Diglyceride
Overview
A diglyceride, more correctly known as a diacylglycerol, is a glyceride consisting of two fatty acid chains covalently bonded to a glycerol molecule through ester linkages. Shown on the right is 1-palmitoyl-2-oleoyl-glycerol but diacylglycerol can have many different combinations of fatty acids attached at the C-1 and C-2 positions.
Food Additive
Mono- and Diacylglycerols are common food additives used to blend together certain ingredients, such as oil and water, which would not otherwise blend well.
The commercial source may be either animal (cow- or hog-derived) or vegetable, derived primarily from soy bean and canola oil. They may also be synthetically produced. They are often found in bakery products, beverages, ice cream, chewing gum, shortening, whipped toppings, margarine, and confections.
Function
Activate PKC
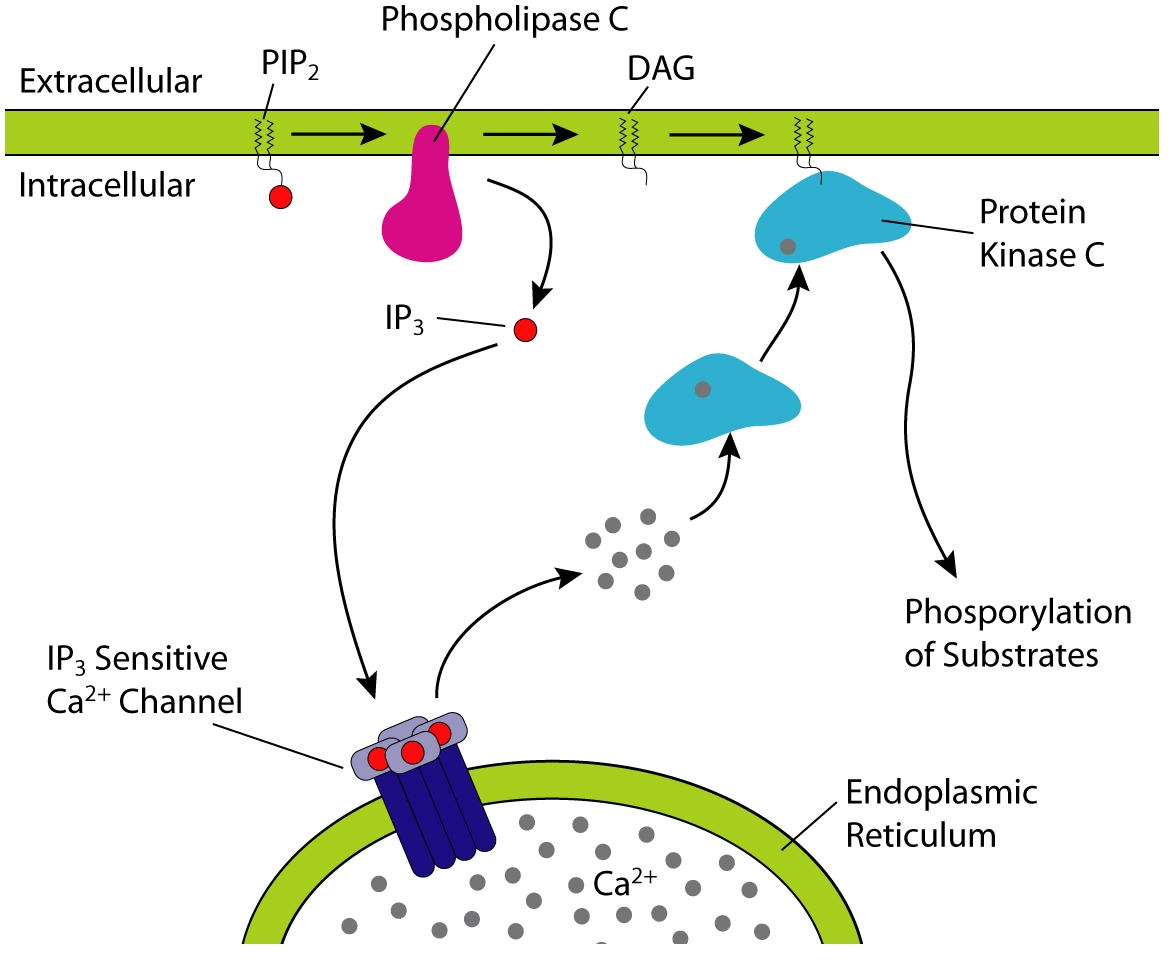
In biochemical signaling, diacylglycerol (DAG) functions as a second messenger signaling lipid, a resultant of a reaction conducted by enzyme phospholipase C (PLC) (a membrane-bound enzyme) that, through the same reaction, produces inositol triphosphate (IP3). Although inositol triphosphate (IP3) diffuses into the cytosol, diacylglycerol (DAG) stays close to the plasma membrane, due to its hydrophobic properties. IP3 stimulates the release of calcium ions from the smooth endoplasmic reticulum, whereas DAG is still membrane-associated and activates protein kinase C (PKC). However, for DAG to activate PKC, there needs to be a cytosolic increase in calcium ions, which is accomplished by IP3. Diacylglycerol can be mimicked by phorbol esters.
Further reading: function of PKC
Other
In addition to activating PKC, diacylglycerol has a number of other functions in the cell:
- a source for prostaglandins
- a precursor of the endocannabinoid 2-arachidonoylglycerol
- an activator of a subfamily of TRPC (Transient Receptor Potential Canonical) cation channels, TRPC3/6/7.
Metabolism
Synthesis of diacylglycerol begins with glycerol-3-phosphate, which is derived primarily from dihydroxyacetone phosphate, a product of glycolysis (usually in the cytoplasm of liver or adipose tissue cells). Glycerol-3-phosphate is first acylated with acyl-coenzyme A (acyl-CoA) to form lysophosphatidic acid, which is then acylated with another molecule of acyl-CoA to yield phosphatidic acid. Phosphatidic acid is then de-phosphorylated to form diacylglycerol.
Diacylglycerol is a precursor to triacylglycerol (triglyceride), which is formed in the addition of a third fatty acid to the diacylglycerol under the catalysis of diglyceride acyltransferase.
Since diacylglycerol is synthesized via phosphatidic acid, it will usually contain a saturated fatty acid at the C-1 position on the glycerol moiety and an unsaturated fatty acid at the C-2 position. [1]
Additional images
-
PIP2 cleavage to IP3 and DAG initiates intracellular calcium release and PKC activation.
References
- ↑ Berg J, Tymoczko JL, Stryer L (2006). Biochemistry (6th ed. ed.). San Francisco: W. H. Freeman. ISBN 0716787245.
da:Diglycerid de:Diacylglycerol he:דיגליצריד ur:دو ایسائل گلسرول Template:WH Template:WS Template:Jb1