Enflurane
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Chetan Lokhande, M.B.B.S [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Enflurane is a general anesthetic that is FDA approved for the {{{indicationType}}} of analgesia for labor/delivery, for vaginal delivery, anesthesia - obstetric procedure, as supplement to other general anesthetic agents during cesarean section, general anesthesia.. Common adverse reactions include gastrointestinal: nausea, vomiting, musculoskeletal: involuntary movement, neurologic: shivering.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Analgesia for labor/delivery, for vaginal delivery: for vaginal delivery, 0.25 to 1% concentration.
- Anesthesia - Obstetric procedure, As supplement to other general anesthetic agents during Cesarean section: during cesarean section, 0.5 to 1% concentration (to supplement other anesthetics).
- General anesthesia: induction, 2 to 4.5% concentration via vaporizer; with oxygen or combination with oxygen-nitrous oxide mixtures.
- General anesthesia: maintenance, 0.5 to 3% concentration; max 3%
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- General anesthesia - Surgical procedure on eye proper
Non–Guideline-Supported Use
There is limited information about Off-Label Non–Guideline-Supported Use of Enflurane in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Enflurane FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information about Off-Label Guideline-Supported Use of Enflurane in pediatric patients.
Non–Guideline-Supported Use
There is limited information about Off-Label Non–Guideline-Supported Use of Enflurane in pediatric patients.
Contraindications
- Seizure disorders (see Warnings)
- Known sensitivity to Enflurane or other halogenated anesthetics. Known or suspected genetic susceptibility to malignant hyperthermia
Warnings
Perioperative Hyperkalemia
- Use of inhaled anesthetic agents has been associated with rare increases in serum potassium levels that have resulted in cardiac arrhythmias and death in pediatric patients during the postoperative period. Patients with latent as well as overt neuromuscular disease, particularly Duchenne muscular dystrophy, appear to be most vulnerable. Concomitant use of succinylcholine has been associated with most, but not all, of these cases. These patients also experienced significant elevations in serum creatinine kinase levels and, in some cases, changes in urine consistent with myoglobinuria.
- Despite the similarity in presentation to malignant hyperthermia, none of these patients exhibited signs or symptoms of muscle rigidity or hypermetabolic state. Early and aggressive intervention to treat the hyperkalemia and resistant arrhythmias is recommended, as is subsequent evaluation for latent neuromuscular disease.
Malignant Hyperthermia
- In susceptible individuals, enflurane anesthesia may trigger a skeletal muscle hypermetabolic state leading to high oxygen demand and the clinical syndrome known as malignant hyperthermia. The syndrome includes nonspecific features such as muscle rigidity, tachycardia, tachypnea, cyanosis, arrhythmias, and unstable blood pressure. (It should also be noted that many of these nonspecificsigns may appear with light anesthesia, acute hypoxia, etc. The syndrome of malignant hyperthermia secondary to enflurane appears to be rare; by March 1980, 35 cases had been reported in North America for an approximate incidence of 1:725,000 enflurane anesthetics.) An increase in overall metabolism may be reflected in an elevated temperature (which may rise rapidly early or late in the case, but usually is not the first sign of augmented metabolism) and an increased usage of the CO2 absorption system (hot cannister). PaO2 and pH may decrease, and hyperkalemia and a base deficit may appear. Treatment includes discontinuance of triggering agents (e.g., enflurane), administration of intravenous dantrolene sodium, and application of supportive therapy. Such therapy includes vigorous efforts to restore body temperature to normal, respiratory and circulatory support as indicated, and management of electrolyte-fluid-acid-base derangement. (Consult prescribing information for dantrolene sodium intravenous for additional information on patient management.) Renal failure may appear later, and urine flow should be sustained if possible.
- Increasing depth of anesthesia with Enflurane may produce a change in the electroencephalogram characterized by high voltage, fast frequency, progressing through spike-dome complexes alternating with periods of electrical silence to frank seizure activity. The latter may or may not be associated with motor movement. Motor activity, when encountered, generally consists of twitching or “jerks” of various muscle groups; it is self-limiting and can be terminated by lowering the anesthetic concentration. This electroencephalographic pattern associated with deep anesthesia is exacerbated by low arterial carbon dioxide tension. A reduction in ventilation and anesthetic concentrations usually suffices to eliminate seizure activity. Cerebral blood flow and metabolism studies in normal volunteers immediately following seizure activity show no evidence of cerebral hypoxia. Mental function testing does not reveal any impairment of performance following prolonged enflurane anesthesia associated with or not associated with seizure activity.
- Since levels of anesthesia may be altered easily and rapidly, only vaporizers producing predictable concentrations should be used.
- Hypotension and respiratory exchange can serve as a guide to depth of anesthesia. Deep levels of anesthesia may produce marked hypotension and respiratory depression When previous exposure to a halogenated anesthetic is known to have been followed by evidence of unexplained hepatic dysfunction, consideration should be given to use of an agent other than enflurane.
Adverse Reactions
Clinical Trials Experience
- Malignant hyperthermia (see Warnings).
- Motor activity exemplified by movements of various muscle groups and/or seizures may be encountered with deep levels of enflurane anesthesia, or light levels with hypocapnia.
- Hypotension, respiratory depression and hypoxia have been reported.
- Arrhythmias, shivering, nausea and vomiting have been reported.
- Elevation of the white blood count has been observed.
- Mild, moderate and severe liver injury, including hepatic failure, may rarely follow anesthesia with enflurane.
- Serum transaminases may be increased and histologic evidence of injury may be found. The histologic changes are neither unique nor consistent. In several of these cases, it has not been possible to exclude enflurane as the cause or as a contributing cause to liver injury. The incidence of unexplained hepatotoxicity following the administration of enflurane is unknown, but it appears to be rare and not dose related. Enflurane has also been associated with perioperative hyperkalemia (see Warnings). There have been rare post-marketing reports of hepatic failure and hepatic necrosis associated with the use of potent volatile anesthetic agents, including Enflurane. Due to the spontaneous nature of these reports, the actual incidence and relationship of Enflurane to these events cannot be established with certainty
Postmarketing Experience
There is limited information regarding Enflurane Postmarketing Experience in the drug label.
Drug Interactions
- The action of nondepolarizing relaxants is augmented by enflurane. Less than the usual amounts of these drugs should be used. If the usual amounts of nondepolarizing relaxants are given, the time for recovery from neuromuscular blockade will be longer in the presence of enflurane than when halothane or nitrous oxide with a balanced technique are used.
Use in Specific Populations
Pregnancy
- Reproduction studies have been performed in rats and rabbits at doses up to four times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to enflurane. There are, however, no adequate and well-controlled studies inpregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Enflurane in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Enflurane during labor and delivery.
Nursing Mothers
- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when enflurane is administered to a nursing woman.
Pediatric Use
There is no FDA guidance on the use of Enflurane in pediatric settings.
Geriatic Use
There is no FDA guidance on the use of Enflurane in geriatric settings.
Gender
There is no FDA guidance on the use of Enflurane with respect to specific gender populations.
Race
There is no FDA guidance on the use of Enflurane with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Enflurane in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Enflurane in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Enflurane in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Enflurane in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Enflurane Administration in the drug label.
Monitoring
There is limited information regarding Enflurane Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Enflurane and IV administrations.
Overdosage
- In the event of overdosage, or what may appear to be overdosage, the following action should be taken: Stop drug administration, establish a clear airway and initiate assisted or controlled ventilation with pure oxygen.
Pharmacology
| |
| |
Enflurane
| |
| Systematic (IUPAC) name | |
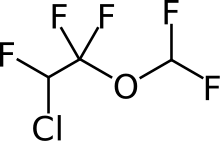
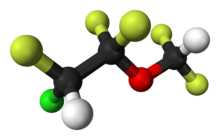
| (RS)-2-chloro-1-(difluoromethoxy)-1,1,2-trifluoro-ethane | |
| Identifiers | |
| CAS number | |
| ATC code | N01 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 184.492 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Protein binding | 97% |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
There is limited information regarding Enflurane Mechanism of Action in the drug label.
Structure
- Enflurane, USP, a nonflammable liquid administered by vaporizing, is a general inhalation anesthetic drug. It is 2-chloro-1,1,2-trifluoroethyl difluoromethyl ether, (CHF2OCF2CHFCl). The boiling point is 56.5°C at 760 mm Hg, and the vapor pressure (in mm Hg) is 175 at 20°C, 218 at 25°C, and 345 at 36°C. Vapor pressures can be calculated using the equation:
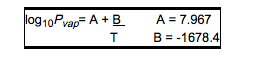
- T = ° C + 273.16 (Kelvin)
- Enflurane is a clear, colorless, stable liquid whose purity exceeds 99.9% (area percent by gas chromatography). No stabilizers are added as these have been found, through controlled laboratory tests, to be unnecessary even in the presence of ultraviolet light. Enflurane is stable to strong base, does not decompose in contact with soda lime (at normal operating temperatures) and does not react with aluminum, tin, brass, iron or copper. The partition coefficients of enflurane at 25°C are 74 in conductive rubber and 120 in polyvinyl chloride.
Pharmacodynamics
- Enflurane is an inhalation anesthetic. The MAC (minimum alveolar concentration) in man is 1.68% in pure oxygen, 0.57 in 70% nitrous oxide, 30% oxygen, and 1.17 in 30% nitrous oxide, 70% oxygen.Induction of and recovery from anesthesia with enflurane are rapid. Enflurane has a mild sweet odor. Enflurane may provide a mild stimulus to salivation or tracheobronchial secretions. Pharyngeal and laryngeal reflexes are readily obtunded. The level of anesthesia can be changed rapidly by changing the inspired enflurane concentration. Enflurane reduces ventilation as depth of anesthesia increases. High PaCO2 levels can be obtained at deeper levels of anesthesia if ventilation is not supported. Enflurane provokes a sigh response reminiscent of that seen with diethyl ether. There is a decrease in blood pressure with induction of anesthesia, followed by a return to near normal with surgical stimulation. Progressive increases in depth of anesthesia produce corresponding increases in hypotension. Heart rate remains relatively constant without significant bradycardia. Electrocardiographic monitoring or recordings indicate that cardiac rhythm remains stable. Elevation of the carbon dioxide level in arterial blood does not alter cardiac rhythm.
- Studies in man indicate a considerable margin of safety in the administration of Epinephrine containing solutions during enflurane anesthesia. Enflurane anesthesia has been used in excision of pheochromocytoma in man without ventricular arrhythmias. On the basis of studies in patients anesthetized with enflurane and injected with epinephrine-containing solutions to achieve hemostasis in a highly vascular area (transsphenoidal surgery), up to 2 micrograms per kilogram (2 μg/kg) of epinephrine may be injected subcutaneously over a 10 minute period in patients judged to have ordinary tolerance to epinephrine administration. This would represent up to 14 mL of 1:100,000 epinephrine-containing solution (10 μg/mL), or the equivalent quantity, in a 70 kilogram patient. This may be repeated up to 3 times per hour (total 42 mL per hour).The concomitant administration of lidocaine enhances the safety of the use of epinephrine during enflurane anesthesia. This effect of lidocain is dose related.All customary precautions in the use of vasoconstrictor substances should be observed. Muscle relaxation may be adequate for intra-abdominal operations at normal levels of anesthesia. Muscle relaxants may be used to achieve greater relaxation and all commonly used muscle relaxants are compatible with enflurane. The Nondepolarizing Muscle Relaxants Are Potentiated. In the normal 70 kg adult, 6 to 9 mg of d-tubocurarine or 1 to 1.5 mg of pancuronium will produce a 90% or greater depression of twitch height. Neostigmine does not reverse the direct effect of enflurane. Enflurane 0.25 to 1% (average 0.5%) provides analgesia equal to that produced by 30 to 60% (average 40%) nitrous oxide for vaginal delivery. With either agent, patients remain awake, cooperative and oriented. Maternal blood losses are comparable. These clinical approaches produce normal Apgar scores. Serial neurobehavioral testing of the newborn during the first 24 hours of life reveals that neither enflurane nor nitrous oxide analgesia is associated with obvious neurobehavioral alterations. Neither enflurane nor nitrous oxide when used for obstetrical analgesia alters BUN, creatinine, uric acid or osmolality.The only difference in the use of these two agents for obstetrical analgesia appears to be higher inspired oxygen concentration that may be used with enflurane. Analgetic doses of enflurane, up to approximately 1.0%, do not significantly depress the rate or force of uterine contraction during labor and delivery. A slowing of the rate of uterine contraction and a diminution of the force of uterine contraction is noted between the administration of 1.0 to 2.0% delivered enflurane; concentrations somewhere between 2.0 and 3.0% delivered enflurane may abolish uterine contractions. Enflurane displaces the myometrial response curve to oxytocin so that at lower concentrations of enflurane oxytocin will restore uterine contractions; however, as the dose of enflurane progresses (somewhere between 1.5 and 3% delivered enflurane) the response to oxytocin is diminished and then abolished. Uterine bleeding may be increased when enflurane is used in higher concentrations for vaginal delivery or to facilitate delivery by Cesarean section; however, this has not been demonstrated within the recommended dosage range (see Dosage And Administration section).
- Mean estimated blood loss in patients anesthetized for therapeutic termination of pregnancy with 1.0% enflurane in 70% nitrous oxide with oxygen is approximately twice that noted following therapeutic termination of pregnancy performed with the use of a local anesthetic technique (40 mL versus 20 mL).
Pharmacokinetics
- Biotransformation of enflurane in man results in low peak levels of serum fluoride averaging 15 μmol/L. These levels are well below the 50 μmol/L threshold level, which can produce minimal renal damage in normal subjects. However, patients chronically ingesting isoniazid or other hydrazine-containing compounds may metabolize greater amounts of enflurane. Although no significant renal dysfunction
has been found thus far in such patients, peak serum fluoride levels can exceed 50 μmol/L, particularly when anesthesia goes beyond 2 MAC hours. Depression of lymphocyte transformation does notfollow prolonged enflurane anesthesia in man in the absence of surgery. Thus enflurane does not depress this aspect of the immune response.
Nonclinical Toxicology
There is limited information regarding Enflurane Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Enflurane Clinical Studies in the drug label.
How Supplied
- Enflurane, USP is packaged in 250 mL amber-colored bottles.250mL - NDC 66794-010-25
Storage
- Store at room controlled room temperature 15° to 30°C (59° to 86°F). Enflurane, USP contains no additives and has been demonstrated to be stable at room temperature for periods in excess of five years.
Images
Drug Images
{{#ask: Page Name::Enflurane |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Enflurane |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Enflurane, as well as other general anesthetics, may cause a slight decrease in intellectual function for 2 or 3 days following anesthesia. As with other anesthetics, small changes in moods and symptoms may persist for several days following administration.
Precautions with Alcohol
Alcohol-Enflurane interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Enflurane Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Enflurane Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Enflurane |Label Name=Enflurane label.png
}}