Doxazosin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: João André Alves Silva, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Doxazosin is a alpha-adrenergic blocker that is FDA approved for the {{{indicationType}}} of benign prostatic hyperplasia (BPH) and hypertension. Common adverse reactions include edema, hypotension, nausea, dizziness, headache, somnolence, vertigo and fatigue.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
BENIGN PROSTATIC HYPERPLASIA
- Initial dose:
- 1 mg once a day
- Depending on the patient's urodynamics and BPH symptomatology, dosage may be increased, with a recommended titration interval of 1-2 weeks, to:
- 2 mg once a day, followed by 4 mg once a day until the maximum dosage of 8 mg once a day.
HYPERTENSION
- Initial dose:
- 1 mg once a day
- Depending on the patient's standing blood pressure response (based on measurements taken at 2–6 hours post-dose and 24 hours post-dose), dosage may then be increased to:
- 2 mg once a day, followed by 4 mg once a day, than 8 mg once a day until the maximum dosage of 16 mg once a day to achieve the desired reduction in blood pressure.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Doxazonin sandbox in adult patients.
Non–Guideline-Supported Use
Cardiac syndrome X
Disorder of the urinary system
- Dosing Information
- 2 mg at bedtime, twice daily
Erectile dysfunction; Diagnosis
- Dosing Information
- 4 mg daily
Pheochromocytoma
- Dosing Information
- 2—16 mg daily
Ureteric stone
- Dosing Information
- 4 mg daily
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Hypertension
- Dosing Information
- Initial: 1 mg/day PO until the maximum dosage of 4 mg/day
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Doxazosin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Doxazosin in pediatric patients.
Contraindications
- Doxazosin tablets are contraindicated in patients with a known sensitivity to quinazolines (e.g., prazosin, terazosin), doxazosin, or any of the inert ingredients.
Warnings
Syncope and "First-dose" Effect
- Doxazosin, like other alpha-adrenergic blocking agents, can cause marked hypotension, especially in the upright position, with syncope and other postural symptoms such as dizziness. Marked orthostatic effects are most common with the first dose but can also occur when there is a dosage increase, or if therapy is interrupted for more than a few days. To decrease the likelihood of excessive hypotension and syncope, it is essential that treatment be initiated with the 1 mg dose. The 2 mg, 4 mg, and 8 mg tablets are not for initial therapy. Dosage should then be adjusted slowly with evaluations and increases in dose every two weeks to the recommended dose. Additional antihypertensive agents should be added with caution.
- Patients being titrated with doxazosin should be cautioned to avoid situations where injury could result should syncope occur, during both the day and night.
- If syncope occurs, the patient should be placed in a recumbent position and treated supportively as necessary.
Priapism
- Rarely (probably less frequently than once in every several thousand patients), alpha1 antagonists, including doxazosin, have been associated with priapism (painful penile erection, sustained for hours and unrelieved by sexual intercourse or masturbation). Because this condition can lead to permanent impotence if not promptly treated, patients must be advised about the seriousness of the condition.
Adverse Reactions
Clinical Trials Experience
Benign Prostatic Hyperplasia (BPH)
The incidence of adverse events has been ascertained from worldwide clinical trials in 965 BPH patients. The incidence rates presented below (Table 3) are based on combined data from seven placebo-controlled trials involving once daily administration of doxazosin in doses of 1 mg to 16 mg in hypertensives and 0.5 mg to 8 mg in normotensives. The adverse events when the incidence in the doxazosin group was at least 1% are summarized in Table 3. No significant difference in the incidence of adverse events compared to placebo was seen except for dizziness, fatigue, hypotension, edema and dyspnea. Dizziness and dyspnea appeared to be dose-related.
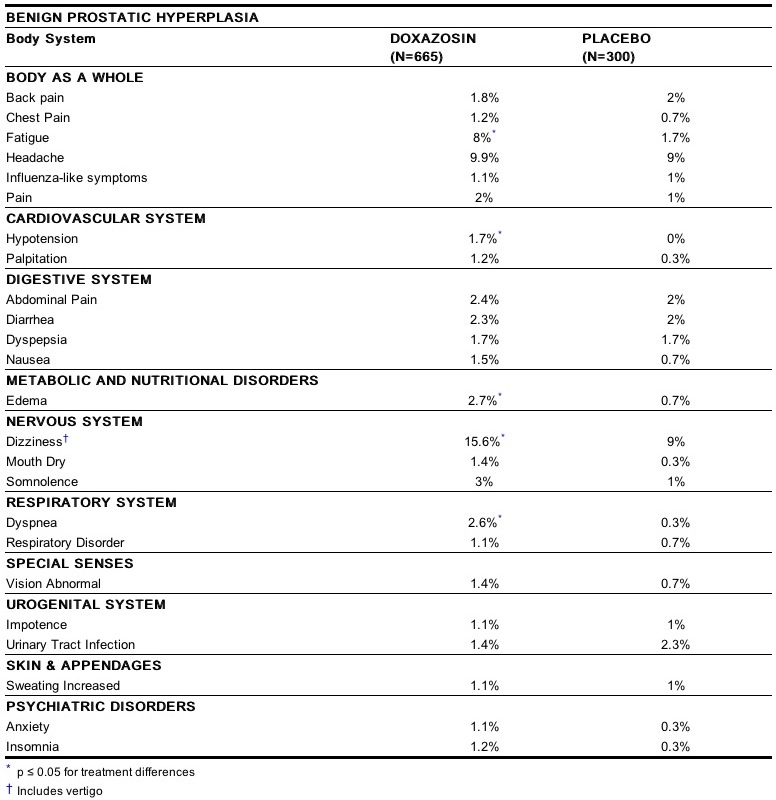
In these placebo-controlled studies of 665 doxazosin patients, treated for a mean of 85 days, additional adverse reactions have been reported. These are less than 1% and not distinguishable from those that occurred in the placebo group. Adverse reactions with an incidence of less than 1% but of clinical interest are (doxazosin vs. placebo): Cardiovascular System: angina pectoris (0.6% vs. 0.7%), postural hypotension (0.3% vs. 0.3%), syncope (0.5% vs. 0%), tachycardia (0.9% vs. 0%); Urogenital System: dysuria (0.5% vs. 1.3%), and Psychiatric Disorders: libido decreased (0.8% vs. 0.3%). The safety profile in patients treated for up to three years was similar to that in the placebo-controlled studies. The majority of adverse experiences with doxazosin were mild.
Hypertension
Doxazosin mesylate has been administered to approximately 4,000 hypertensive patients, of whom 1,679 were included in the hypertension clinical development program. In that program, minor adverse effects were frequent, but led to discontinuation of treatment in only 7% of patients. In placebo-controlled studies adverse effects occurred in 49% and 40% of patients in the doxazosin and placebo groups, respectively, and led to discontinuation in 2% of patients in each group. The major reasons for discontinuation were postural effects (2%), edema, malaise/fatigue, and some heart rate disturbance, each about 0.7%. In controlled hypertension clinical trials directly comparing doxazosin to placebo there was no significant difference in the incidence of side effects, except for dizziness (including postural), weight gain, somnolence and fatigue/malaise. Postural effects and edema appeared to be dose related. The prevalence rates presented below are based on combined data from placebo-controlled studies involving once daily administration of doxazosin at doses ranging from 1 mg to 16 mg. Table 4 summarizes those adverse experiences (possibly/probably related) reported for patients in these hypertension studies where the prevalence rate in the doxazosin group was at least 0.5% or where the reaction is of particular interest.
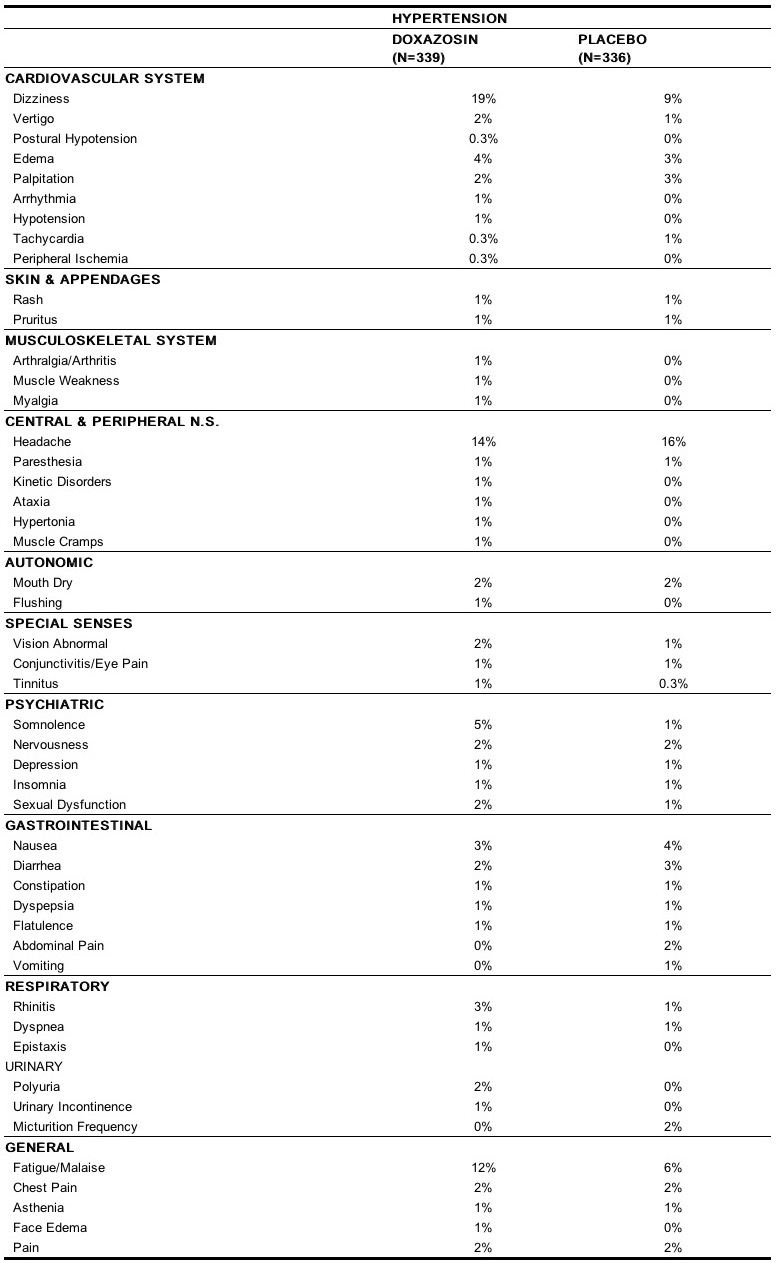
Additional adverse reactions have been reported, but these are, in general, not distinguishable from symptoms that might have occurred in the absence of exposure to doxazosin. The following adverse reactions occurred with a frequency of between 0.5% and 1%: syncope, hypoesthesia, increased sweating, agitation, increased weight. The following additional adverse reactions were reported by < 0.5% of 3,960 patients who received doxazosin in controlled or open, short- or long-term clinical studies, including international studies. Cardiovascular System: angina pectoris, myocardial infarction, cerebrovascular accident; Autonomic Nervous System: pallor; Metabolic: thirst, gout, hypokalemia; Hematopoietic: lymphadenopathy, purpura; Reproductive System: breast pain; Skin Disorders: alopecia, dry skin, eczema; Central Nervous System: paresis, tremor, twitching, confusion, migraine, impaired concentration; Psychiatric: paroniria, amnesia, emotional lability, abnormal thinking, depersonalization; Special Senses: parosmia, earache, taste perversion, photophobia, abnormal lacrimation; Gastrointestinal System: increased appetite, anorexia, fecal incontinence, gastroenteritis; Respiratory System: bronchospasm, sinusitis, coughing, pharyngitis; Urinary System: renal calculus; General Body System: hot flushes, back pain, infection, fever/rigors, decreased weight, influenza-like symptoms. Doxazosin has not been associated with any clinically significant changes in routine biochemical tests. No clinically relevant adverse effects were noted on serum potassium, serum glucose, uric acid, blood urea nitrogen, creatinine or liver function tests. Doxazosin has been associated with decreases in white blood cell counts. In post-marketing experience the following additional adverse reactions have been reported: Autonomic Nervous System: priapism; Central Nervous System: hypoesthesia; Endocrine System: gynecomastia; Gastrointestinal System: vomiting; General Body System: allergic reaction; Heart Rate/Rhythm: bradycardia; Hematopoietic: leukopenia, thrombocytopenia; Liver/Biliary System: hepatitis, hepatitis cholestatic; Respiratory System: bronchospasm aggravated; Skin Disorders: urticaria; Special Senses: Intraoperative Floppy Iris Syndrome (see PRECAUTIONS: General: Cataract Surgery); Urinary System: hematuria, micturition disorder, micturition frequency, nocturia.
Postmarketing Experience
(Description)
Drug Interactions
- Drug 1
- Drug 2
- Drug 3
- Drug 4
- Drug 5
Drug 1
(Description)
Drug 2
(Description)
Drug 3
(Description)
Drug 4
(Description)
Drug 5
(Description)
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
(Description)
Pregnancy Category (AUS):
(Description)
Labor and Delivery
(Description)
Nursing Mothers
(Description)
Pediatric Use
(Description)
Geriatic Use
(Description)
Gender
(Description)
Race
(Description)
Renal Impairment
(Description)
Hepatic Impairment
(Description)
Females of Reproductive Potential and Males
(Description)
Immunocompromised Patients
(Description)
Others
(Description)
Administration and Monitoring
Administration
(Oral/Intravenous/etc)
Monitoring
Condition 1
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section)
IV Compatibility
Solution
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Y-Site
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Admixture
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Syringe
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
TPN/TNA
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Overdosage
- Experience with doxazosin overdosage is limited.
Pharmacology
Doxazosin
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
(Description)
Structure
(Description with picture)
Pharmacodynamics
(Description)
Pharmacokinetics
(Description)
Nonclinical Toxicology
(Description)
Clinical Studies
Condition 1
(Description)
Condition 2
(Description)
Condition 3
(Description)
How Supplied
(Description)
Storage
There is limited information regarding Doxazosin Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Doxazosin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Doxazosin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
(Patient Counseling Information)
Precautions with Alcohol
Alcohol-Doxazonin sandbox interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Doxazosin Brand Names in the drug label.
Look-Alike Drug Names
- (Paired Confused Name 1a) — (Paired Confused Name 1b)
- (Paired Confused Name 2a) — (Paired Confused Name 2b)
- (Paired Confused Name 3a) — (Paired Confused Name 3b)
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.