Treprostinil (inhalant)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1];
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Treprostinil (inhalant) is {{{aOrAn}}} {{{drugClass}}} that is FDA approved for the treatment of {{{indication}}}. Common adverse reactions include {{{adverseReactions}}}.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
Tyvaso is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to improve exercise ability. Studies establishing effectiveness included predominately patients with NYHA Functional Class III symptoms and etiologies of idiopathic or heritable PAH (56%) or PAH associated with connective tissue diseases (33%).
The effects diminish over the minimum recommended dosing interval of 4 hours; treatment timing can be adjusted for planned activities.
While there are long-term data on use of treprostinil by other routes of administration, nearly all controlled clinical experience with inhaled treprostinil has been on a background of bosentan (an endothelin receptor antagonist) or sildenafil (a phosphodiesterase type 5 inhibitor). The controlled clinical experience was limited to 12 weeks in duration [see CLINICAL STUDIES (14)].
Dosage
2.1 Usual Dosage in Adults Tyvaso is intended for oral inhalation using the Tyvaso Inhalation System, which consists of an ultrasonic, pulsed delivery device and its accessories.
Tyvaso is dosed in 4 separate, equally spaced treatment sessions per day, during waking hours. The treatment sessions should be approximately 4 hours apart.
Initial Dosage:
Therapy should begin with 3 breaths of Tyvaso (18 mcg of treprostinil), per treatment session, 4 times daily. If 3 breaths are not tolerated, reduce to 1 or 2 breaths and subsequently increase to 3 breaths, as tolerated.
Maintenance Dosage:
Dosage should be increased by an additional 3 breaths at approximately 1-2 week intervals, if tolerated, until the target dose of 9 breaths (54 mcg of treprostinil) is reached per treatment session, 4 times daily. If adverse effects preclude titration to target dose, Tyvaso should be continued at the highest tolerated dose.
If a scheduled treatment session is missed or interrupted, therapy should be resumed as soon as possible at the usual dose.
The maximum recommended dosage is 9 breaths per treatment session, 4 times daily.
2.2 Patients with Hepatic Insufficiency Plasma clearance of treprostinil is reduced in patients with hepatic insufficiency. Patients with hepatic insufficiency may therefore be at increased risk of dose-dependent adverse reactions because of an increase in systemic exposure [see WARNINGS AND PRECAUTIONS (5.3), USE IN SPECIFIC POPULATIONS (8.6) and CLINICAL PHARMACOLOGY (12.3)].
2.3 Patients with Renal Insufficiency Plasma clearance of treprostinil may be reduced in patients with renal insufficiency, since treprostinil and its metabolites are excreted mainly through the urinary route. Patients with renal insufficiency may therefore be at increased risk of dose-dependent adverse reactions [see WARNINGS AND PRECAUTIONS (5.3), USE IN SPECIFIC POPULATIONS (8.7) and CLINICAL PHARMACOLOGY (12.3)].
2.4 Administration Tyvaso must be used only with the Tyvaso Inhalation System. Patients should follow the instructions for use for operation of the Tyvaso Inhalation System and for daily cleaning of the device components after the last treatment session of the day. To avoid potential interruptions in drug delivery because of equipment malfunction, patients should have access to a back-up Tyvaso Inhalation System device.
Do not mix Tyvaso with other medications in the Tyvaso Inhalation System. Compatibility of Tyvaso with other medications has not been studied.
The Tyvaso Inhalation System should be prepared for use each day according to the instructions for use. One ampule of Tyvaso contains a sufficient volume of medication for all 4 treatment sessions in a single day. Prior to the first treatment session, the patient should twist the top off a single Tyvaso ampule and squeeze the entire contents into the medicine cup. Between each of the 4 daily treatment sessions, the device should be capped and stored upright with the remaining medication inside.
At the end of each day, the medicine cup and any remaining medication must be discarded. The device must be cleaned each day according to the instructions for use.
Avoid skin or eye contact with Tyvaso solution. Do not orally ingest the Tyvaso solution.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Treprostinil (inhalant) in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Treprostinil (inhalant) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Treprostinil (inhalant) FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Treprostinil (inhalant) in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Treprostinil (inhalant) in pediatric patients.
Contraindications
None.
Warnings
5.1 Patients with Pulmonary Disease or Pulmonary Infections The safety and efficacy of Tyvaso have not been established in patients with significant underlying lung disease (e.g., asthma or chronic obstructive pulmonary disease). Patients with acute pulmonary infections should be carefully monitored to detect any worsening of lung disease and loss of drug effect.
5.2 Risk of Symptomatic Hypotension Treprostinil is a pulmonary and systemic vasodilator. In patients with low systemic arterial pressure, treatment with Tyvaso may produce symptomatic hypotension.
5.3 Patients with Hepatic or Renal Insufficiency Titrate slowly in patients with hepatic or renal insufficiency, because such patients will likely be exposed to greater systemic concentrations relative to patients with normal hepatic or renal function [see DOSAGE AND ADMINISTRATION (2.2, 2.3), USE IN SPECIFIC POPULATIONS (8.6, 8.7) and CLINICAL PHARMACOLOGY (12.3)].
5.4 Risk of Bleeding Since Tyvaso inhibits platelet aggregation, there may be an increased risk of bleeding, particularly among patients receiving anticoagulant therapy.
5.5 Effect of Other Drugs on Treprostinil Co-administration of a cytochrome P450 (CYP) 2C8 enzyme inhibitor (e.g., gemfibrozil) may increase exposure (both Cmax and AUC) to treprostinil. Co-administration of a CYP2C8 enzyme inducer (e.g., rifampin) may decrease exposure to treprostinil. Increased exposure is likely to increase adverse events associated with treprostinil administration, whereas decreased exposure is likely to reduce clinical effectiveness [see DRUG INTERACTIONS (7.5) and CLINICAL PHARMACOLOGY (12.3)].
Adverse Reactions
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In a 12-week placebo-controlled study (TRIUMPH I) of 235 patients with PAH (WHO Group 1 and nearly all NYHA Functional Class III), the most commonly reported adverse reactions on Tyvaso included: cough and throat irritation; headache, gastrointestinal effects, muscle, jaw or bone pain, flushing and syncope. Table 1 lists the adverse reactions that occurred at a rate of at least 4% and were more frequent in patients treated with Tyvaso than with placebo.
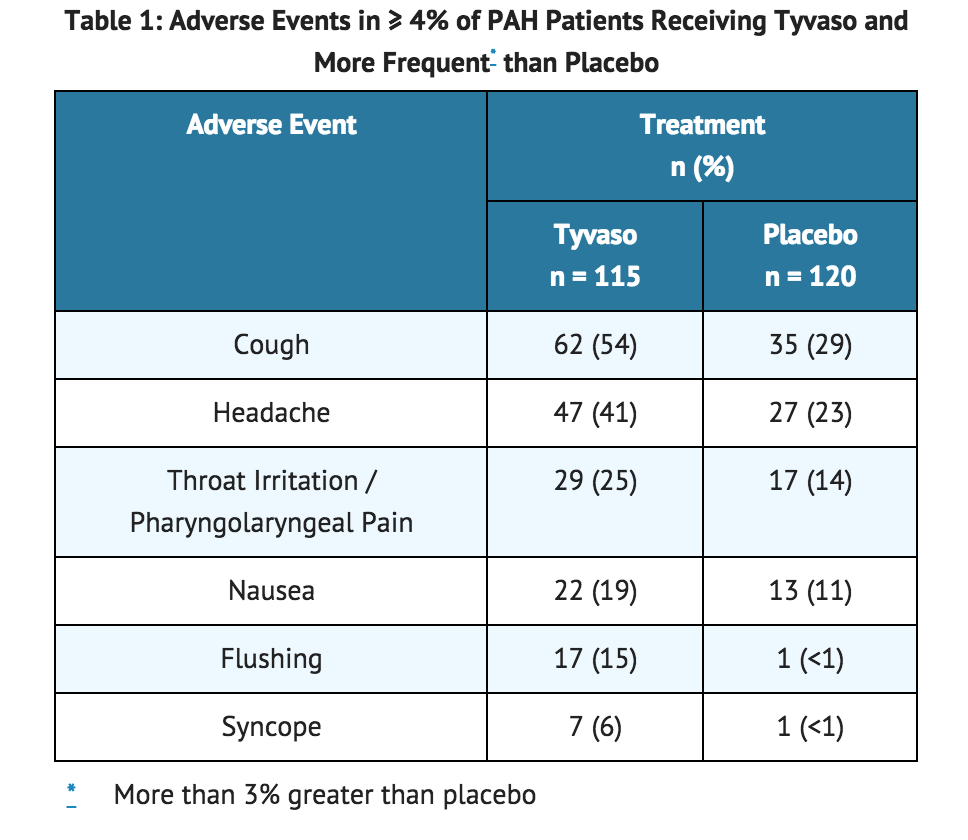
The safety of Tyvaso was also studied in a long-term, open-label extension study in which 206 patients were dosed for a mean duration of 2.3 years, with a maximum exposure of 5.4 years. Eighty-nine (89%) percent of patients achieved the target dose of nine breaths, four times daily. Forty-two (42%) percent achieved a dose of 12 breaths four times daily. The adverse events during this chronic dosing study were qualitatively similar to those observed in the 12-week placebo controlled trial.
Adverse Events Associated with Route of Administration
Adverse events in the treated group during the double-blind and open-label phase reflecting irritation to the respiratory tract included: cough, throat irritation, pharyngeal pain, epistaxis, hemoptysis and wheezing. Serious adverse events during the open-label portion of the study included pneumonia in fifteen subjects. There were three serious episodes of hemoptysis (one fatal) noted during the open-label experience.
Postmarketing Experience
The following adverse reaction has been identified during the postapproval use of Tyvaso. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure:
Angioedema.
Drug Interactions
Pharmacokinetic/pharmacodynamic interaction studies have not been conducted with inhaled treprostinil (Tyvaso); however, some of such studies have been conducted with orally (treprostinil diolamine) and subcutaneously administered treprostinil (Remodulin®).
Pharmacodynamics
7.1 Antihypertensive Agents or Other Vasodilators Concomitant administration of Tyvaso with diuretics, antihypertensive agents or other vasodilators may increase the risk of symptomatic hypotension.
7.2 Anticoagulants Since treprostinil inhibits platelet aggregation, there may be an increased risk of bleeding, particularly among patients receiving anticoagulants.
Pharmacokinetics
7.3 Bosentan In a human pharmacokinetic study conducted with bosentan (250 mg/day) and an oral formulation of treprostinil (treprostinil diolamine), no pharmacokinetic interactions between treprostinil and bosentan were observed.
7.4 Sildenafil In a human pharmacokinetic study conducted with sildenafil (60 mg/day) and an oral formulation of treprostinil (treprostinil diolamine), no pharmacokinetic interactions between treprostinil and sildenafil were observed.
7.5 Effect of Cytochrome P450 Inhibitors and Inducers In vitro studies of human hepatic microsomes showed that treprostinil does not inhibit cytochrome P450 (CYP) isoenzymes CYP1A2, CYP2A6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1 and CYP3A. Additionally, treprostinil does not induce cytochrome P450 isoenzymes CYP1A2, CYP2B6, CYP2C9, CYP2C19, and CYP3A.
Human pharmacokinetic studies with an oral formulation of treprostinil (treprostinil diolamine) indicated that co-administration of the cytochrome P450 (CYP) 2C8 enzyme inhibitor gemfibrozil increases exposure (both Cmax and AUC) to treprostinil. Co-administration of the CYP2C8 enzyme inducer rifampin decreases exposure to treprostinil. It is unclear if the safety and efficacy of treprostinil by the inhalation route are altered by inhibitors or inducers of CYP2C8 [see WARNINGS AND PRECAUTIONS (5.5)].
7.6 Effect of Other Drugs on Treprostinil Drug interaction studies have been carried out with treprostinil (oral or subcutaneous) co-administered with acetaminophen (4 g/day), warfarin (25 mg/day), and fluconazole (200 mg/day), respectively in healthy volunteers. These studies did not show a clinically significant effect on the pharmacokinetics of treprostinil. Treprostinil does not affect the pharmacokinetics or pharmacodynamics of warfarin. The pharmacokinetics of R- and S- warfarin and the INR in healthy subjects given a single 25 mg dose of warfarin were unaffected by continuous subcutaneous infusion of treprostinil at an infusion rate of 10 ng/kg/min.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): B
There are no adequate and well controlled studies with Tyvaso in pregnant women. Animal reproduction studies have not been conducted with treprostinil administered by the inhalation route. However, studies in pregnant rabbits using continuous subcutaneous (sc) infusions of treprostinil sodium at infusion rates higher than the recommended human sc infusion rate resulted in an increased incidence of fetal skeletal variations associated with maternal toxicity [see NONCLINICAL TOXICOLOGY (13.3)]. Animal reproduction studies are not always predictive of human response.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Treprostinil (inhalant) in women who are pregnant.
Labor and Delivery
No treprostinil treatment-related effects on labor and delivery were seen in animal studies. The effect of treprostinil on labor and delivery in humans is unknown.
Nursing Mothers
It is not known whether treprostinil is excreted in human milk.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established. Clinical studies of Tyvaso did not include patients younger than 18 years to determine whether they respond differently from older patients.
Geriatic Use
Clinical studies of Tyvaso did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently from younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of hepatic, renal, or cardiac dysfunction, and of concomitant diseases or other drug therapy.
Gender
There is no FDA guidance on the use of Treprostinil (inhalant) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Treprostinil (inhalant) with respect to specific racial populations.
Renal Impairment
No studies have been performed in patients with renal insufficiency. Since treprostinil and its metabolites are excreted mainly through the urinary route, patients with renal insufficiency may have decreased clearance of the drug and its metabolites and consequently, dose-related adverse outcomes may be more frequent [see CLINICAL PHARMACOLOGY (12.3), DOSAGE AND ADMINISTRATION (2.3) and WARNINGS AND PRECAUTIONS (5.3)].
Hepatic Impairment
Plasma clearance of treprostinil, delivered subcutaneously, was reduced up to 80% in subjects with mild-to-moderate hepatic insufficiency. Uptitrate slowly when treating patients with hepatic insufficiency because of the risk of an increase in systemic exposure which may lead to an increase in dose-dependent adverse effects. Treprostinil has not been studied in patients with severe hepatic insufficiency [see CLINICAL PHARMACOLOGY (12.3), DOSAGE AND ADMINISTRATION (2.2) and WARNINGS AND PRECAUTIONS (5.3)].
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Treprostinil (inhalant) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Treprostinil (inhalant) in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Treprostinil (inhalant) Administration in the drug label.
Monitoring
There is limited information regarding Treprostinil (inhalant) Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Treprostinil (inhalant) and IV administrations.
Overdosage
In general, symptoms of overdose with Tyvaso include flushing, headache, hypotension, nausea, vomiting, and diarrhea. Provide general supportive care until the symptoms of overdose have resolved.
Pharmacology
There is limited information regarding Treprostinil (inhalant) Pharmacology in the drug label.
Mechanism of Action
Treprostinil is a prostacyclin analogue. The major pharmacologic actions of treprostinil are direct vasodilation of pulmonary and systemic arterial vascular beds and inhibition of platelet aggregation.
Structure
Tyvaso is a sterile formulation of treprostinil intended for administration by oral inhalation using the Tyvaso Inhalation System. Tyvaso is supplied in 2.9 mL low density polyethylene (LDPE) ampules, containing 1.74 mg treprostinil (0.6 mg/mL). Each ampule also contains 18.9 mg sodium chloride, 18.3 mg sodium citrate, 0.58 mg sodium hydroxide, 11.7 mg 1 N hydrochloric acid, and water for injection. Sodium hydroxide and hydrochloric acid may be added to adjust pH between 6.0 and 7.2.
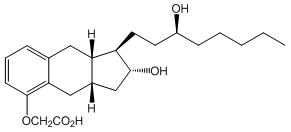
Treprostinil is (1R,2R,3aS,9aS)-[2,3,3a,4,9,9a-hexahydro-2-hydroxy-1-[(3S)-3-hydroxyoctyl]-1H-benz[f]inden-5-yl]oxy]acetic acid. Treprostinil has a molecular weight of 390.51 and a molecular formula of C23H34O5.
The structural formula of treprostinil is:
Pharmacodynamics
In a clinical trial of 240 healthy volunteers, single doses of Tyvaso 54 mcg (the target maintenance dose per session) and 84 mcg (supratherapeutic inhalation dose) prolonged the corrected QTc interval by approximately 10 ms. The QTc effect dissipated rapidly as the concentration of treprostinil decreased.
Pharmacokinetics
Pharmacokinetic information for single doses of inhaled treprostinil was obtained in healthy volunteers in three separate studies. Treprostinil systemic exposure (AUC and Cmax) post-inhalation was shown to be proportional to the doses administered (18 mcg – 90 mcg).
Absorption and Distribution
In a three-period crossover study, the bioavailability of two single doses of Tyvaso (18 mcg and 36 mcg) was compared with that of intravenous treprostinil in 18 healthy volunteers. Mean estimates of the absolute systemic bioavailability of treprostinil after inhalation were approximately 64% (18 mcg) and 72% (36 mcg).
Treprostinil plasma exposure data were obtained from two studies at the target maintenance dose, 54 mcg. The mean Cmax at the target dose was 0.91 and 1.32 ng/mL with corresponding mean Tmax of 0.25 and 0.12 hr, respectively. The mean AUC for the 54 mcg dose was 0.81 and 0.97 hr∙ng/mL, respectively.
Following parenteral infusion, the apparent steady state volume of distribution (Vss) of treprostinil is approximately 14 L/70 kg ideal body weight.
In vitro treprostinil is 91% bound to human plasma proteins over the 330-10,000 mcg/L concentration range.
Metabolism and Excretion
Of subcutaneously administered treprostinil, only 4% is excreted unchanged in urine. Treprostinil is substantially metabolized by the liver, primarily by CYP2C8. Metabolites are excreted in urine (79%) and feces (13%) over 10 days. Five apparently inactive metabolites were detected in the urine, each accounting for 10-15% of the dose administered. Four of the metabolites are products of oxidation of the 3-hydroxyloctyl side chain and one is a glucuroconjugated derivative (treprostinil glucuronide).
The elimination of treprostinil (following subcutaneous administration of treprostinil) is biphasic, with a terminal elimination half-life of approximately 4 hours using a two compartment model.
Special Populations
Hepatic Insufficiency
Plasma clearance of treprostinil, delivered subcutaneously, was reduced up to 80% in subjects presenting with mild-to-moderate hepatic insufficiency. Treprostinil has not been studied in patients with severe hepatic insufficiency [see DOSAGE AND ADMINISTRATION (2.2), WARNINGS AND PRECAUTIONS (5.3) and USE IN SPECIFIC POPULATIONS (8.6)].
Renal Insufficiency
No studies have been performed in patients with renal insufficiency; therefore, since treprostinil and its metabolites are excreted mainly through the urinary route, there is the potential for an increase in both parent drug and its metabolites and an increase in systemic exposure [see DOSAGE AND ADMINISTRATION (2.3), WARNINGS AND PRECAUTIONS (5.3) and USE IN SPECIFIC POPULATIONS (8.7)].
Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility A two-year rat carcinogenicity study was performed with treprostinil inhalation at target doses of 5.26, 10.6, and 34.1 mcg/kg/day. There was no evidence for carcinogenic potential associated with treprostinil inhalation in rats at systemic exposure levels up to 35 times the clinical exposure at the target maintenance dose of 54 mcg. In vitro and in vivo genetic toxicology studies did not demonstrate any mutagenic or clastogenic effects of treprostinil. Treprostinil sodium did not affect fertility or mating performance of male or female rats given continuous subcutaneous (sc) infusions at rates of up to 450 ng treprostinil/kg/min [about 59 times the recommended starting human sc infusion rate (1.25 ng/kg/min) and 8 times the average rate (9.3 ng/kg/min) achieved in clinical trials, on a ng/m2 basis]. In this study, males were dosed from 10 weeks prior to mating and through the 2-week mating period. Females were dosed from 2 weeks prior to mating until gestational day 6.
13.3 Developmental Toxicity In pregnant rats, continuous sc infusions of treprostinil sodium during organogenesis and late gestational development, at rates as high as 900 ng treprostinil/kg/min (about 117 times the recommended starting human sc infusion rate and about 16 times the average rate achieved in clinical trials, on a ng/m2 basis), resulted in no evidence of harm to the fetus. In pregnant rabbits, effects of continuous sc infusions of treprostinil during organogenesis were limited to an increased incidence of fetal skeletal variations (bilateral full rib or right rudimentary rib on lumbar vertebra 1) associated with maternal toxicity (reduction in body weight and food consumption) at an infusion rate of 150 ng treprostinil/kg/min (about 41 times the starting human sc infusion rate and 5 times the average rate achieved in clinical trials, on a ng/m2 basis).
13.4 Inhalational Toxicity Rats and dogs that received daily administrations of treprostinil by inhalation for 3 months developed respiratory tract lesions (respiratory epithelial degeneration, goblet cell hyperplasia/hypertrophy, epithelial ulceration, squamous epithelial degeneration and necrosis, and lung hemorrhage). Some of the same lesions seen in animals sacrificed at the end of treatment (larynx, lung and nasal cavity lesions in rats, and lesions of the larynx in dogs) were also observed in animals sacrificed after a 4-week recovery period. Rats also developed cardiac changes (degeneration/fibrosis). A no-effect dose level for these effects was not demonstrated in rats (doses as low as 7 µg/kg/day were administered); whereas 107 µg/kg/day was a no-effect dose level in dogs.
In a 2-year rat study with treprostinil inhalation at target doses of 5.26, 10.6, and 34.1 mcg/kg/day, there were more deaths (11) in the mid and high dose treprostinil groups during the first 9 weeks of the study, compared to 1 in control groups. At the high dose level, males showed a higher incidence of inflammation in teeth and preputial gland, and females showed higher incidences of inflammation and urothelial hyperplasia in the urinary bladder. The exposures in rats at mid and high dose levels were about 15 and 35 times, respectively, the clinical exposure at the target maintenance dose of 54 mcg.
Clinical Studies
There is limited information regarding Treprostinil (inhalant) Clinical Studies in the drug label.
How Supplied
There is limited information regarding Treprostinil (inhalant) How Supplied in the drug label.
Storage
There is limited information regarding Treprostinil (inhalant) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Treprostinil (inhalant) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Treprostinil (inhalant) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Treprostinil (inhalant) Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Treprostinil (inhalant) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Treprostinil (inhalant) Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Treprostinil (inhalant) Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.