Eliglustat: Difference between revisions
Kiran Singh (talk | contribs) No edit summary |
Kiran Singh (talk | contribs) No edit summary |
||
| Line 143: | Line 143: | ||
[[File: | [[ File:Eliglustat table5.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
|useInPregnancyAUS=* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | |useInPregnancyAUS=* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | ||
| Line 153: | Line 149: | ||
|useInLaborDelivery=There is no FDA guidance on use of {{PAGENAME}} during labor and delivery. | |useInLaborDelivery=There is no FDA guidance on use of {{PAGENAME}} during labor and delivery. | ||
|useInNursing=There is no FDA guidance on the use of {{PAGENAME}} with respect to nursing mothers. | |useInNursing=There is no FDA guidance on the use of {{PAGENAME}} with respect to nursing mothers. | ||
|useInPed= | |useInPed=* Safety and effectiveness in pediatric patients have not been established. | ||
|useInGeri=There is no FDA guidance on the use of {{PAGENAME}} with respect to geriatric patients. | |useInGeri=There is no FDA guidance on the use of {{PAGENAME}} with respect to geriatric patients. | ||
|useInGender=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific gender populations. | |useInGender=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific gender populations. | ||
|useInRace=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific racial populations. | |useInRace=There is no FDA guidance on the use of {{PAGENAME}} with respect to specific racial populations. | ||
|useInRenalImpair=There is no | |useInRenalImpair=* There is no dosage adjustment required for patients with mild renal impairment. CERDELGA has not been studied in patients with moderate to severe renal impairment or end-stage renal disease (ESRD). Use of CERDELGA in these patients is not recommended. | ||
|useInHepaticImpair= | |useInHepaticImpair=* CERDELGA has not been studied in patients with hepatic impairment. Use of CERDELGA is not recommended in all stages of hepatic impairment or cirrhosis. | ||
|useInReproPotential=There is no FDA guidance on the use of {{PAGENAME}} in women of reproductive potentials and males. | |useInReproPotential=There is no FDA guidance on the use of {{PAGENAME}} in women of reproductive potentials and males. | ||
|useInImmunocomp=There is no FDA guidance one the use of {{PAGENAME}} in patients who are immunocompromised. | |useInImmunocomp=There is no FDA guidance one the use of {{PAGENAME}} in patients who are immunocompromised. | ||
| Line 170: | Line 166: | ||
<!--IV Compatibility--> | <!--IV Compatibility--> | ||
|IVCompat=There is limited information regarding <i>IV Compatibility</i> of {{PAGENAME}} in the drug label. | |IVCompat=There is limited information regarding <i>IV Compatibility</i> of {{PAGENAME}} in the drug label. | ||
|overdose= | |overdose=* The highest eliglustat plasma concentration experienced to date occurred in a single-dose, dose escalation study in healthy subjects, in a subject taking a dose equivalent to approximately 21 times the recommended dose for GD1 patients. At the time of the highest plasma concentration (59-fold higher than normal therapeutic conditions), the subject experienced dizziness marked by disequilibrium, hypotension, bradycardia, nausea, and vomiting. | ||
* In the event of acute overdose, the patient should be carefully observed and given symptomatic and supportive treatment. | |||
* Hemodialysis is unlikely to be beneficial given that eliglustat has a large volume of distribution | |||
|PD=There is limited information regarding <i>Pharmacodynamics</i> of {{PAGENAME}} in the drug label. | |PD=There is limited information regarding <i>Pharmacodynamics</i> of {{PAGENAME}} in the drug label. | ||
|PK=There is limited information regarding <i>Pharmacokinetics</i> of {{PAGENAME}} in the drug label. | |PK=There is limited information regarding <i>Pharmacokinetics</i> of {{PAGENAME}} in the drug label. | ||
Revision as of 17:38, 18 February 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Eliglustat is a {{{drugClass}}} that is FDA approved for the treatment of {{{indication}}}. Common adverse reactions include .
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- CERDELGA is indicated for the long-term treatment of adult patients with Gaucher disease type 1 (GD1) who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) as detected by an FDA-cleared test.
Limitations of Use:
- Patients who are CYP2D6 ultra-rapid metabolizers (URMs) may not achieve adequate concentrations of CERDELGA to achieve a therapeutic effect .
- A specific dosage cannot be recommended for those patients whose CYP2D6 genotype cannot be determined (indeterminate metabolizers).
Dosage
Patient Selection
- Select patients with Gaucher disease type 1 based on their CYP2D6 metabolizer status. It is recommended patient genotypes be established using an FDA-cleared test for determining CYP2D6 genotype.
Recommended Adult Dosage
- The recommended dosage of CERDELGA is 84 mg twice daily in CYP2D6 EMs and IMs. The recommended dosage in CYP2D6 PMs is 84 mg once daily; appropriate adverse event monitoring is recommended. The predicted exposures with 84 mg once daily in patients who are CYP2D6 PMs are expected to be similar to exposures observed with 84 mg twice daily in CYP2D6 IMs.
- Some inhibitors of CYP2D6 and CYP3A are contraindicated with CERDELGA depending on the patient's metabolizer status [see CONTRAINDICATIONS (4)]. Co-administration of CERDELGA with other CYP2D6 and CYP3A inhibitors may require dosage adjustment depending on the patient's CYP2D6 metabolizer status to reduce the risk of potentially significant adverse reactions.
- Reduce the dosage of CERDELGA to 84 mg once daily for:
- CYP2D6 EMs and IMs taking strong or moderate CYP2D6 inhibitors
- CYP2D6 EMs taking strong or moderate CYP3A inhibitors
DOSAGE FORMS AND STRENGTHS
- CERDELGA is supplied as 84 mg hard gelatin capsules, with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black. Each capsule contains 100 mg eliglustat tartrate, which is equivalent to 84 mg of eliglustat.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Eliglustat in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Eliglustat in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Eliglustat in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Eliglustat in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Eliglustat in pediatric patients.
Contraindications
- CERDELGA is contraindicated in the following patients due to the risk of significantly increased eliglustat plasma concentrations which may result in prolongation of the PR, QTc, and/or QRS cardiac intervals that could result in cardiac arrhythmias. See TABLE 3 and TABLE 4 for examples of drugs in each of the categories described:
EMs or IMs taking a strong or moderate CYP2D6 inhibitor concomitantly with a strong or moderate CYP3A inhibitor. IMs or PMs taking a strong CYP3A inhibitor.
Warnings
Drug-Drug Interactions
- Eliglustat is a CYP2D6 and CYP3A substrate. Drugs that inhibit CYP2D6 and CYP3A metabolism pathways may significantly increase the exposure to eliglustat and result in prolongation of the PR, QTc, and/or QRS cardiac intervals that could result in cardiac arrhythmias. Some drugs that are inhibitors of CYP2D6 and CYP3A are contraindicated with CERDELGA depending on the patient's CYP2D6 metabolizer status . See TABLE 3 and TABLE 4 for other potentially significant drug interactions.
ECG Changes and Potential for Cardiac Arrhythmias
- Use of CERDELGA in patients with pre-existing cardiac conditions has not been studied during clinical trials. Because CERDELGA is predicted to cause increases in ECG intervals (PR, QTc, and QRS) at substantially elevated eliglustat plasma concentrations, use of CERDELGA is not recommended in patients with pre-existing cardiac disease (congestive heart failure, recent acute myocardial infarction, bradycardia, heart block, ventricular arrhythmia), long QT syndrome, and in combination with Class IA (e.g., quinidine, procainamide) and Class III (e.g., amiodarone, sotalol) antiarrhythmic medications
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- The most common adverse reactions to CERDELGA (occurring in ≥10% of the 126 GD1 patients treated with CERDELGA across Trials 1 and 2) were fatigue, headache, nausea, diarrhea, back pain, pain in extremities, and upper abdominal pain.
The adverse reaction profile of CERDELGA is based on two controlled studies, Trials 1 and 2. Table 1 presents the profile from the 9-month double-blind, randomized, placebo-controlled trial of 40 treatment-naïve patients (Trial 1). Patients were between the ages of 16 and 63 on the date of the first dose of study drug, and included 20 males and 20 females.
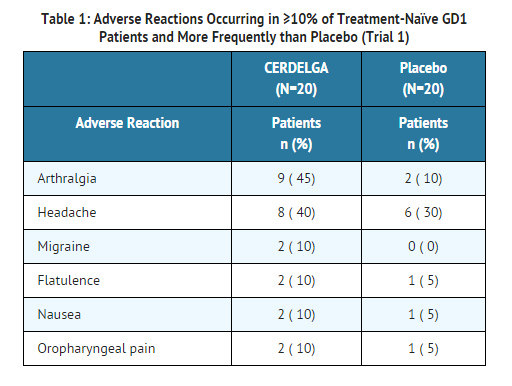
- Table 2 presents the profile from the 12-month open-label, randomized, imiglucerase-controlled trial of 159 treated patients switching from enzyme replacement therapy (ERT) (Trial 2). Patients were between the ages of 18 and 69 on the date of the first dose of CERDELGA, and included 87 females and 72 males.
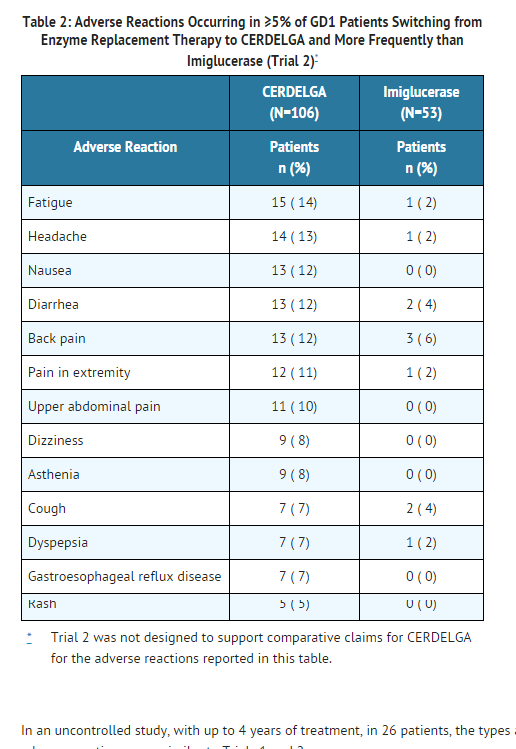
In an uncontrolled study, with up to 4 years of treatment, in 26 patients, the types and incidences of adverse reactions were similar to Trials 1 and 2.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Eliglustat in the drug label.
Cardiovascular
Digestive
Endocrine
Hematologic and Lymphatic
Metabolic and Nutritional
Musculoskeletal
Neurologic
Respiratory
Skin and Hypersensitivy Reactions
Special Senses
Urogenital
Miscellaneous
Drug Interactions
Potential for Other Drugs to Affect CERDELGA
Eliglustat is a CYP2D6 and CYP3A substrate.
CYP2D6 and CYP3A Inhibitors
- Drugs that inhibit CYP2D6 and CYP3A pathways may significantly increase the exposure to eliglustat and result in prolongation of the PR, QTc, and/or QRS cardiac interval which could result in cardiac arrhythmias:
- Some inhibitors of CYP2D6 and CYP3A are contraindicated with CERDELGA depending on the patient's CYP2D6 metabolizer status.
- Co-administration of CERDELGA with other CYP2D6 and CYP3A inhibitors may require dosage adjustment depending on the patient's CYP2D6 metabolizer status to reduce the risk of potential significant adverse reactions.
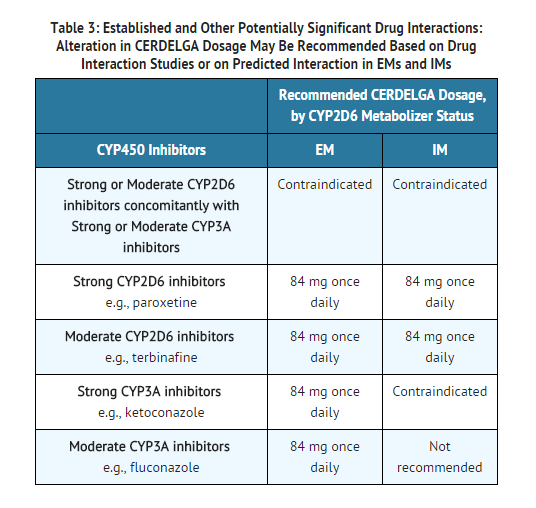
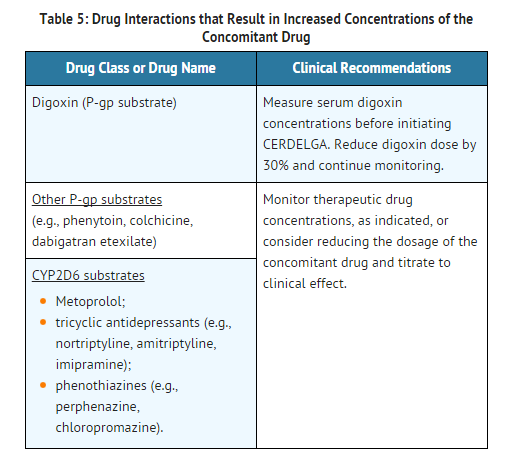
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Eliglustat in women who are pregnant.
Pregnancy Category (AUS):
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Eliglustat in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Eliglustat during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Eliglustat with respect to nursing mothers.
Pediatric Use
- Safety and effectiveness in pediatric patients have not been established.
Geriatic Use
There is no FDA guidance on the use of Eliglustat with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Eliglustat with respect to specific gender populations.
Race
There is no FDA guidance on the use of Eliglustat with respect to specific racial populations.
Renal Impairment
- There is no dosage adjustment required for patients with mild renal impairment. CERDELGA has not been studied in patients with moderate to severe renal impairment or end-stage renal disease (ESRD). Use of CERDELGA in these patients is not recommended.
Hepatic Impairment
- CERDELGA has not been studied in patients with hepatic impairment. Use of CERDELGA is not recommended in all stages of hepatic impairment or cirrhosis.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Eliglustat in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Eliglustat in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- Intravenous
Monitoring
There is limited information regarding Monitoring of Eliglustat in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Eliglustat in the drug label.
Overdosage
- The highest eliglustat plasma concentration experienced to date occurred in a single-dose, dose escalation study in healthy subjects, in a subject taking a dose equivalent to approximately 21 times the recommended dose for GD1 patients. At the time of the highest plasma concentration (59-fold higher than normal therapeutic conditions), the subject experienced dizziness marked by disequilibrium, hypotension, bradycardia, nausea, and vomiting.
- In the event of acute overdose, the patient should be carefully observed and given symptomatic and supportive treatment.
- Hemodialysis is unlikely to be beneficial given that eliglustat has a large volume of distribution
Pharmacology
There is limited information regarding Eliglustat Pharmacology in the drug label.
Mechanism of Action
There is limited information regarding Eliglustat Mechanism of Action in the drug label.
Structure
There is limited information regarding Eliglustat Structure in the drug label.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Eliglustat in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Eliglustat in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Eliglustat in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Eliglustat in the drug label.
How Supplied
Storage
There is limited information regarding Eliglustat Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Eliglustat |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Eliglustat |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Eliglustat in the drug label.
Precautions with Alcohol
- Alcohol-Eliglustat interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Look-Alike Drug Names
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Eliglustat
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Eliglustat |Label Name=Eliglustat11.png
}}
{{#subobject:
|Label Page=Eliglustat |Label Name=Eliglustat11.png
}}