Dirithromycin: Difference between revisions
Gerald Chi (talk | contribs) m (Changed protection level for "Dirithromycin" ([Edit=Allow only autoconfirmed users] (expires 19:46, 22 January 2014 (UTC)) [Move=Allow only autoconfirmed users] (expires 19:46, 22 January 2014 (UTC)))) |
No edit summary |
||
| Line 1: | Line 1: | ||
<div style="float: right;"> | |||
[[File:Dirithromycin.png|thumb|none|400px|This image is provided by the National Library of Medicine.]]</div> | |||
| | __NOTOC__ | ||
| | |||
| | |||
| | |||
{{SI}} | {{SI}} | ||
{{CMG}} | |||
==Overview== | |||
'''Dirithromycin''' is a [[macrolide]] [[glycopeptide antibiotic]]. | '''Dirithromycin''' is a [[macrolide]] [[glycopeptide antibiotic]]. | ||
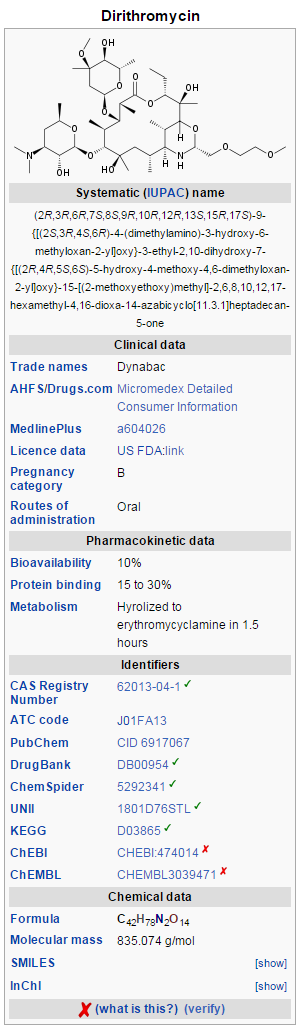
Dirithromycin (Dynabac) is a more lipid-soluble prodrug derivative of 9S-erythromycyclamine prepared by condensation of the latter with 2-(2-methoxyethoxy)acetaldehyde. The 9N, 11O-oxazine ring thus formed is a hemi-aminal that is unstable under both acidic and alkaline aqueous conditions and undergoes spontaneous hydrolysis to form erythromycyclamine. Erythromycyclamine is a semisynthetic derivative of erythromycin in which the 9-ketogroup of the erythronolide ring has been converted to an amino group. Erythromycyclamine retains the antibacterial properties of erythromycin oral administration. The prodrug, dirithromycin, is provided as enteric coated tablets to protect it from acid catalyzed hydrolysis in the stomach. | Dirithromycin (Dynabac) is a more lipid-soluble [[prodrug]] derivative of 9S-erythromycyclamine prepared by condensation of the latter with 2-(2-methoxyethoxy)acetaldehyde. The 9N, 11O-oxazine ring thus formed is a hemi-aminal that is unstable under both acidic and alkaline aqueous conditions and undergoes spontaneous hydrolysis to form erythromycyclamine. Erythromycyclamine is a semisynthetic derivative of [[erythromycin]] in which the 9-ketogroup of the erythronolide ring has been converted to an amino group. Erythromycyclamine retains the antibacterial properties of erythromycin oral administration. The prodrug, dirithromycin, is provided as enteric coated tablets to protect it from acid catalyzed hydrolysis in the stomach. | ||
Orally administered dirithromycin is absorbed rapidly into the plasma, largely from the small intestine. Spontaneous hydrolysis to erythromycyclamine occurs in the plasma. Oral bioavailability is estimated to be about 10%, but food does not affect absorption of the prodrug. | Orally administered dirithromycin is absorbed rapidly into the plasma, largely from the small intestine. Spontaneous hydrolysis to erythromycyclamine occurs in the plasma. Oral bioavailability is estimated to be about 10%, but food does not affect absorption of the prodrug. | ||
==Discontinuation== | |||
Dirithromycin is no longer available in the United States.<ref>{{cite web | title = Dynabac Drug Details | url = http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseaction=Search.SearchAction&SearchTerm=Dirithromycin&SearchType=BasicSearch | publisher = U.S. [[Food and Drug Administration]] | accessdate = 2007-05-25}}</ref> | Dirithromycin is no longer available in the United States.<ref>{{cite web | title = Dynabac Drug Details | url = http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseaction=Search.SearchAction&SearchTerm=Dirithromycin&SearchType=BasicSearch | publisher = U.S. [[Food and Drug Administration]] | accessdate = 2007-05-25}}</ref> Since the production of dirithromycin is discontinued in the U.S, [[National Institutes of Health]] recommend that people taking dirithromycin should consult their physicians to discuss switching to another treatment.<ref>{{cite web | url = http://www.nlm.nih.gov/medlineplus/druginfo/medmaster/a696021.html | title = Dirithromycin | date = January 1, 2006 | work = [[MedlinePlus]] | publisher = U.S. [[National Library of Medicine]] | accessdate = 2007-05-25 |archiveurl = http://web.archive.org/web/20070329044740/http://www.nlm.nih.gov/medlineplus/druginfo/medmaster/a696021.html <!-- Bot retrieved archive --> |archivedate = 2007-03-29}}</ref> However, dirithromycin is still available in many european countries. | ||
==References== | ==References== | ||
{{reflist|2}} | |||
{{GlycopeptideAntiBio}} | {{GlycopeptideAntiBio}} | ||
| Line 44: | Line 23: | ||
[[Category:Macrolide antibiotics]] | [[Category:Macrolide antibiotics]] | ||
[[Category:Prodrugs]] | [[Category:Prodrugs]] | ||
[[Category:Drug]] | |||
[[ | |||
Revision as of 20:20, 7 April 2015
|
WikiDoc Resources for Dirithromycin |
|
Articles |
|---|
|
Most recent articles on Dirithromycin Most cited articles on Dirithromycin |
|
Media |
|
Powerpoint slides on Dirithromycin |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Dirithromycin at Clinical Trials.gov Trial results on Dirithromycin Clinical Trials on Dirithromycin at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Dirithromycin NICE Guidance on Dirithromycin
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Dirithromycin Discussion groups on Dirithromycin Patient Handouts on Dirithromycin Directions to Hospitals Treating Dirithromycin Risk calculators and risk factors for Dirithromycin
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Dirithromycin |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Dirithromycin is a macrolide glycopeptide antibiotic.
Dirithromycin (Dynabac) is a more lipid-soluble prodrug derivative of 9S-erythromycyclamine prepared by condensation of the latter with 2-(2-methoxyethoxy)acetaldehyde. The 9N, 11O-oxazine ring thus formed is a hemi-aminal that is unstable under both acidic and alkaline aqueous conditions and undergoes spontaneous hydrolysis to form erythromycyclamine. Erythromycyclamine is a semisynthetic derivative of erythromycin in which the 9-ketogroup of the erythronolide ring has been converted to an amino group. Erythromycyclamine retains the antibacterial properties of erythromycin oral administration. The prodrug, dirithromycin, is provided as enteric coated tablets to protect it from acid catalyzed hydrolysis in the stomach. Orally administered dirithromycin is absorbed rapidly into the plasma, largely from the small intestine. Spontaneous hydrolysis to erythromycyclamine occurs in the plasma. Oral bioavailability is estimated to be about 10%, but food does not affect absorption of the prodrug.
Discontinuation
Dirithromycin is no longer available in the United States.[1] Since the production of dirithromycin is discontinued in the U.S, National Institutes of Health recommend that people taking dirithromycin should consult their physicians to discuss switching to another treatment.[2] However, dirithromycin is still available in many european countries.
References
- ↑ "Dynabac Drug Details". U.S. Food and Drug Administration. Retrieved 2007-05-25.
- ↑ "Dirithromycin". MedlinePlus. U.S. National Library of Medicine. January 1, 2006. Archived from the original on 2007-03-29. Retrieved 2007-05-25.