Sandbox: Maria 13: Difference between revisions
(→Causes) |
|||
| Line 13: | Line 13: | ||
==Classification== | ==Classification== | ||
*Nasopharyngeal angiofibroma may be classified according to | *Nasopharyngeal angiofibroma may be classified according to Radkowski Classification System into 3 categories: | ||
:* | *'''Stage I''' | ||
:* | :*Ia: limited to nasal cavity/nasopharynx | ||
:* | :*Ib: extension into one or more paranasal sinuses | ||
* | *'''Stage II''' | ||
:*IIa: minimal extension through sphenopalatine foramen into pterygomaxillary fossa | |||
:*IIb: fills pterygomaxillary fossa bowing the posterior wall of the maxiallary antrum anteriorly or extending into the orbit via the inferior orbital fissure. | |||
:*IIc: extends beyond pterygomaxillary fossa into infratemporal fossa (modified by Radkowski 4) | |||
*'''Stage III''' | |||
:*Stage IIIA: intracranial extension | |||
==Pathophysiology== | ==Pathophysiology== | ||
*The pathogenesis of nasopharyngeal angiofibroma is characterized by the following features: | *The pathogenesis of nasopharyngeal angiofibroma is characterized by the following features: | ||
Revision as of 20:58, 14 April 2016
|
WikiDoc Resources for Sandbox: Maria 13 |
|
Articles |
|---|
|
Most recent articles on Sandbox: Maria 13 Most cited articles on Sandbox: Maria 13 |
|
Media |
|
Powerpoint slides on Sandbox: Maria 13 |
|
Evidence Based Medicine |
|
Cochrane Collaboration on Sandbox: Maria 13 |
|
Clinical Trials |
|
Ongoing Trials on Sandbox: Maria 13 at Clinical Trials.gov Trial results on Sandbox: Maria 13 Clinical Trials on Sandbox: Maria 13 at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Sandbox: Maria 13 NICE Guidance on Sandbox: Maria 13
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Sandbox: Maria 13 Discussion groups on Sandbox: Maria 13 Patient Handouts on Sandbox: Maria 13 Directions to Hospitals Treating Sandbox: Maria 13 Risk calculators and risk factors for Sandbox: Maria 13
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Sandbox: Maria 13 |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Associate Editor(s)-in-Chief: Maria Fernanda Villarreal, M.D. [2]
Synonyms and keywords: Juvenile nasopharyngeal angiofibroma; angiofibroma of the nasopharynx;
Overview
Nasopharyngeal angiofibroma (also called juvenile nasopharyngeal angiofibroma) is a histologically benign but locally aggressive vascular tumor that grows in the back of the nasal cavity. It almost exclusively affects adolescent males. Patients with nasopharyngeal angiofibroma usually present with one-sided nasal obstruction and recurrent bleeding.
Historical Perspective
- Nasopharyngeal angiofibroma was first discovered by [scientist name], a [nationality + occupation], in [year] during/following [event].
Classification
- Nasopharyngeal angiofibroma may be classified according to Radkowski Classification System into 3 categories:
- Stage I
- Ia: limited to nasal cavity/nasopharynx
- Ib: extension into one or more paranasal sinuses
- Stage II
- IIa: minimal extension through sphenopalatine foramen into pterygomaxillary fossa
- IIb: fills pterygomaxillary fossa bowing the posterior wall of the maxiallary antrum anteriorly or extending into the orbit via the inferior orbital fissure.
- IIc: extends beyond pterygomaxillary fossa into infratemporal fossa (modified by Radkowski 4)
- Stage III
- Stage IIIA: intracranial extension
Pathophysiology
- The pathogenesis of nasopharyngeal angiofibroma is characterized by the following features:
- Vascular neoplasm
- Originates from the pterygopalatine fossa
- Genetic alterations associated with the development of nasopharyngeal angiofibroma, include:[1]
- Overexpression PDGF-B
- Overexpression bFGF
- Overexpression bFGF
- Deletion of chromosome 17
- Tumor suppressor gene p53
- Overexpression of Her-2/neu oncogene
- On gross pathology, characteristic findings of nasopharyngeal angiofibroma, include:
- Unencapsulated
- Polypoid fibrous mass
- Bleeding on manipulation
- On microscopic histopathological analysis, characteristic findings of nasopharyngeal angiofibroma, include:
- Fibroblastic cells with plump (near cuboidal) nuclei
- Fibrous stroma
- Abundant capillaries
Causes
- There are no known causes of nasopharyngeal angiofibroma.
Differentiating Nasopharyngeal Angiofibroma from Other Diseases
- Nasopharyngeal angiofibroma must be differentiated from other diseases that cause [clinical feature 1], [clinical feature 2], and [clinical feature 3], such as:
- Antro-choanal polyp (antral-choanal polyp)
- Rhinosporidiosis
- Malignancy
- Chordoma
- Nasopharanageal cyst
- Pyogenic granuloma
Epidemiology and Demographics
- Nasopharyngeal angiofibroma is rare
- Nasopharyngeal angiofibroma accounts for 0.05% of all head and neck tumors.
- The prevalence of nasopharyngeal angiofibroma is approximately 1 per 100,000 individuals worldwide.
Age
- Nasopharyngeal angiofibroma is more commonly observed among patients aged 7-19 years
- Nasopharyngeal angiofibroma is more commonly observed among children and adolescents .
Gender
- Males are more commonly affected with nasopharyngeal angiofibroma than females.
- The male to female ratio for nasopharyngeal angiofibroma is approximately 4 to 1.
Race
- There is no racial predilection for nasopharyngeal angiofibroma.
- Nasopharyngeal angiofibroma usually affects individuals of the [race 1] race.
- [Race 2] individuals are less likely to develop nasopharyngeal angiofibroma.
Risk Factors
- Common risk factors in the development of nasopharyngeal angiofibroma are [risk factor 1], [risk factor 2], [risk factor 3], and [risk factor 4].
Natural History, Complications and Prognosis
- The majority of patients with nasopharyngeal angiofibroma are symptomatic at diagnosis.
- Early clinical features include epistaxis, facial pain, and headache.
- If left untreated, the majority of patients with nasopharyngeal angiofibroma may progress to develop
- The most common complication of nasopharyngeal angiofibroma is
- Prognosis is generally [excellent/good/poor], and the [1/5/10year mortality/survival rate] of patients with nasopharyngeal angiofibroma is approximately [#%].
Diagnosis
Diagnostic Criteria
- The diagnosis of nasopharyngeal angiofibroma is made when at least [number] of the following [number] diagnostic criteria are met:
- [criterion 1]
- [criterion 2]
- [criterion 3]
- [criterion 4]
Symptoms
- Common symptoms of nasopharyngeal angiofibroma, may include:
- Epistaxis or blood-tinged nasal discharge
- Unilateral nasal obstruction
- Rhinorrhea
- Hearing loss
- Diplopia
- Rarely anosmia
- Eye pain
Physical Examination
- Patients with nasopharyngeal angiofibroma usually are well-appearing.
- Physical examination may be remarkable for:
- A smooth submucosal mass in the posterior nasal cavity
Laboratory Findings
- Laboratory findings consistent with the diagnosis of nasopharyngeal angiofibroma, include:
Imaging Findings
- Computed tomography is the imaging modality of choice for nasopharyngeal angiofibroma.
- On conventional radiography, findings of nasopharyngeal angiofibroma, include:
- Visualisation of a nasopharyngeal mass
- Opacification of the sphenoid sinus
- Anterior bowing of the posterior wall of the maxillary antrum
- Holman-miller sign: the anterior bowing of the posterior wall of the maxillary antrum which is seen on lateral skull film or cross-sectional imaging
- Widening of the pterygomaxillary fissure and pterygopalatine fossa
- Erosion of the medial pterygoid plate
- On CT, findings of nasopharyngeal angiofibroma, include:
- Bony changes
- Non-encapsulated soft tissue mass
- Bowing the posterior wall of the maxillary antrum anteriorly
- On MRI, findings of nasopharyngeal angiofibroma, include:
- T1: intermediate signal
- T2: heterogeneous signal: flow voids appear dark
- T1 C+ (Gd): shows prominent enhancement
- The images below demonstrate findings of nasopharyngeal angiofibroma.
-
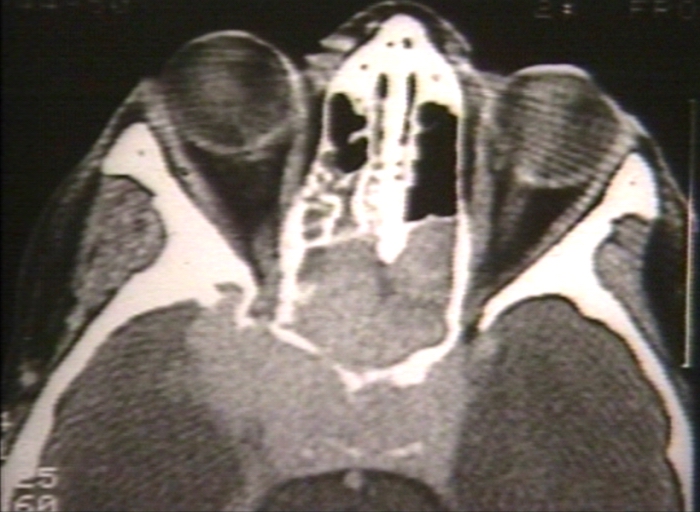
Nasopharyngeal angiofibroma. Skull base invasionImage courtesy of Professor Peter Anderson DVM PhD and published with permission © PEIR, University of Alabama at Birmingham, Department of Pathology
-
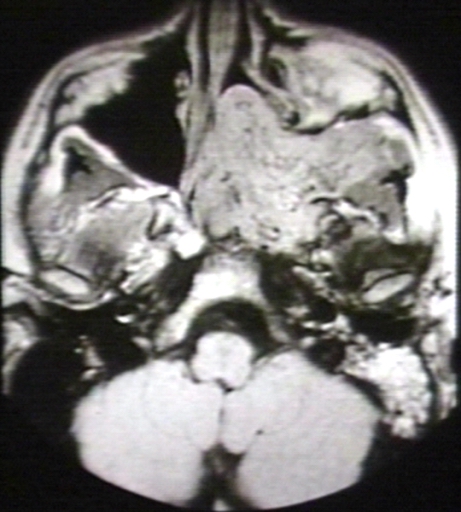
MRI (T1): Nasopharyngeal angiofibroma 1/3 Image courtesy of Professor Peter Anderson DVM PhD and published with permission © PEIR, University of Alabama at Birmingham, Department of Pathology
-
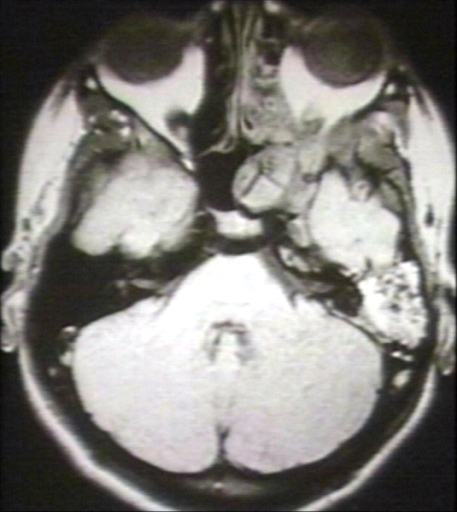
MRI (T1): Nasopharyngeal angiofibroma Image courtesy of Professor Peter Anderson DVM PhD and published with permission © PEIR, University of Alabama at Birmingham, Department of Pathology
-
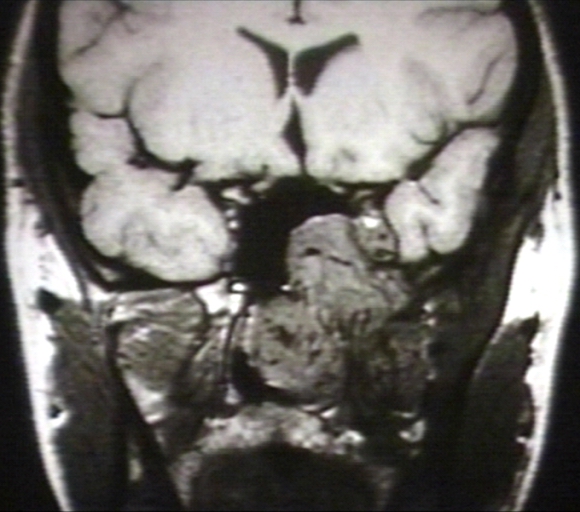
MRI (T1): Nasopharyngeal angiofibroma 3/3 Image courtesy of Professor Peter Anderson DVM PhD and published with permission © PEIR, University of Alabama at Birmingham, Department of Pathology
-
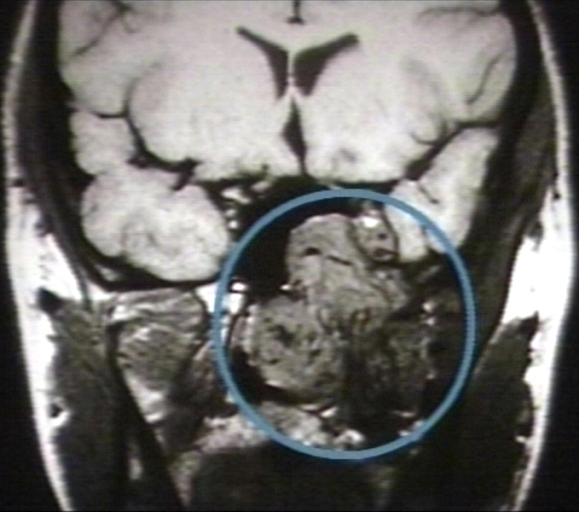
MRI (T1): Nasopharyngeal angiofibroma 3/3 (circled)Image courtesy of Professor Peter Anderson DVM PhD and published with permission © PEIR, University of Alabama at Birmingham, Department of Pathology
Other Diagnostic Studies
- Nasopharyngeal angiofibroma may also be diagnosed using [diagnostic study name].
- Findings on [diagnostic study name] include [finding 1], [finding 2], and [finding 3].
Treatment
Medical Therapy
- There is no treatment for nasopharyngeal angiofibroma; the mainstay of therapy is supportive care.
- The mainstay of therapy for nasopharyngeal angiofibroma is [medical therapy 1] and [medical therapy 2].
- [Medical therapy 1] acts by [mechanism of action1].
- Response to [medical therapy 1] can be monitored with [test/physical finding/imaging] every [frequency/duration].
Surgery
- Surgery is the mainstay of therapy for nasopharyngeal angiofibroma.
- [Surgical procedure] in conjunction with [chemotherapy/radiation] is the most common approach to the treatment of nasopharyngeal angiofibroma.
- [Surgical procedure] can only be performed for patients with [disease stage] nasopharyngeal angiofibroma.
Prevention
- There are no primary preventive measures available for nasopharyngeal angiofibroma.
- Effective measures for the primary prevention of nasopharyngeal angiofibroma include [measure1], [measure2], and [measure3].
- Once diagnosed and successfully treated, patients with nasopharyngeal angiofibroma are followed-up every [duration]. Follow-up testing includes [test 1], [test 2], and [test 3].
References
- ↑ Coutinho-Camillo CM, Brentani MM, Nagai MA (2008). "Genetic alterations in juvenile nasopharyngeal angiofibromas". Head Neck. 30 (3): 390–400. doi:10.1002/hed.20775. PMID 18228521.