Toloxatone
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C11H13NO3 |
| Molar mass | 207.226 |
| tradename = | pregnancy_category = | legal_status = Uncontrolled | routes_of_administration = Oral
| bioavailability = | protein_bound = | metabolism = | elimination_half-life = | excretion =
| CAS_number_Ref =
| CAS_number = 29218-27-7
| ATC_prefix = N06
| ATC_suffix = AG03
| PubChem = 34521
| DrugBank_Ref =
| DrugBank =
| ChemSpiderID_Ref =
| ChemSpiderID = 31769
| UNII_Ref =
| UNII = 5T206015T5
| KEGG_Ref =
| KEGG = D02559
| ChEMBL_Ref =
| ChEMBL = 18116
| C=11 | H=13 | N=1 | O=3
| molecular_weight = 207.226 g/mol
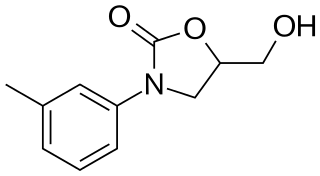
| smiles = O=C2OC(CO)CN2c1cccc(c1)C
| InChI = 1/C11H13NO3/c1-8-3-2-4-9(5-8)12-6-10(7-13)15-11(12)14/h2-5,10,13H,6-7H2,1H3
| InChIKey = MXUNKHLAEDCYJL-UHFFFAOYAQ
| StdInChI_Ref =
| StdInChI = 1S/C11H13NO3/c1-8-3-2-4-9(5-8)12-6-10(7-13)15-11(12)14/h2-5,10,13H,6-7H2,1H3
| StdInChIKey_Ref =
| StdInChIKey = MXUNKHLAEDCYJL-UHFFFAOYSA-N
}}
}}
|
WikiDoc Resources for Toloxatone |
|
Articles |
|---|
|
Most recent articles on Toloxatone |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Toloxatone at Clinical Trials.gov Clinical Trials on Toloxatone at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Toloxatone
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Toloxatone Discussion groups on Toloxatone Patient Handouts on Toloxatone Directions to Hospitals Treating Toloxatone Risk calculators and risk factors for Toloxatone
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Toloxatone |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Toloxatone (Humoryl) is an antidepressant launched in 1984 in France for the treatment of depression. It acts as a selective reversible inhibitor of MAO-A (RIMA).[1]
References
- ↑ Berlin I, Zimmer R, Thiede HM; et al. (December 1990). "Comparison of the monoamine oxidase inhibiting properties of two reversible and selective monoamine oxidase-A inhibitors moclobemide and toloxatone, and assessment of their effect on psychometric performance in healthy subjects". British Journal of Clinical Pharmacology. 30 (6): 805–16. doi:10.1111/j.1365-2125.1990.tb05445.x. PMC 1368300. PMID 1705137.
- Pages with script errors
- CS1 maint: Explicit use of et al.
- CS1 maint: Multiple names: authors list
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Articles with changed CASNo identifier
- Monoamine oxidase inhibitors
- Alcohols
- Carbamates
- Drug