Fluorometholone: Difference between revisions
No edit summary |
No edit summary |
||
| (8 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
{{DrugProjectFormSinglePage | {{DrugProjectFormSinglePage | ||
|authorTag= | |authorTag={{DB}} | ||
|genericName=Fluorometholone | |||
|aOrAn=a | |||
|drugClass=adrenal cortex hormone | |||
|indicationType=treatment | |||
|genericName= | |indication=corticosteroid-responsive inflammation of the palpebral and bulbar conjunctiva, cornea and anterior segment of the globe | ||
|adverseReactions=burning sensation in eye, [[cataract]], eye irritation, impaired wound healing, [[raised intraocular pressure]], [[infectious disease]] | |||
|blackBoxWarningTitle=Title | |||
|blackBoxWarningBody=<i><span style="color:#FF0000;">ConditionName: </span></i> | |||
|aOrAn= | |||
a | |||
|drugClass= | |||
| | |||
| | |||
|adverseReactions= | |||
|blackBoxWarningTitle= | |||
Title | |||
|blackBoxWarningBody= | |||
<i><span style="color:#FF0000;">ConditionName: </span></i> | |||
* Content | * Content | ||
| Line 42: | Line 15: | ||
<!--FDA-Labeled Indications and Dosage (Adult)--> | <!--FDA-Labeled Indications and Dosage (Adult)--> | ||
|fdaLIADAdult======[[Inflammatory disorder of the eye]]===== | |||
*FML® suspension is indicated for the treatment of corticosteroid-responsive inflammation of the palpebral and bulbar conjunctiva, cornea and anterior segment of the globe. | |||
* Dosing Information | * Dosing Information | ||
:* | :*Shake well before using. Instill one drop into the conjunctival sac two to four times daily. During the initial 24 to 48 hours, the dosing frequency may be increased to one application every four hours. Care should be taken not to discontinue therapy prematurely. | ||
|offLabelAdultGuideSupport=*There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Fluorometholone in adult patients. | |||
===== | |offLabelAdultNoGuideSupport=*There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of Fluorometholone in adult patients. | ||
|fdaLIADPed======Inflammatory disorder of the eye===== | |||
* Dosing Information | * Dosing Information | ||
:* | :*Shake well before using. Instill one drop into the conjunctival sac two to four times daily. During the initial 24 to 48 hours, the dosing frequency may be increased to one application every four hours. Care should be taken not to discontinue therapy prematurely. | ||
=====Condition2===== | =====Condition2===== | ||
*There is limited information regarding <i>FDA-Labeled Use</i> of {{PAGENAME}} in pediatric patients. | |||
* | |||
There is limited information regarding <i>FDA-Labeled Use</i> of {{PAGENAME}} in pediatric patients. | |||
<!--Off-Label Use and Dosage (Pediatric)--> | <!--Off-Label Use and Dosage (Pediatric)--> | ||
<!--Guideline-Supported Use (Pediatric)--> | <!--Guideline-Supported Use (Pediatric)--> | ||
|offLabelPedGuideSupport=*There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Fluorometholone in pediatric patients. | |||
|offLabelPedNoGuideSupport=*There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of Fluorometholone in pediatric patients. | |||
|contraindications=*FML® suspension is contraindicated in most viral diseases of the cornea and conjunctiva, including epithelial herpes simplex [[keratitis]] (dendritic keratitis), [[vaccinia]], and varicella, and also in mycobacterial infection of the eye, and fungal diseases of ocular structures. | |||
| | *FML® suspension is also contraindicated in individuals with known or suspected [[hypersensitivity]] to any of the ingredients of this preparation and to other corticosteroids. | ||
|warnings=*Prolonged use of corticosteroids may increase intraocular pressure in susceptible individuals, resulting in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision, and in posterior subcapsular [[cataract]] formation. Prolonged use may also suppress the host immune response and thus increase the hazard of secondary ocular infections. | |||
*Various ocular diseases and long-term use of topical corticosteroids have been known to cause corneal and scleral thinning. Use of topical corticosteroids in the presence of thin corneal or scleral tissue may lead to perforation. | |||
* | *Acute purulent infections of the eye may be masked or activity enhanced by the presence of corticosteroid medication. | ||
* | *If this product is used for 10 days or longer, intraocular pressure should be routinely monitored even though it may be difficult in children and uncooperative patients. Steroids should be used with caution in the presence of [[glaucoma]]. Intraocular pressure should be checked frequently. | ||
* | *The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation. | ||
* | *Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including [[herpes simplex]]). Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution; frequent slit lamp microscopy is recommended. | ||
====Precautions==== | ====Precautions==== | ||
'''General''' | |||
*The initial prescription and renewal of the medication order beyond 20 milliliters of FML® suspension should be made by a physician only after examination of the patient with the aid of magnification, such as slit lamp biomicroscopy and, where appropriate, fluorescein staining. If signs and symptoms fail to improve after two days, the patient should be re-evaluated. | |||
*As fungal infections of the cornea are particularly prone to develop coincidentally with long-term local corticosteroid applications, fungal invasion should be suspected in any persistent corneal ulceration where a corticosteroid has been used or is in use. Fungal cultures should be taken when appropriate. | |||
*If this product is used for 10 days or longer, intraocular pressure should be monitored (see WARNINGS). | |||
'''Information for Patients''' | |||
*If inflammation or pain persists longer than 48 hours or becomes aggravated, the patient should be advised to discontinue use of the medication and consult a physician. | |||
*This product is sterile when packaged. To prevent contamination, care should be taken to avoid touching the bottle tip to eyelids or to any other surface. The use of this bottle by more than one person may spread infection. Keep bottle tightly closed when not in use. Keep out of the reach of children. | |||
= | *The preservative in FML® suspension, benzalkonium chloride, may be absorbed by soft contact lenses. Patients wearing soft contact lenses should be instructed to wait at least 15 minutes after instilling FML® suspension to insert soft contact lenses. | ||
|clinicalTrials=*Adverse reactions include, in decreasing order of frequency, elevation of [[intraocular pressure]]] (IOP) with possible development of glaucoma and infrequent optic nerve damage, posterior subcapsular cataract formation, and delayed wound healing. | |||
*Although systemic effects are extremely uncommon, there have been rare occurrences of systemic [[hypercorticoidism]] after use of topical dermatologic steroids applied to the skin. | |||
*Corticosteroid-containing preparations have also been reported to cause acute anterior uveitis and perforation of the globe. [[Keratitis]], [[conjunctivitis]], [[corneal ulcers]], [[mydriasis]], [[conjunctival hyperemia]], loss of accommodation and [[ptosis]] have occasionally been reported following local use of corticosteroids. | |||
*The development of secondary ocular infection (bacterial, fungal and viral) has occurred. Fungal and viral infections of the cornea are particularly prone to develop coincidentally with long-term applications of steroids. The possibility of fungal invasion should be considered in any persistent corneal ulceration where steroid treatment has been used (see WARNINGS). | |||
*Transient burning and stinging upon instillation and other minor symptoms of ocular irritation have been reported with the use of FML® suspension. Other adverse events reported with the use of fluorometholone include: allergic reactions; foreign body sensation; erythema of eyelid; eyelid edema/eye swelling; eye discharge; eye pain; eye [[pruritus]]; lacrimation increased; [[rash]]; taste perversion; visual disturbance (blurry vision); and visual field defect. | |||
|postmarketing=*There is limited information regarding <i>Postmarketing Experience</i> of {{PAGENAME}} in the drug label. | |||
<!--Drug Interactions--> | <!--Drug Interactions--> | ||
|FDAPregCat=C | |||
|useInPregnancyFDA='''Teratogenic effects. Pregnancy Category C''' | |||
*Fluorometholone has been shown to be embryocidal and teratogenic in rabbits when administered at low multiples of the human ocular dose. Fluorometholone was applied ocularly to rabbits daily on days 6-18 of gestation, and dose-related fetal loss and fetal abnormalities including cleft palate, deformed rib cage, anomalous limbs and neural abnormalities such as [[encephalocele]], [[craniorachischisis]], and [[spina bifida]] were observed. There are no adequate and well-controlled studies of fluorometholone in pregnant women, and it is not known whether fluorometholone can cause fetal harm when administered to a pregnant woman. Fluorometholone should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. | |||
|useInPregnancyAUS=* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | |||
* | |||
|useInPregnancyAUS= | |||
* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | |||
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of {{PAGENAME}} in women who are pregnant. | There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of {{PAGENAME}} in women who are pregnant. | ||
|useInLaborDelivery=*There is no FDA guidance on use of {{PAGENAME}} during labor and delivery. | |||
|useInLaborDelivery= | |useInNursing=*It is not known whether topical ophthalmic administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Systemically‑administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. Because of the potential for serious adverse reactions in nursing infants from fluorometholone, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. | ||
There is no FDA guidance on use of {{PAGENAME}} during labor and delivery. | |useInPed=*Safety and effectiveness in infants below the age of two years have not been established. | ||
|useInGeri=*No overall differences in safety or effectiveness have been observed between elderly and younger patients. | |||
|useInNursing= | |useInGender=*There is no FDA guidance on the use of {{PAGENAME}} with respect to specific gender populations. | ||
|useInRace=*There is no FDA guidance on the use of {{PAGENAME}} with respect to specific racial populations. | |||
|useInRenalImpair=*There is no FDA guidance on the use of {{PAGENAME}} in patients with renal impairment. | |||
|useInPed= | |useInHepaticImpair=*There is no FDA guidance on the use of {{PAGENAME}} in patients with hepatic impairment. | ||
|useInReproPotential=*There is no FDA guidance on the use of {{PAGENAME}} in women of reproductive potentials and males. | |||
|useInImmunocomp=*There is no FDA guidance one the use of {{PAGENAME}} in patients who are immunocompromised. | |||
|useInGeri= | |||
|useInGender= | |||
There is no FDA guidance on the use of {{PAGENAME}} with respect to specific gender populations. | |||
|useInRace= | |||
There is no FDA guidance on the use of {{PAGENAME}} with respect to specific racial populations. | |||
|useInRenalImpair= | |||
There is no FDA guidance on the use of {{PAGENAME}} in patients with renal impairment. | |||
|useInHepaticImpair= | |||
There is no FDA guidance on the use of {{PAGENAME}} in patients with hepatic impairment. | |||
|useInReproPotential= | |||
There is no FDA guidance on the use of {{PAGENAME}} in women of reproductive potentials and males. | |||
|useInImmunocomp= | |||
There is no FDA guidance one the use of {{PAGENAME}} in patients who are immunocompromised. | |||
<!--Administration and Monitoring--> | <!--Administration and Monitoring--> | ||
|administration=*Shake well before using. Instill one drop into the conjunctival sac two to four times daily. During the initial 24 to 48 hours, the dosing frequency may be increased to one application every four hours. Care should be taken not to discontinue therapy prematurely. | |||
*If signs and symptoms fail to improve after two days, the patient should be re-evaluated (see PRECAUTIONS). | |||
* | *The dosing of FML® suspension may be reduced, but care should be taken not to discontinue therapy prematurely. In chronic conditions, withdrawal of treatment should be carried out by gradually decreasing the frequency of applications. | ||
|monitoring=*If this product is used for 10 days or longer, intraocular pressure should be routinely monitored even though it may be difficult in children and uncooperative patients. Steroids should be used with caution in the presence of glaucoma. Intraocular pressure should be checked frequently. | |||
|IVCompat=*There is limited information regarding <i>IV Compatibility</i> of {{PAGENAME}} in the drug label. | |||
|drugBox=[[File:Fluorometholone image.png|600px|thumbnail|left]] | |||
{{clear}} | |||
|mechAction=*Corticosteroids inhibit the inflammatory response to a variety of inciting agents and probably delay or slow healing. They inhibit the edema, fibrin deposition, capillary dilation, leukocyte migration, capillary proliferation, fibroblast proliferation, deposition of collagen, and scar formation associated with inflammation. | |||
* | *There is no generally accepted explanation for the mechanism of action of ocular corticosteroids. However, corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor, arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2. | ||
| | *Corticosteroids are capable of producing a rise in intraocular pressure. In clinical studies of documented steroid-responders, fluorometholone demonstrated a significantly longer average time to produce a rise in intraocular pressure than dexamethasone phosphate; however, in a small percentage of individuals, a significant rise in intraocular pressure occurred within one week. The ultimate magnitude of the rise was equivalent for both drugs. | ||
|structure=*FML® (fluorometholone ophthalmic suspension, USP) 0.1% is a sterile, topical anti-inflammatory agent for ophthalmic use. | |||
'''Chemical Name''' | |||
(Fluorometholone: 9-Fluoro-11β,17-dihydroxy-6α-methylpregna-1,4-diene-3,20-dione. | |||
[[File:Fluorometholone structure.png|600px|thumbnail|left]] | |||
{{clear}} | |||
'''Contains''' | |||
There is limited information regarding <i> | *Active: fluorometholone 0.1%. Preservative: benzalkonium chloride 0.004%. Inactives: edetate disodium; polysorbate 80; polyvinyl alcohol 1.4%; purified water; sodium chloride; sodium phosphate, dibasic; sodium phosphate, monobasic; and sodium hydroxide to adjust pH. FML® suspension is formulated with a pH from 6.2 to 7.5. It has an osmolality range of 290-350 mOsm/kg. | ||
|PD=*There is limited information regarding <i>Pharmacodynamics</i> of {{PAGENAME}} in the drug label. | |||
|PK=*There is limited information regarding <i>Pharmacokinetics</i> of {{PAGENAME}} in the drug label. | |||
|nonClinToxic='''Carcinogenesis, Mutagenesis, Impairment of Fertility''' | |||
< | *No studies have been conducted in animals or in humans to evaluate the possibility of these effects with fluorometholone. | ||
|clinicalStudies=*There is limited information regarding <i>Clinical Studies</i> of {{PAGENAME}} in the drug label. | |||
|howSupplied=*FML® (fluorometholone ophthalmic suspension, USP) 0.1% is supplied sterile in opaque white LDPE plastic bottles with white high impact polystyrene (HIPS) caps as follows: | |||
:*5 mL in 10 mL bottle NDC 11980-211-05 | |||
:*10 mL in 15 mL bottle NDC 11980-211-10 | |||
|storage=*Storage: Store at 2°-25°C (36°-77°F); protect from freezing. Store in an upright position. | |||
|packLabel=[[File:Fluorometholone pdp.png|600px|thumbnail|left]] | |||
{{clear}} | |||
* | |||
| | |||
* | |||
| | |||
[[File:Fluorometholone label.png|600px|thumbnail|left]] | |||
{{clear}} | |||
|fdaPatientInfo=*There is limited information regarding <i>Patient Counseling Information</i> of {{PAGENAME}} in the drug label. | |||
|alcohol=* Alcohol-{{PAGENAME}} interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | |||
|brandNames=FML Forte Liquifilm, | |||
FML Liquifilm, | |||
FML S.O.P., | |||
Fluor-OP. | |||
|lookAlike=* A® — B®<ref name="www.ismp.org">{{Cite web | last = | first = | title = http://www.ismp.org | url = http://www.ismp.org | publisher = | date = }}</ref> | |||
|drugShortage= | |drugShortage= | ||
}} | }} | ||
{{PillImage | |||
|fileName=No image.jpg | |||
}} | |||
{{LabelImage | |||
|fileName={{PAGENAME}}11.png | |||
}} | |||
{{LabelImage | |||
|fileName={{PAGENAME}}11.png | |||
}} | |||
<!--Pill Image--> | |||
<!--Label Display Image--> | <!--Label Display Image--> | ||
<!--Category--> | <!--Category--> | ||
[[Category:Drug]] | [[Category:Drug]] | ||
Latest revision as of 18:27, 6 October 2014
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Deepika Beereddy, MBBS [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Fluorometholone is a adrenal cortex hormone that is FDA approved for the treatment of corticosteroid-responsive inflammation of the palpebral and bulbar conjunctiva, cornea and anterior segment of the globe. Common adverse reactions include burning sensation in eye, cataract, eye irritation, impaired wound healing, raised intraocular pressure, infectious disease.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Inflammatory disorder of the eye
- FML® suspension is indicated for the treatment of corticosteroid-responsive inflammation of the palpebral and bulbar conjunctiva, cornea and anterior segment of the globe.
- Dosing Information
- Shake well before using. Instill one drop into the conjunctival sac two to four times daily. During the initial 24 to 48 hours, the dosing frequency may be increased to one application every four hours. Care should be taken not to discontinue therapy prematurely.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Fluorometholone in adult patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Fluorometholone in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Inflammatory disorder of the eye
- Dosing Information
- Shake well before using. Instill one drop into the conjunctival sac two to four times daily. During the initial 24 to 48 hours, the dosing frequency may be increased to one application every four hours. Care should be taken not to discontinue therapy prematurely.
Condition2
- There is limited information regarding FDA-Labeled Use of Fluorometholone in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Fluorometholone in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Fluorometholone in pediatric patients.
Contraindications
- FML® suspension is contraindicated in most viral diseases of the cornea and conjunctiva, including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, and also in mycobacterial infection of the eye, and fungal diseases of ocular structures.
- FML® suspension is also contraindicated in individuals with known or suspected hypersensitivity to any of the ingredients of this preparation and to other corticosteroids.
Warnings
- Prolonged use of corticosteroids may increase intraocular pressure in susceptible individuals, resulting in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision, and in posterior subcapsular cataract formation. Prolonged use may also suppress the host immune response and thus increase the hazard of secondary ocular infections.
- Various ocular diseases and long-term use of topical corticosteroids have been known to cause corneal and scleral thinning. Use of topical corticosteroids in the presence of thin corneal or scleral tissue may lead to perforation.
- Acute purulent infections of the eye may be masked or activity enhanced by the presence of corticosteroid medication.
- If this product is used for 10 days or longer, intraocular pressure should be routinely monitored even though it may be difficult in children and uncooperative patients. Steroids should be used with caution in the presence of glaucoma. Intraocular pressure should be checked frequently.
- The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation.
- Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex). Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution; frequent slit lamp microscopy is recommended.
Precautions
General
- The initial prescription and renewal of the medication order beyond 20 milliliters of FML® suspension should be made by a physician only after examination of the patient with the aid of magnification, such as slit lamp biomicroscopy and, where appropriate, fluorescein staining. If signs and symptoms fail to improve after two days, the patient should be re-evaluated.
- As fungal infections of the cornea are particularly prone to develop coincidentally with long-term local corticosteroid applications, fungal invasion should be suspected in any persistent corneal ulceration where a corticosteroid has been used or is in use. Fungal cultures should be taken when appropriate.
- If this product is used for 10 days or longer, intraocular pressure should be monitored (see WARNINGS).
Information for Patients
- If inflammation or pain persists longer than 48 hours or becomes aggravated, the patient should be advised to discontinue use of the medication and consult a physician.
- This product is sterile when packaged. To prevent contamination, care should be taken to avoid touching the bottle tip to eyelids or to any other surface. The use of this bottle by more than one person may spread infection. Keep bottle tightly closed when not in use. Keep out of the reach of children.
- The preservative in FML® suspension, benzalkonium chloride, may be absorbed by soft contact lenses. Patients wearing soft contact lenses should be instructed to wait at least 15 minutes after instilling FML® suspension to insert soft contact lenses.
Adverse Reactions
Clinical Trials Experience
- Adverse reactions include, in decreasing order of frequency, elevation of intraocular pressure] (IOP) with possible development of glaucoma and infrequent optic nerve damage, posterior subcapsular cataract formation, and delayed wound healing.
- Although systemic effects are extremely uncommon, there have been rare occurrences of systemic hypercorticoidism after use of topical dermatologic steroids applied to the skin.
- Corticosteroid-containing preparations have also been reported to cause acute anterior uveitis and perforation of the globe. Keratitis, conjunctivitis, corneal ulcers, mydriasis, conjunctival hyperemia, loss of accommodation and ptosis have occasionally been reported following local use of corticosteroids.
- The development of secondary ocular infection (bacterial, fungal and viral) has occurred. Fungal and viral infections of the cornea are particularly prone to develop coincidentally with long-term applications of steroids. The possibility of fungal invasion should be considered in any persistent corneal ulceration where steroid treatment has been used (see WARNINGS).
- Transient burning and stinging upon instillation and other minor symptoms of ocular irritation have been reported with the use of FML® suspension. Other adverse events reported with the use of fluorometholone include: allergic reactions; foreign body sensation; erythema of eyelid; eyelid edema/eye swelling; eye discharge; eye pain; eye pruritus; lacrimation increased; rash; taste perversion; visual disturbance (blurry vision); and visual field defect.
Postmarketing Experience
- There is limited information regarding Postmarketing Experience of Fluorometholone in the drug label.
Drug Interactions
There is limited information regarding Fluorometholone Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C Teratogenic effects. Pregnancy Category C
- Fluorometholone has been shown to be embryocidal and teratogenic in rabbits when administered at low multiples of the human ocular dose. Fluorometholone was applied ocularly to rabbits daily on days 6-18 of gestation, and dose-related fetal loss and fetal abnormalities including cleft palate, deformed rib cage, anomalous limbs and neural abnormalities such as encephalocele, craniorachischisis, and spina bifida were observed. There are no adequate and well-controlled studies of fluorometholone in pregnant women, and it is not known whether fluorometholone can cause fetal harm when administered to a pregnant woman. Fluorometholone should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Fluorometholone in women who are pregnant.
Labor and Delivery
- There is no FDA guidance on use of Fluorometholone during labor and delivery.
Nursing Mothers
- It is not known whether topical ophthalmic administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Systemically‑administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. Because of the potential for serious adverse reactions in nursing infants from fluorometholone, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- Safety and effectiveness in infants below the age of two years have not been established.
Geriatic Use
- No overall differences in safety or effectiveness have been observed between elderly and younger patients.
Gender
- There is no FDA guidance on the use of Fluorometholone with respect to specific gender populations.
Race
- There is no FDA guidance on the use of Fluorometholone with respect to specific racial populations.
Renal Impairment
- There is no FDA guidance on the use of Fluorometholone in patients with renal impairment.
Hepatic Impairment
- There is no FDA guidance on the use of Fluorometholone in patients with hepatic impairment.
Females of Reproductive Potential and Males
- There is no FDA guidance on the use of Fluorometholone in women of reproductive potentials and males.
Immunocompromised Patients
- There is no FDA guidance one the use of Fluorometholone in patients who are immunocompromised.
Administration and Monitoring
Administration
- Shake well before using. Instill one drop into the conjunctival sac two to four times daily. During the initial 24 to 48 hours, the dosing frequency may be increased to one application every four hours. Care should be taken not to discontinue therapy prematurely.
- If signs and symptoms fail to improve after two days, the patient should be re-evaluated (see PRECAUTIONS).
- The dosing of FML® suspension may be reduced, but care should be taken not to discontinue therapy prematurely. In chronic conditions, withdrawal of treatment should be carried out by gradually decreasing the frequency of applications.
Monitoring
- If this product is used for 10 days or longer, intraocular pressure should be routinely monitored even though it may be difficult in children and uncooperative patients. Steroids should be used with caution in the presence of glaucoma. Intraocular pressure should be checked frequently.
IV Compatibility
- There is limited information regarding IV Compatibility of Fluorometholone in the drug label.
Overdosage
There is limited information regarding Fluorometholone overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology
Mechanism of Action
- Corticosteroids inhibit the inflammatory response to a variety of inciting agents and probably delay or slow healing. They inhibit the edema, fibrin deposition, capillary dilation, leukocyte migration, capillary proliferation, fibroblast proliferation, deposition of collagen, and scar formation associated with inflammation.
- There is no generally accepted explanation for the mechanism of action of ocular corticosteroids. However, corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor, arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
- Corticosteroids are capable of producing a rise in intraocular pressure. In clinical studies of documented steroid-responders, fluorometholone demonstrated a significantly longer average time to produce a rise in intraocular pressure than dexamethasone phosphate; however, in a small percentage of individuals, a significant rise in intraocular pressure occurred within one week. The ultimate magnitude of the rise was equivalent for both drugs.
Structure
- FML® (fluorometholone ophthalmic suspension, USP) 0.1% is a sterile, topical anti-inflammatory agent for ophthalmic use.
Chemical Name
(Fluorometholone: 9-Fluoro-11β,17-dihydroxy-6α-methylpregna-1,4-diene-3,20-dione.
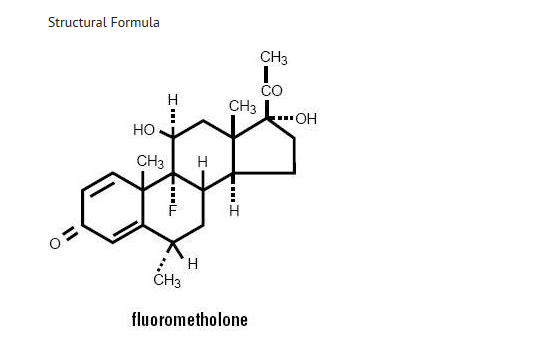
Contains
- Active: fluorometholone 0.1%. Preservative: benzalkonium chloride 0.004%. Inactives: edetate disodium; polysorbate 80; polyvinyl alcohol 1.4%; purified water; sodium chloride; sodium phosphate, dibasic; sodium phosphate, monobasic; and sodium hydroxide to adjust pH. FML® suspension is formulated with a pH from 6.2 to 7.5. It has an osmolality range of 290-350 mOsm/kg.
Pharmacodynamics
- There is limited information regarding Pharmacodynamics of Fluorometholone in the drug label.
Pharmacokinetics
- There is limited information regarding Pharmacokinetics of Fluorometholone in the drug label.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- No studies have been conducted in animals or in humans to evaluate the possibility of these effects with fluorometholone.
Clinical Studies
- There is limited information regarding Clinical Studies of Fluorometholone in the drug label.
How Supplied
- FML® (fluorometholone ophthalmic suspension, USP) 0.1% is supplied sterile in opaque white LDPE plastic bottles with white high impact polystyrene (HIPS) caps as follows:
- 5 mL in 10 mL bottle NDC 11980-211-05
- 10 mL in 15 mL bottle NDC 11980-211-10
Storage
- Storage: Store at 2°-25°C (36°-77°F); protect from freezing. Store in an upright position.
Images
Drug Images
{{#ask: Page Name::Fluorometholone |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel

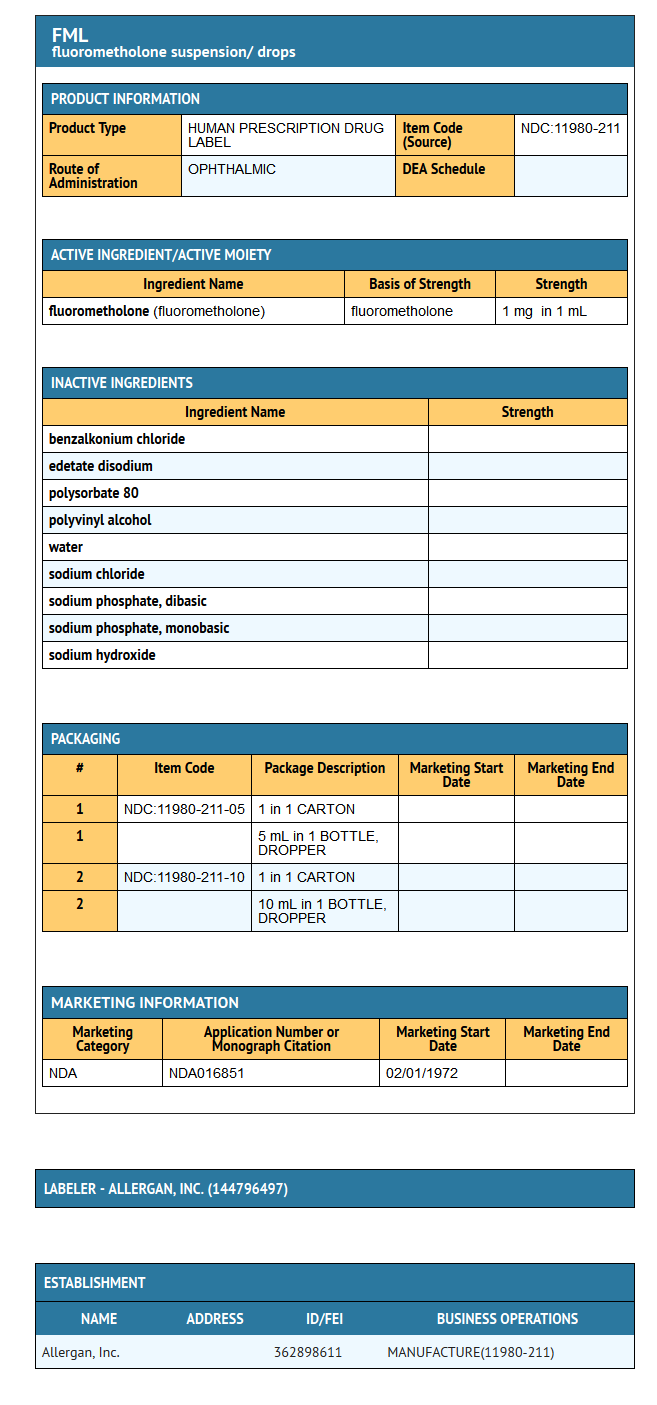
{{#ask: Label Page::Fluorometholone |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- There is limited information regarding Patient Counseling Information of Fluorometholone in the drug label.
Precautions with Alcohol
- Alcohol-Fluorometholone interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
FML Forte Liquifilm, FML Liquifilm, FML S.O.P., Fluor-OP.
Look-Alike Drug Names
- A® — B®[1]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Fluorometholone
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Fluorometholone |Label Name=Fluorometholone11.png
}}
{{#subobject:
|Label Page=Fluorometholone |Label Name=Fluorometholone11.png
}}