Ingenol mebutate: Difference between revisions
No edit summary |
No edit summary |
||
| Line 121: | Line 121: | ||
*Local skin reactions, including erythema, flaking/scaling, crusting, swelling, vesiculation/pustulation, and erosion/ulceration were assessed within the selected treatment area and graded by the investigator on a scale of 0 to 4. A grade of 0 represented no reaction present in the treated area, and a grade of 4 indicated a marked and severe skin reaction that extended beyond the treated area. | *Local skin reactions, including erythema, flaking/scaling, crusting, swelling, vesiculation/pustulation, and erosion/ulceration were assessed within the selected treatment area and graded by the investigator on a scale of 0 to 4. A grade of 0 represented no reaction present in the treated area, and a grade of 4 indicated a marked and severe skin reaction that extended beyond the treated area. | ||
: [[File:{{PAGENAME}}01.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
: [[File:{{PAGENAME}}02.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
*Local skin reactions typically occurred within 1 day of treatment initiation, peaked in intensity up to 1 week following completion of treatment, and resolved within 2 weeks for areas treated on the face and scalp, and within 4 weeks for areas treated on the trunk and extremities. | *Local skin reactions typically occurred within 1 day of treatment initiation, peaked in intensity up to 1 week following completion of treatment, and resolved within 2 weeks for areas treated on the face and scalp, and within 4 weeks for areas treated on the trunk and extremities. | ||
| Line 129: | Line 129: | ||
*Adverse reactions that occurred in ≥2% of subjects treated with Picato® gel and at a higher frequency than the vehicle are presented in Table 3 and Table 4. | *Adverse reactions that occurred in ≥2% of subjects treated with Picato® gel and at a higher frequency than the vehicle are presented in Table 3 and Table 4. | ||
: [[File:{{PAGENAME}}03.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
: [[File:{{PAGENAME}}04.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
*Less common adverse reactions in subjects treated with Picato® included: eyelid edema, eye pain, conjunctivitis. | *Less common adverse reactions in subjects treated with Picato® included: eyelid edema, eye pain, conjunctivitis. | ||
| Line 298: | Line 298: | ||
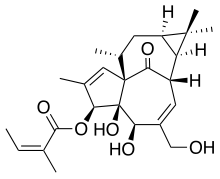
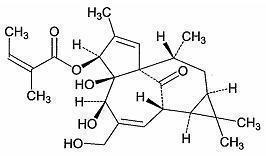
*The molecular formula is C25H34O6 and molecular weight is 430.5. Ingenol mebutate is represented by the following structural formula: | *The molecular formula is C25H34O6 and molecular weight is 430.5. Ingenol mebutate is represented by the following structural formula: | ||
: [[File:{{PAGENAME}} | : [[File:{{PAGENAME}}16.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
*Ingenol mebutate is a white to pale yellow crystalline powder. | *Ingenol mebutate is a white to pale yellow crystalline powder. | ||
| Line 339: | Line 339: | ||
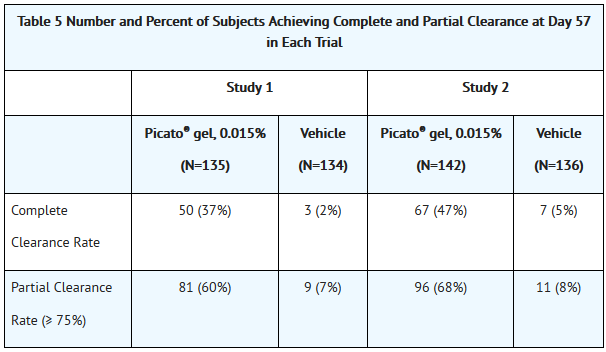
*Efficacy was assessed at Day 57. Complete clearance rate was defined as the proportion of subjects with no (zero) clinically visible AK lesions in the treatment area. Partial clearance rate was defined as the proportion of subjects with 75% or greater reduction in the number of AK lesions at baseline in the selected treatment area. Table 5 presents the efficacy results for each trial. | *Efficacy was assessed at Day 57. Complete clearance rate was defined as the proportion of subjects with no (zero) clinically visible AK lesions in the treatment area. Partial clearance rate was defined as the proportion of subjects with 75% or greater reduction in the number of AK lesions at baseline in the selected treatment area. Table 5 presents the efficacy results for each trial. | ||
: [[File:{{PAGENAME}}05.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
: [[File:{{PAGENAME}}06.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
*Subjects who achieved complete clearance at Day 57 in Study 1 and Study 2 entered a 12-month follow-up period. Based on 108 Picato® gel-treated subjects who achieved complete clearance in Study 1 and Study 2, the recurrence rate at 12 months was 54% where recurrence was defined as the percentage of subjects with any identified AK lesion in the previously treated area who achieved complete clearance at Day 57. | *Subjects who achieved complete clearance at Day 57 in Study 1 and Study 2 entered a 12-month follow-up period. Based on 108 Picato® gel-treated subjects who achieved complete clearance in Study 1 and Study 2, the recurrence rate at 12 months was 54% where recurrence was defined as the percentage of subjects with any identified AK lesion in the previously treated area who achieved complete clearance at Day 57. | ||
| Line 349: | Line 349: | ||
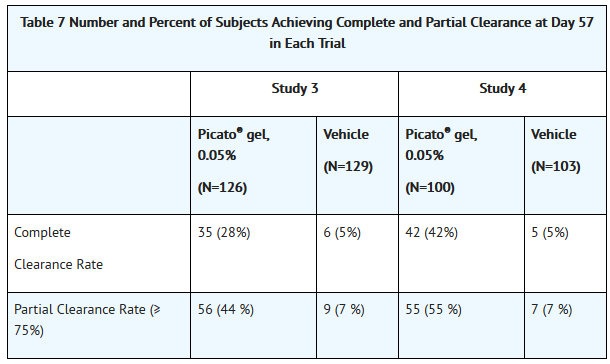
:*Efficacy was assessed at Day 57. Complete clearance rate was defined as the proportion of subjects with no (zero) clinically visible AK lesions in the treatment area. The partial clearance rate was defined as the proportion of subjects with 75% or greater reduction in the number of AK lesions at baseline in the selected treatment area. Table 7 presents the efficacy results for each study. | :*Efficacy was assessed at Day 57. Complete clearance rate was defined as the proportion of subjects with no (zero) clinically visible AK lesions in the treatment area. The partial clearance rate was defined as the proportion of subjects with 75% or greater reduction in the number of AK lesions at baseline in the selected treatment area. Table 7 presents the efficacy results for each study. | ||
: [[File:{{PAGENAME}}07.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
: [[File:{{PAGENAME}}08.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
<!--How Supplied--> | <!--How Supplied--> | ||
| Line 360: | Line 360: | ||
:*Picato® gel is available in 2 dosage strengths: 0.015% and 0.05%. | :*Picato® gel is available in 2 dosage strengths: 0.015% and 0.05%. | ||
: [[File:{{PAGENAME}}09.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
*Store Picato® gel in a refrigerator at 36ºF – 46ºF (2ºC – 8ºC); excursions permitted between 32ºF – 59ºF (0ºC – 15ºC) (see USP for controlled cold temperature). Protect from freezing. | *Store Picato® gel in a refrigerator at 36ºF – 46ºF (2ºC – 8ºC); excursions permitted between 32ºF – 59ºF (0ºC – 15ºC) (see USP for controlled cold temperature). Protect from freezing. | ||
| Line 377: | Line 377: | ||
:*avoid washing and touching the treated area, or participating in activities that cause excessive sweating, for 6 hours after treatment. Following this time, patients may wash the area with a mild soap. | :*avoid washing and touching the treated area, or participating in activities that cause excessive sweating, for 6 hours after treatment. Following this time, patients may wash the area with a mild soap. | ||
:*keep out of the reach of children. | :*keep out of the reach of children. | ||
: [[File:{{PAGENAME}}10.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
: [[File:{{PAGENAME}}11.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | |||
<!--Precautions with Alcohol--> | <!--Precautions with Alcohol--> | ||
| Line 419: | Line 423: | ||
{{LabelImage | {{LabelImage | ||
|fileName={{PAGENAME}} | |fileName={{PAGENAME}}12.png|This image is provided by the National Library of Medicine. | ||
}} | |||
{{LabelImage | |||
|fileName={{PAGENAME}}13.png|This image is provided by the National Library of Medicine. | |||
}} | |||
{{LabelImage | |||
|fileName={{PAGENAME}}14.png|This image is provided by the National Library of Medicine. | |||
}} | }} | ||
{{LabelImage | {{LabelImage | ||
|fileName={{PAGENAME}} | |fileName={{PAGENAME}}15.png|This image is provided by the National Library of Medicine. | ||
}} | }} | ||
Revision as of 15:00, 13 February 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Ingenol mebutate is an inducer of cell death that is FDA approved for the {{{indicationType}}} of actinic keratosis. Common adverse reactions include local skin reactions, application site pain, application site pruritus, application site irritation, application site infection, periorbital edema, nasopharyngitis and headache.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Actinic Keratosis
- For topical use only; Picato® gel is not for oral, ophthalmic, or intravaginal use.
- For the treatment of actinic keratosis on the face and scalp Picato® gel, 0.015% should be applied to the affected area once daily for 3 consecutive days.
- For the treatment of actinic keratosis on the trunk and extremities Picato® gel, 0.05% should be applied to the affected area once daily for 2 consecutive days.
- Picato® gel may be applied to the affected area, up to one contiguous skin area of approximately 25 cm2 (e.g., 5 cm x 5 cm) using one unit dose tube. After spreading evenly over the treatment area, the gel should be allowed to dry for 15 minutes. Patients should wash their hands immediately after applying Picato® gel and take care not to transfer the applied drug to other areas, including the eye. Patients should avoid washing and touching the treated area for a period of 6 hours after application of Picato® gel. Following this time, patients may wash the area with a mild soap.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ingenol mebutate in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ingenol mebutate in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Ingenol mebutate in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ingenol mebutate in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ingenol mebutate in pediatric patients.
Contraindications
- None.
Warnings
Precautions
- Eye Exposure
- Eye disorders, including severe eye pain, eyelid edema, eyelid ptosis, periorbital edema can occur after exposure. Patients should wash hands well after applying Picato® gel, and avoid transfer of the drug to the periocular area during and after application. If accidental exposure occurs, the area should be flushed with water and the patient should seek medical care as soon as possible.
- Local Skin Reactions
- Severe skin reactions in the treated area, including erythema, crusting, swelling, vesiculation/postulation, and erosion/ulceration, can occur after topical application of Picato® gel. Administration of Picato® gel is not recommended until the skin is healed from any previous drug or surgical treatment.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
- The data described below reflect exposure to Picato® gel in 499 subjects with actinic keratosis, including 274 subjects exposed to Picato® gel field treatment (skin area of 25 cm2 in the face or scalp regions) at a concentration of 0.015% once daily for 3 consecutive days, and 225 subjects exposed to Picato® gel field treatment (skin area of 25 cm2 in the trunk or extremities regions) at a concentration of 0.05% once daily for 2 consecutive days.
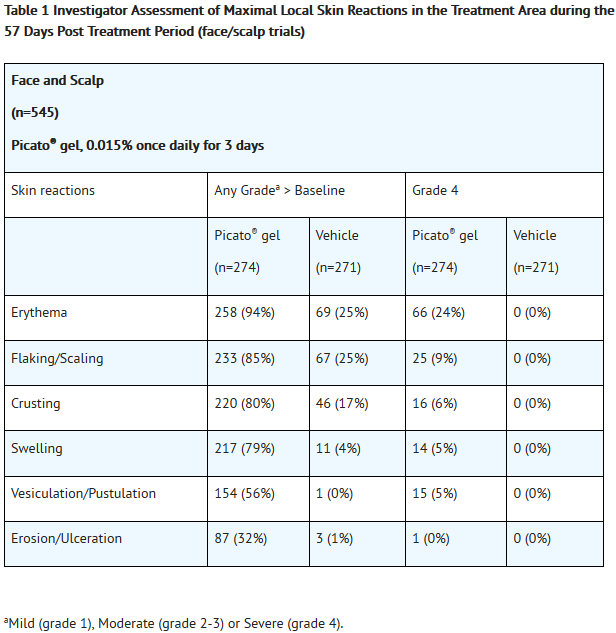
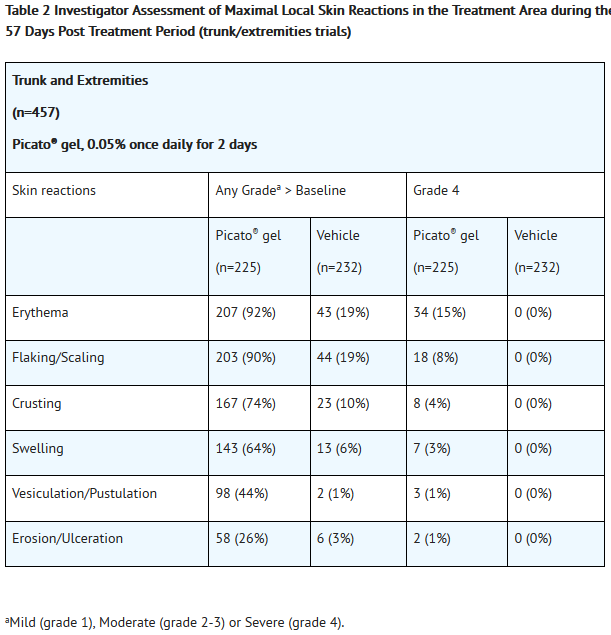
- Local skin reactions, including erythema, flaking/scaling, crusting, swelling, vesiculation/pustulation, and erosion/ulceration were assessed within the selected treatment area and graded by the investigator on a scale of 0 to 4. A grade of 0 represented no reaction present in the treated area, and a grade of 4 indicated a marked and severe skin reaction that extended beyond the treated area.
- Local skin reactions typically occurred within 1 day of treatment initiation, peaked in intensity up to 1 week following completion of treatment, and resolved within 2 weeks for areas treated on the face and scalp, and within 4 weeks for areas treated on the trunk and extremities.
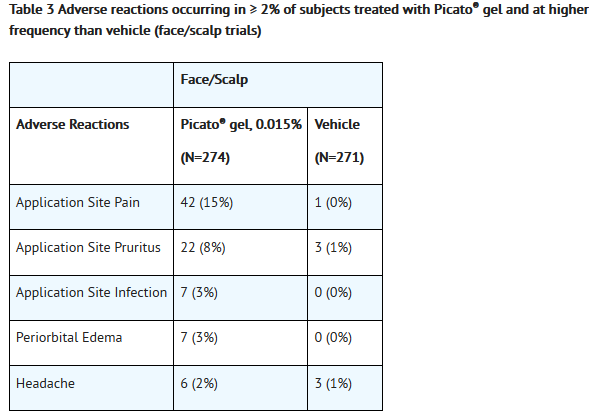
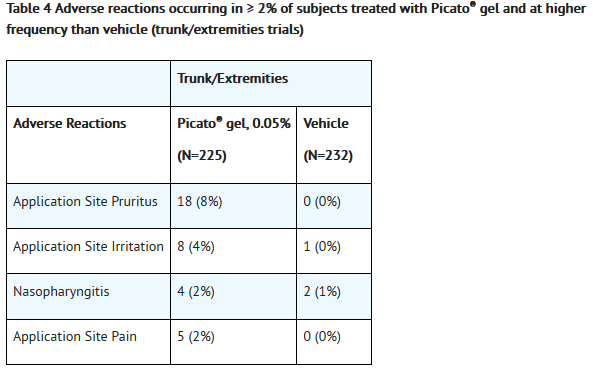
- Adverse reactions that occurred in ≥2% of subjects treated with Picato® gel and at a higher frequency than the vehicle are presented in Table 3 and Table 4.
- Less common adverse reactions in subjects treated with Picato® included: eyelid edema, eye pain, conjunctivitis.
- A total of 108 subjects treated with Picato® gel on the face/scalp and 38 subjects treated on the trunk/extremities were followed for 12 months. Results from these studies did not change the safety profile of Picato® gel.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Ingenol mebutate in the drug label.
Drug Interactions
There is limited information regarding Ingenol mebutate Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Pregnancy Category C
- There are no adequate and well-controlled studies of Picato® gel in pregnant women. Picato® gel should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Systemic embryofetal development studies were conducted with ingenol mebutate in rats and rabbits. Intravenous doses of 1.5, 3, and 5 µg/kg/day (9, 18, and 30 μg/m2/day) ingenol mebutate were administered during the period of organogenesis (gestational days 6 – 16) to pregnant female rats. No treatment related effects on embryofetal toxicity or teratogenicity were noted at doses up to 5 µg/kg/day (30 μg/m2/day). Intravenous doses of 1, 2, and 4 µg/kg/day (12, 24, and 48 μg/m2/day) ingenol mebutate were administered during the period of organogenesis (gestational days 6 – 18) to pregnant female rabbits. An increase in embryo-fetal mortality was noted at 4 µg/kg/day (48 μg/m2/day). An increased incidence of fetal visceral and skeletal variations was noted in all three ingenol mebutate dose groups. The clinical relevance of these findings is unclear since systemic exposure of ingenol mebutate was not detected in subjects with actinic keratosis treated with Picato® gel, 0.05% applied to a 100 cm2 treatment area [see Clinical Pharmacology ( 12.3)].
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Ingenol mebutate in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Ingenol mebutate during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Ingenol mebutate with respect to nursing mothers.
Pediatric Use
- Actinic keratosis is not a condition generally seen within the pediatric population.
- The safety and effectiveness of Picato® gel for actinic keratosis in patients less than 18 years of age have not been established.
Geriatic Use
- Of the 1165 subjects treated with Picato® gel in the clinical trials, 56% were 65 years and older and, 21% were 75 years and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects.
Gender
There is no FDA guidance on the use of Ingenol mebutate with respect to specific gender populations.
Race
There is no FDA guidance on the use of Ingenol mebutate with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Ingenol mebutate in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Ingenol mebutate in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Ingenol mebutate in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Ingenol mebutate in patients who are immunocompromised.
Administration and Monitoring
Administration
- Topical
Monitoring
There is limited information regarding Monitoring of Ingenol mebutate in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Ingenol mebutate in the drug label.
Overdosage
Acute Overdose
- Topical overdosing of Picato® gel could result in an increased incidence of local skin reactions.
Chronic Overdose
There is limited information regarding Chronic Overdose of Ingenol mebutate in the drug label.
Pharmacology
Mechanism of Action
- The mechanism of action by which Picato® gel induces cell death in treating AK lesions is unknown.
Structure
- Picato® (ingenol mebutate) gel, 0.015% or 0.05% is a clear colorless gel for topical administration, which contains the active substance ingenol mebutate, an inducer of cell death.
- The chemical name of ingenol mebutate is:
- 2-Butenoic acid, 2-methyl-, (1aR,2S,5R,5aS,6S,8aS,9R,10aR)-1a,2,5,5a,6,9,10,10a-octahydro-5,5a-dihydroxy-4-(hydroxymethyl)-1,1,7,9-tetramethyl-11-oxo-1H-2,8a-methanocyclopenta [a]cyclopropa[e]cyclodecen-6-yl ester, (2Z) -
or
- (1aR,2S,5R,5aS,6S,8aS,9R,10aR)-5,5a-dihydroxy-4-(hydroxymethyl)-1,1,7,9-tetramethyl-11-oxo-1a,2,5,5a,6,9,10,10a-octahydro-1H 2,8a-methanocyclopenta[a]cyclopropa[e]cyclodecen-6-yl (2Z) 2 methylbut-2-enoate.
- The molecular formula is C25H34O6 and molecular weight is 430.5. Ingenol mebutate is represented by the following structural formula:
- Ingenol mebutate is a white to pale yellow crystalline powder.
- Picato® gel, 0.015% and 0.05% contains 150 mcg and 500 mcg of ingenol mebutate, respectively in each gram of gel consisting of isopropyl alcohol, hydroxyethyl cellulose, citric acid monohydrate, sodium citrate, benzyl alcohol and purified water.
- Picato® gel is clear colorless gel and supplied in unit dose laminate tubes, for single use, containing a nominal fill weight of 0.47 g, with a deliverable weight of 0.25 g. The tubes should be discarded after single use.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Ingenol mebutate in the drug label.
Pharmacokinetics
- Absorption
- The systemic exposure to Picato® gel, 0.05% was assessed in two studies in a total of 16 subjects with AK, following application of approximately 1 g of Picato® gel, 0.05% to an area of 100 cm2 of the dorsal forearm once daily for two consecutive days. In these studies, the blood levels of ingenol mebutate and two of its metabolites (acyl isomers of ingenol mebutate) were measured. Blood levels of ingenol mebutate and the two metabolites were below the lower limit of quantification (0.1 ng/mL) in all the blood samples of the subjects evaluated.
- Drug Interactions
- In vitro studies demonstrated that [3H]-ingenol mebutate undergoes extensive metabolism in human hepatocytes.
- In vitro studies to assess the potential of ingenol mebutate to inhibit or induce human cytochrome P450 (CYP) enzymes demonstrated that ingenol mebutate does not inhibit CYP 1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A4 or induce CYP 1A2, 2C9, and 3A4. The estimated expected systemic exposure (< 0.1 ng/mL) following topical application of Picato® gel, 0.05% to AK subjects in the pharmacokinetic studies described above is negligible compared to the concentrations of ingenol mebutate evaluated in the in vitro studies.
Nonclinical Toxicology
- Long-term animal studies have not been performed to evaluate the carcinogenic potential of Picato® gel or ingenol mebutate. The effects of ingenol mebutate on fertility have not been evaluated.
- Ingenol mebutate was negative in the Ames test, in vitro mouse lymphoma assay, and in vivo rat micronucleus test, but positive in the Syrian hamster embryo (SHE) cell transformation assay.
Clinical Studies
- In two double-blind, vehicle-controlled, clinical trials, 547 adult subjects with AK on the face or scalp were randomized to treatment with either Picato® gel, 0.015% or vehicle gel for 3 consecutive days, followed by an 8 week follow-up period. The studies enrolled subjects with 4 to 8 clinically typical, visible, discrete AK lesions within a 25 cm2 contiguous treatment area. Hypertrophic and hyperkeratotic lesions were excluded from treatment. On each scheduled dosing day, the study gel was applied to the entire treatment area. A total of 536 subjects (98%) completed these studies. Study subjects ranged from 34 to 89 years of age (mean 64 years) and 94% had Fitzpatrick skin type I, II, or III. Approximately 85% of subjects were male, and all Picato®-treated subjects were Caucasian.
- Efficacy was assessed at Day 57. Complete clearance rate was defined as the proportion of subjects with no (zero) clinically visible AK lesions in the treatment area. Partial clearance rate was defined as the proportion of subjects with 75% or greater reduction in the number of AK lesions at baseline in the selected treatment area. Table 5 presents the efficacy results for each trial.
- Subjects who achieved complete clearance at Day 57 in Study 1 and Study 2 entered a 12-month follow-up period. Based on 108 Picato® gel-treated subjects who achieved complete clearance in Study 1 and Study 2, the recurrence rate at 12 months was 54% where recurrence was defined as the percentage of subjects with any identified AK lesion in the previously treated area who achieved complete clearance at Day 57.
- Actinic Keratosis of the Trunk and Extremities
- In two double-blind, vehicle-controlled clinical trials, 458 adult subjects with AK on the trunk or extremities were randomized to treatment with either Picato® gel, 0.05% or vehicle gel for 2 consecutive days, followed by an 8 week follow-up period. The studies enrolled subjects with 4 to 8 clinically typical, visible, discrete AK lesions within a 25 cm2 contiguous treatment area. Hypertrophic and hyperkeratotic lesions were excluded from treatment. On each scheduled dosing day, the study gel was applied to the entire treatment area. A total of 447 subjects (98%) completed these studies. Study subjects ranged from 34 to 89 years of age (mean 66 years) and 94% had Fitzpatrick skin type I, II, or III. Approximately 62% of subjects were male, and all Picato®-treated subjects were Caucasian.
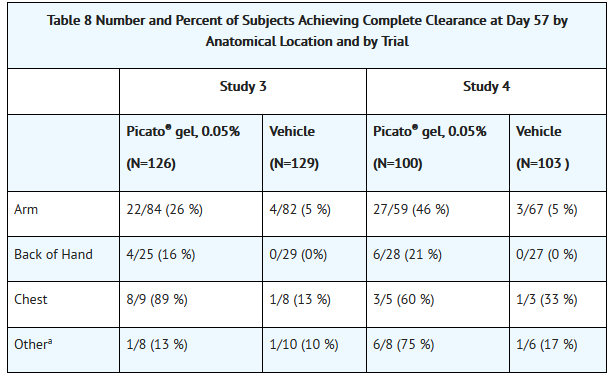
- Efficacy was assessed at Day 57. Complete clearance rate was defined as the proportion of subjects with no (zero) clinically visible AK lesions in the treatment area. The partial clearance rate was defined as the proportion of subjects with 75% or greater reduction in the number of AK lesions at baseline in the selected treatment area. Table 7 presents the efficacy results for each study.
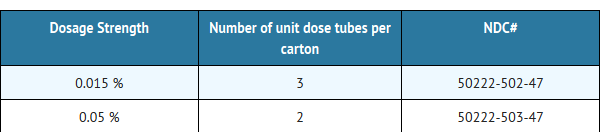
How Supplied
- Picato® gel is a clear colorless gel and is supplied in unit dose laminate tubes containing a nominal fill weight of 0.47 g, with a deliverable weight of 0.25 g. The tubes should be discarded after single use.
- Picato® gel is available in 2 dosage strengths: 0.015% and 0.05%.
- Store Picato® gel in a refrigerator at 36ºF – 46ºF (2ºC – 8ºC); excursions permitted between 32ºF – 59ºF (0ºC – 15ºC) (see USP for controlled cold temperature). Protect from freezing.
Storage
There is limited information regarding Ingenol mebutate Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Ingenol mebutate |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Ingenol mebutate |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Picato® gel should be used as directed by a physician.
- Picato® gel is for external use only. Advise patients to avoid contact with the eyes. Inform patients that treatment with Picato® gel may lead to local skin reactions.
- Patients should avoid inadvertent transfer of Picato® gel to other areas, or to another person. Instruct patients to:
- allow the treated area to dry for 15 minutes after application.
- avoid washing and touching the treated area, or participating in activities that cause excessive sweating, for 6 hours after treatment. Following this time, patients may wash the area with a mild soap.
- keep out of the reach of children.
Precautions with Alcohol
- Alcohol-Ingenol mebutate interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- PICATO®[1]
Look-Alike Drug Names
There is limited information regarding Ingenol mebutate Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Ingenol mebutate |Pill Name=No image.jpg |Drug Name= |Pill Ingred=|+sep=; |Pill Imprint= |Pill Dosage= |Pill Color=|+sep=; |Pill Shape= |Pill Size (mm)= |Pill Scoring= |Pill Image= |Drug Author= |NDC=
}}
{{#subobject:
|Label Page=Ingenol mebutate |Label Name=Ingenol mebutate12.png
}}
{{#subobject:
|Label Page=Ingenol mebutate |Label Name=Ingenol mebutate13.png
}}
{{#subobject:
|Label Page=Ingenol mebutate |Label Name=Ingenol mebutate14.png
}}
{{#subobject:
|Label Page=Ingenol mebutate |Label Name=Ingenol mebutate15.png
}}