Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alonso Alvarado, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS and POST TREATMENT ACUTE EXACERBATION OF HEPATITIS B
See full prescribing information for complete Boxed Warning.
LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS and POST TREATMENT ACUTE EXACERBATION OF HEPATITIS B: Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs, including tenofovir disoproxil fumarate, a component of STRIBILD, in combination with other antiretrovirals.
STRIBILD is not approved for the treatment of chronic hepatitis B virus (HBV) infection and the safety and efficacy of STRIBILD have not been established in patients coinfected with HBV and HIV-1. Severe acute exacerbations of hepatitis B have been reported in patients who are coinfected with HBV and human immunodeficiency virus-1 (HIV-1) and have discontinued EMTRIVA or VIREAD, which are components of STRIBILD. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who are coinfected with HIV-1 and HBV and discontinue STRIBILD. If appropriate, initiation of anti-hepatitis B therapy may be warranted
|
Overview
Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate is a atiretroviral that is FDA approved for the treatment of HIV-1 infection in adults who are antiretroviral treatment-naïve. There is a Black Box Warning for this drug as shown here. Common adverse reactions include a list of adverse reactions, separated by commas..
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Dosage Information
The recommended dosage of STRIBILD is one tablet taken orally once daily with food.
Dosage Adjustment in Patients with Renal Impairment
Initiation of STRIBILD in patients with estimated creatinine clearance below 70 mL per minute is not recommended. Because STRIBILD is a fixed-dose combination tablet, STRIBILD should be discontinued if estimated creatinine clearance declines below 50 mL per min during treatment with STRIBILD as dose interval adjustment required for emtricitabine and tenofovir disoproxil fumarate (DF) cannot be achieved.
Dosage in Patients with Hepatic Impairment
No dosage adjustment of STRIBILD is required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. No pharmacokinetic or safety data are available regarding the use of STRIBILD in patients with severe hepatic impairment (Child-Pugh Class C). Therefore, STRIBILD is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
Testing Prior to Initiation of STRIBILD
Prior to initiation of STRIBILD, patients should be tested for hepatitis B infection and estimated creatinine clearance, urine glucose and urine protein should be documented in all patients.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate in pediatric patients.
Contraindications
Coadministration of STRIBILD is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events. These drugs and other contraindicated drugs (which may lead to reduced efficacy of STRIBILD and possible resistance) are listed in Table 1
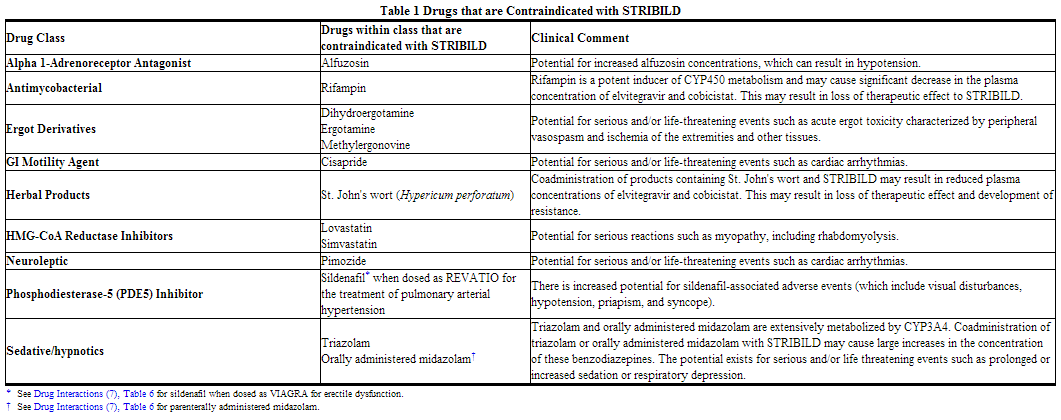
Warnings
|
WARNING: LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS and POST TREATMENT ACUTE EXACERBATION OF HEPATITIS B
See full prescribing information for complete Boxed Warning.
LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS and POST TREATMENT ACUTE EXACERBATION OF HEPATITIS B: Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs, including tenofovir disoproxil fumarate, a component of STRIBILD, in combination with other antiretrovirals.
STRIBILD is not approved for the treatment of chronic hepatitis B virus (HBV) infection and the safety and efficacy of STRIBILD have not been established in patients coinfected with HBV and HIV-1. Severe acute exacerbations of hepatitis B have been reported in patients who are coinfected with HBV and human immunodeficiency virus-1 (HIV-1) and have discontinued EMTRIVA or VIREAD, which are components of STRIBILD. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who are coinfected with HIV-1 and HBV and discontinue STRIBILD. If appropriate, initiation of anti-hepatitis B therapy may be warranted
|
Lactic Acidosis/Severe Hepatomegaly with Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs, including tenofovir DF, a component of STRIBILD, in combination with other antiretrovirals. A majority of these cases have been in women. Obesity and prolonged nucleoside exposure may be risk factors. Particular caution should be exercised when administering nucleoside analogs to any patient with known risk factors for liver disease; however, cases have also been reported in patients with no known risk factors. Treatment with STRIBILD should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
Patients Coinfected with HIV-1 and HBV
It is recommended that all patients with HIV-1 be tested for the presence of chronic hepatitis B virus (HBV) before initiating antiretroviral therapy. STRIBILD is not approved for the treatment of chronic HBV infection and the safety and efficacy of STRIBILD have not been established in patients coinfected with HBV and HIV-1. Severe acute exacerbations of hepatitis B have been reported in patients who are coinfected with HBV and HIV-1 and have discontinued emtricitabine or tenofovir DF, two of the components of STRIBILD. In some patients infected with HBV and treated with EMTRIVA, the exacerbations of hepatitis B were associated with liver decompensation and liver failure. Patients who are coinfected with HIV-1 and HBV should be closely monitored with both clinical and laboratory follow-up for at least several months after stopping treatment with STRIBILD. If appropriate, initiation of anti-hepatitis B therapy may be warranted.
New Onset or Worsening Renal Impairment
Renal impairment, including cases of acute renal failure and Fanconi syndrome (renal tubular injury with severe hypophosphatemia), has been reported with the use of tenofovir DF, a component of STRIBILD, and with the use of STRIBILD.
In the clinical trials of STRIBILD over 96 weeks, 10 (1.4%) subjects in the STRIBILD group (N=701) and 2 (0.3%) subjects in the combined comparator groups (N = 707) discontinued study drug due to a renal adverse reaction. Of these discontinuations, 8 in the STRIBILD group and 1 in the combined comparator groups occurred during the first 48 week. Four (0.6%) of the subjects who received STRIBILD developed laboratory findings consistent with proximal renal tubular dysfunction leading to discontinuation of STRIBILD compared to none in the comparator groups. Two of these four subjects had renal impairment (i.e. estimated creatinine clearance less than 70 mL per minute) at baseline. The laboratory findings in these 4 subjects with evidence of proximal tubulopathy improved but did not completely resolve in all subjects upon discontinuation of STRIBILD. Renal replacement therapy was not required for these subjects. Estimated creatinine clearance, urine glucose and urine protein should be documented in all patients prior to initiating therapy. Initiation of STRIBILD in patients with estimated creatinine clearance below 70 mL per minute is not recommended.
STRIBILD should be avoided with concurrent or recent use of a nephrotoxic agent (e.g., high-dose or multiple non-steroidal anti-inflammatory drugs (NSAIDs)). Cases of acute renal failure after initiation of high dose or multiple NSAIDs have been reported in HIV-infected patients with risk factors for renal dysfunction who appeared stable on tenofovir DF. Some patients required hospitalization and renal replacement therapy. Alternatives to NSAIDs should be considered, if needed, in patients at risk for renal dysfunction.
Persistent or worsening bone pain, pain in extremities, fractures and/or muscular pain or weakness may be manifestations of proximal renal tubulopathy and should prompt an evaluation of renal function in at-risk patients.
Routine monitoring of estimated creatinine clearance, urine glucose, and urine protein should be performed during STRIBILD therapy in all patients. Additionally, serum phosphorus should be measured in patients at risk for renal impairment. Although cobicistat (a component of STRIBILD) may cause modest increases in serum creatinine and modest declines in estimated creatinine clearance without affecting renal glomerular function, patients who experience a confirmed increase in serum creatinine of greater than 0.4 mg per dL from baseline should be closely monitored for renal safety.
The emtricitabine and tenofovir DF components of STRIBILD are primarily excreted by the kidney. STRIBILD should be discontinued if estimated creatinine clearance declines below 50 mL per minute as dose interval adjustment required for emtricitabine and tenofovir DF cannot be achieved with the fixed-dose combination tablet.
Avoid Use with Other Antiretroviral Products
STRIBILD is indicated for use as a complete regimen for the treatment of HIV-1 infection and coadministration with other antiretroviral products is not recommended.
STRIBILD is not recommended for coadministration with the following: emtricitabine or tenofovir DF (ATRIPLA, COMPLERA, EMTRIVA, TRUVADA, VIREAD); products containing lamivudine (COMBIVIR, EPIVIR, EPIVIR-HBV, EPZICOM, TRIZIVIR) or adefovir dipivoxil (HEPSERA); ritonavir (NORVIR, KALETRA).
Bone Effects of Tenofovir DF
Bone Mineral Density
In clinical trials in HIV-1 infected adults, tenofovir DF (a component of STRIBILD) was associated with slightly greater decreases in bone mineral density (BMD) and increases in biochemical markers of bone metabolism, suggesting increased bone turnover relative to comparators. Serum parathyroid hormone levels and 1.25 Vitamin D levels were also higher in subjects receiving tenofovir DF. For additional information, see Adverse Reactions (6.1) and consult the VIREAD prescribing information.
The effects of tenofovir DF-associated changes in BMD and biochemical markers on long-term bone health and future fracture risk are unknown. Assessment of BMD should be considered for HIV-1 infected patients who have a history of pathologic bone fracture or other risk factors for osteoporosis or bone loss. Although the effect of supplementation with calcium and vitamin D was not studied, such supplementation may be beneficial in all patients. If bone abnormalities are suspected, then appropriate consultation should be obtained.
Mineralization Defects
Cases of osteomalacia associated with proximal renal tubulopathy, manifested as bone pain or pain in extremities and which may contribute to fractures, have been reported in association with the use of tenofovir DF. Arthralgias and muscle pain or weakness have also been reported in cases of proximal renal tubulopathy. Hypophosphatemia and osteomalacia secondary to proximal renal tubulopathy should be considered in patients at risk of renal dysfunction who present with persistent or worsening bone or muscle symptoms while receiving products containing tenofovir DF.
Fat Redistribution
Redistribution/accumulation of body fat including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including STRIBILD. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections [such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia (PCP), or tuberculosis], which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution, however, the time to onset is more variable, and can occur many months after initiation of treatment.
Adverse Reactions
Clinical Trials Experience
The following adverse drug reactions are discussed in other sections of the labeling:
- Lactic Acidosis/Severe Hepatomegaly with Steatosis.
- Severe Acute Exacerbations of Hepatitis B.
- New Onset or Worsening Renal Impairment.
- Bone Effects of Tenofovir DF.
- Immune Reconstitution Syndrome.
Adverse Reactions from Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The safety assessment of STRIBILD is based on the Week 96 pooled data from 1408 subjects in two randomized, double-blind, active-controlled clinical trials, Study 102 and Study 103, in antiretroviral treatment-naïve HIV-1 infected adult subjects [see Clinical Studies (14)]. A total of 701 subjects received STRIBILD once daily for at least 96 weeks.
The proportion of subjects who discontinued treatment with STRIBILD (elvitegravir 150 mg/cobicistat 150 mg/emtricitabine 200 mg/tenofovir DF 300 mg); ATRIPLA (efavirenz 600 mg/emtricitabine 200 mg/tenofovir DF 300 mg); or atazanavir (ATV) + ritonavir (RTV) + TRUVADA (emtricitabine 200 mg/tenofovir DF 300 mg) due to adverse events, regardless of severity, was 4.6%, 6.8% and 5.9%, respectively. Table 2 displays the frequency of adverse drug reactions greater than or equal to 5% of subjects in any treatment arm.

See Warnings and Precautions (5.3), for a discussion of renal adverse reactions from clinical trials experience with STRIBILD.
Adverse Reactions from Clinical Trials of the Components of STRIBILD
Emtricitabine and Tenofovir Disoproxil Fumarate: In addition to the adverse reactions observed with STRIBILD, the following adverse reactions occurred in at least 5% of treatment-experienced or treatment-naïve subjects receiving emtricitabine or tenofovir DF with other antiretroviral agents in other clinical trials: depression, abdominal pain, dyspepsia, vomiting, fever, pain, nasopharyngitis, pneumonia, sinusitis, upper respiratory tract infection, arthralgia, back pain, myalgia, paresthesia, peripheral neuropathy (including peripheral neuritis and neuropathy), anxiety, increased cough, and rhinitis.
Skin discoloration has been reported with higher frequency among emtricitabine-treated subjects; it was manifested by hyperpigmentation on the palms and/or soles and was generally mild and asymptomatic. The mechanism and clinical significance are unknown.
Laboratory Abnormalities
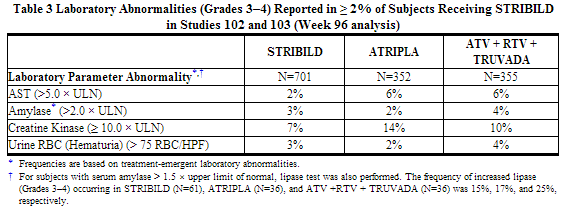
The frequency of laboratory abnormalities (Grades 3–4) occurring in at least 2% of subjects receiving STRIBILD in Studies 102 and 103 are presented in Table 3.
In Study 103, BMD was assessed by DEXA in a non-random subset of 120 subjects. Mean percentage decreases in BMD from baseline to Week 96 in the STRIBILD group (N = 47) were comparable to the ATV + RTV + TRUVADA group (N = 53) at the lumbar spine (-2.0% versus -3.5%, respectively) and at the hip (-3.2% versus -4.2%, respectively). In Studies 102 and 103, bone fractures occurred in 14 subjects (2.0%) in the STRIBILD group, 8 subjects (2.3%) in the ATRIPLA group, and 14 subjects (3.9%) in the ATV + RTV + TRUVADA group. These findings were consistent with data from an earlier 144-week trial of treatment-naïve subjects receiving tenofovir DF + lamivudine + efavirenz.
Proteinuria (all grades) occurred in 46% of subjects receiving STRIBILD, 38% of subjects receiving ATRIPLA, and 37% of subjects receiving ATV + RTV + TRUVADA.
The cobicistat component of STRIBILD has been shown to increase serum creatinine and decrease estimated creatinine clearance due to inhibition of tubular secretion of creatinine without affecting renal glomerular function. In Studies 102 and 103, increases in serum creatinine and decreases in estimated creatinine clearance occurred early in treatment with STRIBILD, after which they stabilized. Table 4 displays the mean changes in serum creatinine and eGFR levels at Week 96 and the percentage of subjects with elevations in serum creatinine (All Grades).

Emtricitabine or Tenofovir DF: In addition to the laboratory abnormalities observed with STRIBILD, the following laboratory abnormalities have been previously reported in subjects treated with emtricitabine or tenofovir DF with other antiretroviral agents in other clinical trials: Grade 3 or 4 laboratory abnormalities of ALT (M: greater than 215 U per L; F: greater than 170 U per L), alkaline phosphatase (greater than 550 U per L), bilirubin (greater than 2.5 × ULN), serum glucose (less than 40 or greater than 250 mg per dL), glycosuria (greater than or equal to 3+), neutrophils (less than 750 per mm3), fasting cholesterol (greater than 240 mg per dL), and fasting triglycerides (greater than 750 mg per dL).
Serum Lipids: In the clinical trials of STRIBILD, a similar percentage of subjects receiving STRIBILD, ATRIPLA, and ATV + RTV + TRUVADA were on lipid lowering agents at baseline (11%, 11%, and 12%, respectively). While receiving study drug through Week 96, an additional 8% of STRIBILD subjects were started on lipid lowering agents, compared to 9% of ATRIPLA and 8% of ATV + RTV + TRUVADA subjects.
Changes from baseline in total cholesterol, HDL-cholesterol, LDL-cholesterol, and triglycerides are presented in Table 5.

Postmarketing Experience
Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following adverse reactions have been identified during post approval use of tenofovir DF. No additional postmarketing adverse reactions specific for emtricitabine have been identified.
- Immune System Disorders: allergic reaction, including angioedema
- Metabolism and Nutrition Disorders: lactic acidosis, hypokalemia, hypophosphatemia
- Respiratory, Thoracic, and Mediastinal Disorders: dyspnea
- Gastrointestinal Disorders: pancreatitis, increased amylase, abdominal pain
- Hepatobiliary Disorders: hepatic steatosis, hepatitis, increased liver enzymes (most commonly AST, ALT gamma GT)
- Skin and Subcutaneous Tissue Disorders: rash
- Musculoskeletal and Connective Tissue Disorders: rhabdomyolysis, osteomalacia (manifested as bone pain and which may contribute to fractures), muscular weakness, myopathy
- Renal and Urinary Disorders: acute renal failure, renal failure, acute tubular necrosis, Fanconi syndrome, proximal renal tubulopathy, interstitial nephritis (including acute cases), nephrogenic diabetes insipidus, renal insufficiency, increased creatinine, proteinuria, polyuria
- General Disorders and Administration Site Conditions: asthenia
The following adverse reactions, listed under the body system headings above, may occur as a consequence of proximal renal tubulopathy: rhabdomyolysis, osteomalacia, hypokalemia, muscular weakness, myopathy, hypophosphatemia.
Drug Interactions
Other Antiretroviral Medications
STRIBILD is a complete regimen for the treatment of HIV-1 infection; therefore, STRIBILD should not be administered with other antiretroviral medications for treatment of HIV-1 infection. Complete information regarding potential drug-drug interactions with other antiretroviral medications is not provided.
Potential for STRIBILD to Affect Other Drugs
Cobicistat, a component of STRIBILD, is an inhibitor of CYP3A and CYP2D6 and an inhibitor of the following transporters: p-glycoprotein (P-gp), BCRP, OATP1B1 and OATP1B3. Thus, coadministration of STRIBILD with drugs that are primarily metabolized by CYP3A or CYP2D6, or are substrates of P-gp, BCRP, OATP1B1 or OATP1B3 may result in increased plasma concentrations of such drugs. Elvitegravir is a modest inducer of CYP2C9 and may decrease the plasma concentrations of CYP2C9 substrates.
Potential for Other Drugs to Affect One or More Components of STRIBILD
Elvitegravir and cobicistat, components of STRIBILD, are metabolized by CYP3A. Cobicistat is also metabolized, to a minor extent, by CYP2D6. Drugs that induce CYP3A activity are expected to increase the clearance of elvitegravir and cobicistat, resulting in decreased plasma concentration of cobicistat and elvitegravir, which may lead to loss of therapeutic effect of STRIBILD and development of resistance (see Table 6). Coadministration of STRIBILD with other drugs that inhibit CYP3A may decrease the clearance and increase the plasma concentration of cobicistat (see Table 6).
Drugs Affecting Renal Function
Because emtricitabine and tenofovir, components of STRIBILD are primarily excreted by the kidneys by a combination of glomerular filtration and active tubular secretion, coadministration of STRIBILD with drugs that reduce renal function or compete for active tubular secretion may increase concentrations of emtricitabine, tenofovir, and other renally eliminated drugs and this may increase the risk of adverse reactions. Some examples of drugs that are eliminated by active tubular secretion include, but are not limited to acyclovir, cidofovir, ganciclovir, valacyclovir, valganciclovir, aminoglycosides (e.g. gentamicin), and high-dose or multiple NSAIDs.
Established and Other Potentially Significant Interactions
Table 6 provides a listing of established or potentially clinically significant drug interactions. The drug interactions described are based on studies conducted with either STRIBILD, the components of STRIBILD, (elvitegravir, cobicistat, emtricitabine, and tenofovir DF) as individual agents and/or in combination, or are predicted drug interactions that may occur with STRIBILD. The table includes potentially significant interactions but is not all inclusive.
Drugs without Clinically Significant Interactions with STRIBILD
Based on drug interaction studies conducted with the components of STRIBILD, no clinically significant drug interactions have been either observed or are expected when STRIBILD is combined with the following drugs: entecavir, famciclovir, H2 receptor antagonists, methadone, proton pump inhibitors and ribavirin.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): B There are no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, STRIBILD should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Antiretroviral Pregnancy Registry: To monitor fetal outcomes of pregnant women exposed to STRIBILD, an Antiretroviral Pregnancy Registry has been established. Healthcare providers are encouraged to register patients by calling 1-800-258-4263.
Animal Data
Elvitegravir: Studies in animals have shown no evidence of teratogenicity or an effect on reproductive function. In offspring from rat and rabbit dams treated with elvitegravir during pregnancy, there were no toxicologically significant effects on developmental endpoints. The exposures (AUC) at the embryo-fetal No Observed Adverse Effects Levels (NOAELs) in rats and rabbits were respectively 23 and 0.2 times higher than the exposure in humans at the recommended daily dose of 150 mg.
Cobicistat: Studies in animals have shown no evidence of teratogenicity or an effect on reproductive function. In offspring from rat and rabbit dams treated with cobicistat during pregnancy, there were no toxicologically significant effects on developmental endpoints. The exposures (AUC) at the embryo-fetal NOAELs in rats and rabbits were respectively 1.8 and 4.3 times higher than the exposure in humans at the recommended daily dose of 150 mg.
Emtricitabine: The incidence of fetal variations and malformations was not increased in embryo-fetal toxicity studies performed with emtricitabine in mice at exposures (AUC) approximately 60 times higher and in rabbits at approximately 120 times higher than human exposures at the recommended daily dose.
Tenofovir DF: Reproduction studies have been performed in rats and rabbits at doses up to 14 and 19 times the human dose based on body surface area comparisons and revealed no evidence of impaired fertility or harm to the fetus due to tenofovir.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate during labor and delivery.
Nursing Mothers
The Centers for Disease Control and Prevention recommend that HIV-infected mothers not breastfeed their infants to avoid risking postnatal transmission of HIV. Studies in rats have demonstrated that elvitegravir, cobicistat, and tenofovir are secreted in milk. It is not known whether elvitegravir or cobicistat is excreted in human milk.
In humans, samples of breast milk obtained from five HIV-1 infected mothers show that emtricitabine is secreted in human milk. Breastfeeding infants whose mothers are being treated with emtricitabine may be at risk for developing viral resistance to emtricitabine. Other emtricitabine-associated risks in infants breastfed by mothers being treated with emtricitabine are unknown.
Samples of breast milk obtained from five HIV-1 infected mothers show that tenofovir is secreted in human milk. Tenofovir-associated risks, including the risk of viral resistance to tenofovir, in infants breastfed by mothers being treated with tenofovir disoproxil fumarate are unknown. Because of both the potential for HIV transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breastfeed if they are receiving STRIBILD.
Pediatric Use
Safety and effectiveness of STRIBILD in pediatric patients less than 18 years of age have not been established
Geriatic Use
Clinical studies of STRIBILD did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, caution should be exercised in the administration of STRIBILD in elderly patients, keeping in mind the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Gender
There is no FDA guidance on the use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate with respect to specific gender populations.
Race
There is no FDA guidance on the use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate with respect to specific racial populations.
Renal Impairment
Initiation of STRIBILD in patients with estimated creatinine clearance below 70 mL per min is not recommended. Because STRIBILD is a fixed-dose combination tablet, STRIBILD should be discontinued if estimated creatinine clearance declines below 50 mL per min during treatment with STRIBILD as dose interval adjustment required for emtricitabine and tenofovir DF cannot be achieved
Hepatic Impairment
No dose adjustment of STRIBILD is required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. No pharmacokinetic or safety data are available regarding the use of STRIBILD in patients with severe hepatic impairment (Child-Pugh Class C). Therefore, STRIBILD is not recommended for use in patients with severe hepatic impairment
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate in patients who are immunocompromised.
Administration and Monitoring
Administration
Oral
Monitoring
Condition 1
(Description regarding monitoring, from Warnings section)
STRIBILD is not approved for the treatment of chronic hepatitis B virus (HBV) infection. Severe acute exacerbations of hepatitis B have been reported in patients coinfected with HIV-1 and HBV who have discontinued EMTRIVA or VIREAD, two of the components of STRIBILD. Hepatic function should be monitored closely in these patients. If appropriate, initiation of anti-hepatitis B therapy may be warranted.
IV Compatibility
There is limited information regarding the compatibility of Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate and IV administrations.
Overdosage
No data are available on overdose of STRIBILD in patients. If overdose occurs the patient must be monitored for evidence of toxicity. Treatment of overdose with STRIBILD consists of general supportive measures including monitoring of vital signs as well as observation of the clinical status of the patient.
Elvitegravir: Limited clinical experience is available at doses higher than the therapeutic dose of elvitegravir. In one study, boosted elvitegravir equivalent to 2 times the therapeutic dose of 150 mg once daily for 10 days was administered to 42 healthy subjects. No severe adverse reactions were reported. The effects of higher doses are not known. As elvitegravir is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis.
Cobicistat: Limited clinical experience is available at doses higher than the therapeutic dose of cobicistat. In two studies, a single dose of cobicistat 400 mg (2.7 times the dose in STRIBILD) was administered to a total of 60 healthy subjects. No severe adverse reactions were reported. The effects of higher doses are not known. As cobicistat is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis.
Emtricitabine: Limited clinical experience is available at doses higher than the therapeutic dose of EMTRIVA. In one clinical pharmacology study, single doses of emtricitabine 1200 mg (6 times the dose in STRIBILD) were administered to 11 subjects. No severe adverse reactions were reported. The effects of higher doses are not known. Hemodialysis treatment removes approximately 30% of the emtricitabine dose over a 3 hour dialysis period starting within 1.5 hours of emtricitabine dosing (blood flow rate of 400 mL per minute and a dialysate flow rate of 600 mL per minute). It is not known whether emtricitabine can be removed by peritoneal dialysis.
Tenofovir DF: Limited clinical experience at doses higher than the therapeutic dose of VIREAD 300 mg is available. In one study, 600 mg tenofovir DF (2 times the dosage in STRIBILD) was administered to 8 subjects orally for 28 days, and no severe adverse reactions were reported. The effects of higher doses are not known. Tenofovir is efficiently removed by hemodialysis with an extraction coefficient of approximately 54%. Following a single 300 mg dose of VIREAD, a 4-hour hemodialysis session removed approximately 10% of the administered tenofovir dose.
Pharmacology
Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
(Description)
Structure
(Description with picture)
Pharmacodynamics
(Description)
Pharmacokinetics
(Description)
Nonclinical Toxicology
(Description)
Clinical Studies
Condition 1
(Description)
Condition 2
(Description)
Condition 3
(Description)
How Supplied
(Description)
Storage
There is limited information regarding Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
(Patient Counseling Information)
Precautions with Alcohol
Alcohol-Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Elvitegravir, Cobicistat, Emtricitabine, And Tenofovir Disoproxil Fumarate Brand Names in the drug label.
Look-Alike Drug Names
- (Paired Confused Name 1a) — (Paired Confused Name 1b)
- (Paired Confused Name 2a) — (Paired Confused Name 2b)
- (Paired Confused Name 3a) — (Paired Confused Name 3b)
Drug Shortage Status
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.