levels. In case of hyperphosphatemia, withhold CRYSVITA and initiate appropriate medical treatment.
Pharmacology
Mechanism of Action
- X-linked hypophosphatemia is caused by excess fibroblast growth factor 23 (FGF23) which suppresses renal tubular phosphate reabsorption and the renal production of 1,25 dihydroxy vitamin D. Burosumab-twza binds to and inhibits the biological activity of FGF23 restoring renal phosphate reabsorption and increasing the serum concentration of 1,25 dihydroxy vitamin D.
Structure
There is limited information regarding Burosumab Structure in the drug label.
Pharmacodynamics
- Following SC administration in XLH patients, higher burosumab-twza concentrations were associated with greater increase of serum phosphorus levels. The increase in serum phosphorus was reversible and returned to baseline with elimination of systemic burosumab-twza.
- Ratio of renal tubular maximum reabsorption rate of phosphate to glomerular filtration rate (TmP/GFR) showed dose-dependent increases from baseline.
- Elevation in serum total FGF23 was observed after initiation of burosumab-twza treatment, however, the clinical implication is unknown.
Pharmacokinetics
- The following pharmacokinetic parameters were observed in patients with XLH administered the approved recommended starting dosage based on a 70 kg patient, unless otherwise specified.
- Burosumab-twza exhibited linear pharmacokinetics following SC injections within the dose range of 0.1 to 1 mg/kg (0.08 to 0.8 times the maximum approved recommended dosage based on a 70 kg patient).
- The steady-state trough mean (± SD) concentration of burosumab-twza was 5.8 (± 3.4) mcg/mL in adult patients.
Absorption
- The burosumab-twza mean Tmax values ranged from 8 to 11 days.
Distribution
- The apparent volume of distribution of burosumab-twza is 8 L.
Elimination
- The apparent clearance is 0.290 L/day. The half-life of burosumab-twza is approximately 19 days.
Metabolism
- The exact pathway for burosumab-twza metabolism has not been characterized. Burosumab-twza is expected to be degraded into small peptides and amino acids via catabolic pathways.
Specific Populations
- No clinical significant difference in burosumab-twza pharmacokinetics was observed based on age.
- The effect of renal or hepatic impairment on the pharmacokinetics of burosumab-twza is unknown.
Pediatric Patients
- The steady-state trough concentration was 15.8 (± 9.4) mcg/mL in patients aged 5-12 years, and 11.2 (± 4.6) mcg/mL in patients aged 1-4 years.
Body Weight
- Clearance and volume of distribution of burosumab-twza increases with body weight.
Drug Interaction Studies
- No drug interaction studies have been conducted with CRYSVITA.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- The carcinogenic potential of burosumab-twza has not been evaluated in long term animal studies.
- Studies have not been performed to evaluate the mutagenic potential of burosumab-twza.
- No specific fertility studies have been performed in animals to evaluate the effects of burosumab-twza.
- Toxicology studies with burosumab-twza of up to 40 weeks duration in non-XLH cynomolgus monkeys did not show significant adverse effects on female reproductive organs at doses up to 65-fold human exposure at the dose of 1 mg/kg every 4 weeks. In male monkeys, minimal mineralization of the rete testis or seminiferous tubules associated with hyperphosphatemia was observed at 11- to 37-fold human exposure, but semen analysis did not show any adverse effects.
Animal Toxicology and/or Pharmacology
- In rabbits and cynomolgus monkeys, inhibition of FGF23 signaling by burosumab-twza increased serum phosphate and 1,25 dihydroxy vitamin D. Ectopic mineralization in multiple tissues and organs was observed at doses of burosumab-twza that resulted in supra-physiologic serum phosphate levels in the non-XLH animals. In a study in wild type (WT) and hypophosphatemic Hyp mice, a murine model of XLH, ectopic mineralization was markedly less in Hyp mice.
- In adult cynomolgus monkeys, burosumab-twza increased bone turnover, mineral content and/or mineral density and cortical thickness at 37- to 65-fold human exposure at the dose of 1 mg/kg every 4 weeks. Adverse effects on bone including reductions in bone mineral density, bone mineralization and bone strength were observed in adult male monkeys at 37- to 47-fold human exposure at the dose of 1 mg/kg every 4 weeks.
- In juvenile cynomolgus monkeys, burosumab-twza increased bone turnover, mineral content and/or mineral density and/or cortical thickness at 0.5- to 5-fold clinical pediatric exposure. Bone mineralization was decreased in a male monkey at 5-fold pediatric exposure but there was no effect on bone strength. Burosumab-twza did not affect bone development in juvenile monkeys at doses up to 5-fold pediatric exposure.
Clinical Studies
Pediatric X-linked Hypophosphatemia
- CRYSVITA has been evaluated in 65 pediatric patients with XLH.
- Study 1 (NCT 02163577) is a randomized, open-label study in 52 prepubescent XLH patients, 5 to 12 years old, which compared treatment with CRYSVITA administered every 2 weeks versus every 4 weeks. Following an initial 16-week dose titration phase, patients completed 48-weeks of treatment with CRYSVITA every 2 weeks. All 52 patients completed at least 64 weeks on study; no patient discontinued. Burosumab-twza dose was adjusted to target a fasting serum phosphorus concentration of 3.5 to 5.0 mg/dL based on the fasting phosphorus level the day of dosing. Twenty-six of 52 patients received CRYSVITA every two weeks up to a maximum dose of 2 mg/kg. The average dose was 0.73 mg/kg (range: 0.3, 1.5) at week 16, 0.98 mg/kg (range: 0.4, 2.0) at week 40 and 1.04 mg/kg (range: 0.4, 2.0) at week 60. The remaining 26 patients received CRYSVITA every four weeks. At study entry, the mean age of patients was 8.5 years and 46% were male. Ninety-six percent had received oral phosphate and active vitamin D analogs for a mean (SD) duration of 7 (2.4) years. Oral phosphate and active vitamin D analogs were discontinued prior to study enrollment. Ninety-four percent of patients had radiographic evidence of rickets at baseline.
- Study 2 (NCT 02750618) is a 64-week open-label study in 13 pediatric XLH patients, 1 to 4 years old. Patients received CRYSVITA at a dose of 0.8 mg/kg every two weeks with titration up to 1.2 mg/kg based on serum phosphorus measurements. All patients completed at least 40 weeks on study; no patients discontinued. At study entry, the mean age of patients was 2.9 years and 69% were male. All patients had radiographic evidence of rickets at baseline and had received oral phosphate and active vitamin D analogs for a mean (SD) duration of 16.9 (13.9) months. Oral phosphate and active vitamin D analogs were discontinued prior to study enrollment.
Serum Phosphorus
- In Study 1, CRYSVITA increased mean (SD) serum phosphorus levels from 2.4 (0.40) at baseline to 3.3 (0.40) and 3.4 (0.45) mg/dL at week 40 and week 64 in the patients who received CRYSVITA every 2 weeks (Figure 1). The ratio of renal tubular maximum reabsorption rate of phosphate to glomerular filtration rate (TmP/GFR) increased in these patients from mean (SD) of 2.2 (0.49) at baseline to 3.3 (0.60) and 3.4 (0.53) mg/dL at week 40 and week 64.
- In Study 2, CRYSVITA increased mean (SD) serum phosphorus levels from 2.5 (0.28) mg/dL at baseline to 3.5 (0.49) mg/dL at week 40.

Radiographic Evaluation of Rickets
- Radiographs from 52 CRYSVITA-treated XLH patients in Study 1 and 13 patients in Study 2 were examined to assess XLH-related rickets using the 10-point Thacher Rickets Severity Score (RSS) and the 7-point Radiographic Global Impression of Change (RGI-C). The RSS score is assigned based on images of the wrist and knee from a single timepoint, with higher scores indicating greater rickets severity. The RGI-C score is assigned based on side-by-side comparisons of wrist and knee radiographs from two timepoints, with higher scores indicating greater improvement in radiographic evidence of rickets. A RGI-C score of +2.0 was defined as radiographic evidence of substantial healing.
- In Study 1, baseline mean (SD) RSS total score was 1.9 (1.17) in patients receiving CRYSVITA every two weeks. After 40 weeks of treatment with CRYSVITA, mean total RSS decreased from 1.9 to 0.8 (see Table 6). After 40 weeks of treatment with CRYSVITA, the mean RGI-C Global score was +1.7 in patients receiving CRYSVITA every two weeks. Eighteen out of 26 patients achieved an RGI-C score of ≥ +2.0. These findings were maintained at week 64 as shown in Table 6.
- In Study 2, baseline mean (SD) total RSS was 2.9 (1.37) in 13 patients. After 40 weeks of treatment with CRYSVITA, mean total RSS decreased from 2.9 to 1.2 and the mean (SE) RGI-C Global score was +2.3 (0.08). All 13 patients achieved a RGI-C global score ≥ +2.0. The mean (SE) lower limb deformity as assessed by RGI-C, using standing long leg radiographs, was +1.3 (0.14) (see Table 6).

Serum Alkaline Phosphatase Activity
- For Study 1, mean (SD) serum total alkaline phosphatase activity was 462 (110) U/L at baseline and decreased to 354 (73) U/L at Week 64 (-23%, p < 0.0001) in the patients who received CRYSVITA every 2 weeks.
- For Study 2, mean (SD) serum total alkaline phosphatase activity was 549 (194) U/L at baseline and decreased to 335 (88) U/L at Week 40 (mean change: -36%).
Growth
- In Study 1, CRYSVITA treatment for 64 weeks increased standing mean (SD) height Z score from -1.72 (1.03) at baseline to -1.54 (1.13) in the patients who received CRYSVITA every two weeks (LS mean change of +0.19 (95% CI: 0.09 to 0.29).
Adult X-linked Hypophosphatemia
- Study 3 (NCT 02526160) is a randomized, double-blind, placebo-controlled study in 134 adult XLH patients. The study comprises a 24-week placebo-controlled treatment phase. CRYSVITA was administered at a dose of 1 mg/kg every 4 weeks. At study entry, the mean age of patients was 40 years (range 19 to 66 years) and 35% were male. All patients had skeletal pain associated with XLH/osteomalacia at baseline. The baseline mean (SD) serum phosphorus concentration was below the lower limit of normal at 1.98 (0.31) mg/dL. Oral phosphate and active vitamin D analogs were not allowed during the study. One patient in the CRYSVITA group discontinued treatment.
- Study 4 (NCT 02537431) is a 48-week, open-label, single-arm study in 14 adult XLH patients to assess the effects of CRYSVITA on improvement of osteomalacia as determined by histologic and histomorphometric evaluation of iliac crest bone biopsies. Patients received 1 mg/kg CRYSVITA every four weeks. At study entry, the mean age of patients was 40 years (range 25 to 52 years) and 43% were male. Oral phosphate and active vitamin D analogs were not allowed during the study.
Serum Phosphorus
- In Study 3 at baseline, mean (SD) serum phosphorus was 1.9 (0.32) and 2.0 (0.30) mg/dL in the placebo and CRYSVITA groups respectively. During the initial 24 weeks of treatment, mean (SD) serum phosphorus across the midpoints of dose intervals (2 weeks post dose) was 2.1 (0.30) and 3.2 (0.53) mg/dL in the placebo and CRYSVITA groups, and mean (SD) serum phosphorus across the ends of dose intervals was 2.0 (0.30) and 2.7 (0.45) mg/dL in the placebo and CRYSVITA groups.
- A total of 94% of patients treated with CRYSVITA achieved a serum phosphorus level above the lower limit of normal (LLN) compared to 8% in the placebo group through week 24 (Table 7).

- At baseline, the mean (SD) ratio of renal tubular maximum reabsorption rate of phosphate to glomerular filtration rate (TmP/GFR) was 1.60 (0.37) and 1.68 (0.40) mg/dL in the placebo and CRYSVITA groups respectively. At week 22 (midpoint of a dose interval), mean (SD) TmP/GFR was 1.69 (0.37) and 2.73 (0.75) mg/dL in the placebo and CRYSVITA groups. At week 24 (end of a dose interval), mean (SD) TmP/GFR was 1.73 (0.42) and 2.21 (0.48) mg/dL in the placebo and CRYSVITA groups.
Radiographic Evaluation of Osteomalacia
- In Study 3, a skeletal survey was conducted at baseline to identify osteomalacia-related fractures and pseudofractures. Osteomalacia-related fractures are defined as atraumatic lucencies extending across both bone cortices and pseudofractures are defined as atraumatic lucencies extending across one cortex. There were 52% of patients who had either active (unhealed) fractures (12%) or active pseudofractures (47%) at baseline. The active fractures and pseudofractures were predominantly located in the femurs, tibia/fibula, and metatarsals of the feet. Assessment of these active fracture/pseudofracture sites at week 24 demonstrated a higher rate of complete healing in the CRYSVITA group compared to placebo as shown in Table 8. During treatment through week 24, a total of 6 new fractures or pseudofractures appeared in 68 patients receiving CRYSVITA, compared to 8 new abnormalities in 66 patients receiving placebo.

Bone Histomorphometry
- In Study 4, after 48 weeks of treatment, healing of osteomalacia was observed in ten patients as demonstrated by decreases in Osteoid volume/Bone volume (OV/BV) from a mean (SD) score of 26% (12.4) at baseline to 11% (6.5), a change of -57%. Osteoid thickness (O.Th) declined in eleven patients from a mean (SD) of 17 (4.1) micrometers to 12 (3.1) micrometers, a change of -33%. Mineralization lag time (MLt) declined in 6 patients from a mean (SD) of 594 (675) days to 156 (77) days, a change of -74%.
How Supplied
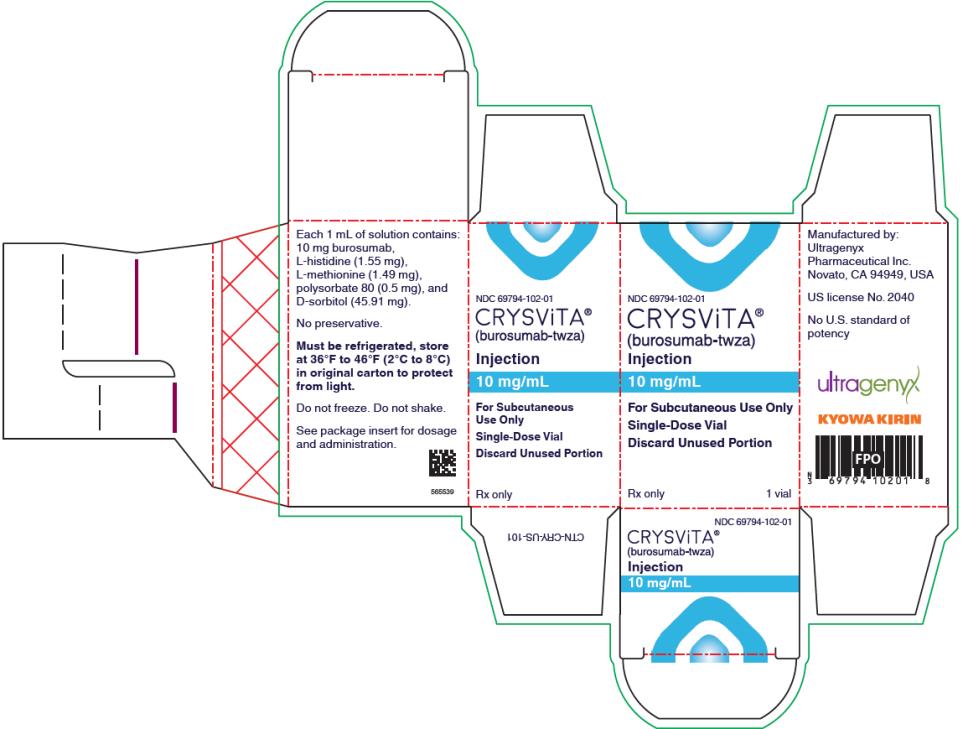
- CRYSVITA (burosumab-twza) injection for subcutaneous administration is supplied as a sterile, preservative-free, clear to slightly opalescent and colorless to pale brown-yellow solution. The product is available as one single-dose vial per carton in the following strengths:
- 10 mg/mL (NDC# 69794-102-01).
- 20 mg/mL (NDC# 69794-203-01).
- 30 mg/mL (NDC# 69794-304-01).
Storage
- CRYSVITA vials must be stored in the original carton until the time of use under refrigerated conditions at 36°F to 46°F (2°C to 8°C). Keep CRYSVITA vial in the original carton to protect from light until time of use.
- Do not freeze or shake CRYSVITA.
- Do not use CRYSVITA beyond the expiration date stamped on the carton.
- CRYSVITA vials are single-dose only. Discard any unused product.
Images
Drug Images
{{#ask: Page Name::Burosumab |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Burosumab |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Hypersensitivity Reactions
- Advise patients that CRYSVITA may cause hypersensitivity events such as rash, injection site rash and urticaria. Instruct the patients to contact their physician if such reactions occur.
Injection Site Reactions
- Inform patients that injection site reactions (e.g. erythema, rash, swelling, bruising, pain, pruritus, urticaria, and hematoma) have occurred at the site of CRYSVITA injection. Instruct the patients to contact their physician if such reactions occur.
Restless Leg Syndrome
- Advise patients that CRYSVITA can induce RLS or worsen the symptoms of existing RLS. Instruct the patients to contact their physician if such a reaction occurs.
Pregnancy
- Report pregnancies to the Ultragenyx Adverse Event reporting line at 1-888-756-8657.
Precautions with Alcohol
Alcohol-Burosumab interaction has not been established. Talk to your doctor regarding the effects of taking alcohol with this medication.
Brand Names
- Crysvita
Look-Alike Drug Names
There is limited information regarding Burosumab Look-Alike Drug Names in the drug label.
Drug Shortage Status
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.