Rosuvastatin: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{DrugProjectFormSinglePage | |||
{{ | |genericName=generic name | ||
|aOrAn=a | |||
|drugClass=Adrenergic receptor agonist | |||
|indication=a list of indications, separated by commas. | |||
|hasBlackBoxWarning=Yes | |||
|adverseReactions=a list of adverse reactions, separated by commas. | |||
|blackBoxWarningTitle=Warning Title | |||
|blackBoxWarningBody=<i><span style="color:#FF0000;">Condition Name:</span></i> (Content) | |||
|fdaLIADAdult======Condition 1===== | |||
* Dosing Information | |||
:* (Dosage) | |||
== | =====Condition 2===== | ||
* Dosing Information | |||
:* (Dosage) | |||
|offLabelAdultGuideSupport======Condition 1===== | |||
* Developed by: (Organisation) | |||
* Class of Recommendation: (Class) (Link) | |||
* Strength of Evidence: (Category A/B/C) (Link) | |||
* Dosing Information/Recommendation | |||
:* (Dosage) | |||
=====Condition 2===== | |||
* Developed by: (Organisation) | |||
* Class of Recommendation: (Class) (Link) | |||
* Strength of Evidence: (Category A/B/C) (Link) | |||
* Dosing Information/Recommendation | |||
:* (Dosage) | |||
|offLabelAdultNoGuideSupport======Condition 1===== | |||
* Dosing Information | |||
:* (Dosage) | |||
=====Condition 2===== | |||
* Dosing Information | |||
:* (Dosage) | |||
=====Condition 3===== | |||
* Dosing Information | |||
:* (Dosage) | |||
|fdaLIADPed======Condition 1===== | |||
* Dosing Information | |||
:* (Dosage) | |||
=====Condition 2===== | |||
* Dosing Information | |||
:* (Dosage) | |||
|offLabelPedGuideSupport======Condition 1===== | |||
* Developed by: (Organisation) | |||
* Class of Recommendation: (Class) (Link) | |||
* Strength of Evidence: (Category A/B/C) (Link) | |||
* Dosing Information/Recommendation | |||
:* (Dosage) | |||
=====Condition 2===== | |||
* Developed by: (Organisation) | |||
* Class of Recommendation: (Class) (Link) | |||
* Strength of Evidence: (Category A/B/C) (Link) | |||
* Dosing Information/Recommendation | |||
:* (Dosage) | |||
|offLabelPedNoGuideSupport======Condition 1===== | |||
* Dosing Information | |||
:* (Dosage) | |||
=====Condition 2===== | |||
* Dosing Information | |||
:* (Dosage) | |||
=====Condition 3===== | |||
* Dosing Information | |||
:* (Dosage) | |||
|contraindications=* Condition 1 | |||
* Condition 2 | |||
* Condition 3 | |||
* Condition 4 | |||
* Condition 5 | |||
|warnings======Conidition 1===== | |||
(Description) | |||
|clinicalTrials=======Central Nervous System====== | |||
: (list/description of adverse reactions) | |||
======Cardiovascular====== | |||
: (list/description of adverse reactions) | |||
======Respiratory====== | |||
: (list/description of adverse reactions) | |||
======Gastrointestinal====== | |||
: (list/description of adverse reactions) | |||
======Hypersensitive Reactions====== | |||
: (list/description of adverse reactions) | |||
======Miscellaneous====== | |||
: (list/description of adverse reactions) | |||
=====Condition 2===== | |||
======Central Nervous System====== | |||
: (list/description of adverse reactions) | |||
======Cardiovascular====== | |||
: (list/description of adverse reactions) | |||
======Respiratory====== | |||
: (list/description of adverse reactions) | |||
======Gastrointestinal====== | |||
: (list/description of adverse reactions) | |||
======Hypersensitive Reactions====== | |||
: (list/description of adverse reactions) | |||
======Miscellaneous====== | |||
: (list/description of adverse reactions) | |||
|postmarketing=(Description) | |||
|drugInteractions=* Drug 1 | |||
* Drug 2 | |||
* Drug 3 | |||
* Drug 4 | |||
* Drug 5 | |||
=====Drug 1===== | |||
(Description) | |||
=====Drug 2===== | |||
(Description) | |||
=====Drug 3===== | |||
(Description) | |||
=====Drug 4===== | |||
(Description) | |||
=====Drug 5===== | |||
(Description) | |||
|useInPregnancyFDA=(Description) | |||
|useInPregnancyAUS=(Description) | |||
|useInLaborDelivery=(Description) | |||
|useInNursing=(Description) | |||
|useInPed=(Description) | |||
|useInGeri=(Description) | |||
|useInGender=(Description) | |||
|useInRace=(Description) | |||
|useInRenalImpair=(Description) | |||
|useInHepaticImpair=(Description) | |||
|useInReproPotential=(Description) | |||
|useInImmunocomp=(Description) | |||
|othersTitle=Others | |||
|useInOthers=(Description) | |||
|administration=(Oral/Intravenous/etc) | |||
|monitoring======Condition 1===== | |||
(Description regarding monitoring, from ''Warnings'' section) | |||
=====Condition 2===== | |||
(Description regarding monitoring, from ''Warnings'' section) | |||
=====Condition 3===== | |||
(Description regarding monitoring, from ''Warnings'' section) | |||
|IVCompat====Solution=== | |||
====Compatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Not Tested==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Variable==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Incompatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
===Y-Site=== | |||
====Compatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Not Tested==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Variable==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Incompatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
===Admixture=== | |||
====Compatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Not Tested==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Variable==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Incompatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
===Syringe=== | |||
====Compatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Not Tested==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Variable==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Incompatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
===TPN/TNA=== | |||
====Compatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Not Tested==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Variable==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
====Incompatible==== | |||
* Solution 1 | |||
* Solution 2 | |||
* Solution 3 | |||
|overdose====Acute Overdose=== | |||
====Signs and Symptoms==== | |||
(Description) | |||
====Management==== | |||
(Description) | |||
===Chronic Overdose=== | |||
====Signs and Symptoms==== | |||
(Description) | |||
====Management==== | |||
(Description) | |||
|drugBox={{Drugbox2 | |||
| verifiedrevid = | |||
| IUPAC_name = | |||
| image = | |||
| drug_name = | |||
<!--Clinical data--> | |||
| tradename = | |||
| MedlinePlus = | |||
| licence_US = | |||
| pregnancy_AU = | |||
| pregnancy_US = | |||
| legal_status = | |||
| routes_of_administration = | |||
<!--Pharmacokinetic data--> | |||
| bioavailability = | |||
| metabolism = | |||
| elimination_half-life = | |||
| excretion = | |||
<!--Identifiers--> | |||
| CAS_number_Ref = | |||
| CAS_number = | |||
| ATC_prefix = | |||
| ATC_suffix = | |||
| PubChem = | |||
| IUPHAR_ligand = | |||
| DrugBank_Ref = | |||
| DrugBank = | |||
| ChemSpiderID_Ref = | |||
| ChemSpiderID = | |||
| UNII_Ref = | |||
| UNII = | |||
| KEGG_Ref = | |||
| KEGG = | |||
| ChEBI_Ref = | |||
| ChEBI = | |||
| ChEMBL_Ref = | |||
| ChEMBL = | |||
<!--Chemical data--> | |||
| C= | H= | N= | O= | |||
| molecular_weight = | |||
| smiles = | |||
| InChI = | |||
| InChIKey = | |||
| StdInChI_Ref = | |||
| StdInChI = | |||
| StdInChIKey_Ref = | |||
| StdInChIKey = | |||
| melting_point = | |||
}} | |||
|mechAction=(Description) | |||
|structure=(Description with picture) | |||
|PD=(Description) | |||
|PK=(Description) | |||
|nonClinToxic=(Description) | |||
|clinicalStudies======Condition 1===== | |||
(Description) | |||
=====Condition 2===== | |||
(Description) | |||
=====Condition 3===== | |||
(Description) | |||
|howSupplied=(Description) | |||
|fdaPatientInfo=(Patient Counseling Information) | |||
|nlmPatientInfo=(Link to patient information page) | |||
|lookAlike=* (Paired Confused Name 1a) — (Paired Confused Name 1b) | |||
* (Paired Confused Name 2a) — (Paired Confused Name 2b) | |||
* (Paired Confused Name 3a) — (Paired Confused Name 3b) | |||
|drugShortage=Drug Shortage | |||
}} | |||
Revision as of 14:03, 30 June 2014
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1];
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Warning Title
See full prescribing information for complete Boxed Warning.
Condition Name: (Content)
|
Overview
Rosuvastatin is a Adrenergic receptor agonist that is FDA approved for the {{{indicationType}}} of a list of indications, separated by commas.. There is a Black Box Warning for this drug as shown here. Common adverse reactions include a list of adverse reactions, separated by commas..
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Contraindications
- Condition 1
- Condition 2
- Condition 3
- Condition 4
- Condition 5
Warnings
|
Warning Title
See full prescribing information for complete Boxed Warning.
Condition Name: (Content)
|
Conidition 1
(Description)
Adverse Reactions
Clinical Trials Experience
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Condition 2
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Postmarketing Experience
(Description)
Drug Interactions
- Drug 1
- Drug 2
- Drug 3
- Drug 4
- Drug 5
Drug 1
(Description)
Drug 2
(Description)
Drug 3
(Description)
Drug 4
(Description)
Drug 5
(Description)
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
(Description)
Pregnancy Category (AUS):
(Description)
Labor and Delivery
(Description)
Nursing Mothers
(Description)
Pediatric Use
(Description)
Geriatic Use
(Description)
Gender
(Description)
Race
(Description)
Renal Impairment
(Description)
Hepatic Impairment
(Description)
Females of Reproductive Potential and Males
(Description)
Immunocompromised Patients
(Description)
Others
(Description)
Administration and Monitoring
Administration
(Oral/Intravenous/etc)
Monitoring
Condition 1
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section)
IV Compatibility
Solution
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Y-Site
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Admixture
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Syringe
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
TPN/TNA
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Overdosage
Acute Overdose
Signs and Symptoms
(Description)
Management
(Description)
Chronic Overdose
Signs and Symptoms
(Description)
Management
(Description)
Pharmacology
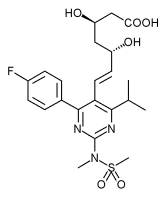
| |
Rosuvastatin
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
(Description)
Structure
(Description with picture)
Pharmacodynamics
(Description)
Pharmacokinetics
(Description)
Nonclinical Toxicology
(Description)
Clinical Studies
Condition 1
(Description)
Condition 2
(Description)
Condition 3
(Description)
How Supplied
(Description)
Storage
There is limited information regarding Rosuvastatin Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Rosuvastatin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Rosuvastatin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
(Patient Counseling Information)
Precautions with Alcohol
Alcohol-Rosuvastatin interaction has not been established. Talk to your doctor regarding the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Rosuvastatin Brand Names in the drug label.
Look-Alike Drug Names
- (Paired Confused Name 1a) — (Paired Confused Name 1b)
- (Paired Confused Name 2a) — (Paired Confused Name 2b)
- (Paired Confused Name 3a) — (Paired Confused Name 3b)
Drug Shortage Status
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.