Deferasirox: Difference between revisions
No edit summary |
No edit summary |
||
| Line 179: | Line 179: | ||
|warnings= | |warnings= | ||
====Precautions==== | ====Precautions==== | ||
* | * Renal Toxicity, Renal Failure, and Proteinuria | ||
:*Exjade can cause acute renal failure, fatal in some patients and requiring dialysis in others. Postmarketing experience showed that most fatalities occurred in patients with multiple comorbidities and who were in advanced stages of their hematological disorders. In the clinical trials, Exjade-treated patients experienced dose-dependent increases in serum creatinine. In patients with transfusional iron overload, these increases in creatinine occurred at a greater frequency compared to deferoxamine-treated patients (38% versus 14%, respectively, in Study 1 and 36% versus 22%, respectively, in Study 3). | |||
:*Measure serum creatinine in duplicate (due to variations in measurements) and determine the creatinine clearance (estimated by the Cockcroft-Gault method) before initiating therapy in all patients in order to establish a reliable pretreatment baseline. Monitor serum creatinine weekly during the first month after initiation or modification of therapy and at least monthly thereafter. Monitor serum creatinine and/or creatinine clearance more frequently if creatinine levels are increasing. Dose reduction, interruption, or discontinuation based on increases in serum creatinine may be necessary. | |||
:*Exjade is contraindicated in patients with creatinine clearance less than 40 mL/minute or serum creatinine greater than 2 times the age appropriate upper limit of normal. | |||
:*Renal tubular damage, including Fanconi’s Syndrome, has been reported in patients treated with Exjade, most commonly in children and adolescents with beta-thalassemia and serum ferritin levels <;<1500 mcg/L. | |||
:*Intermittent proteinuria (urine protein/creatinine ratio >0.6 mg/mg) occurred in 18.6% of Exjade-treated patients compared to 7.2% of deferoxamine-treated patients in Study 1. In clinical trials in patients with transfusional iron overload, Exjade was temporarily withheld until the urine protein/creatinine ratio fell below 0.6 mg/mg. Monthly monitoring for proteinuria is recommended. The mechanism and clinical significance of the proteinuria are uncertain. | |||
*Hepatic Toxicity and Failure | |||
:*Exjade can cause hepatic injury, fatal in some patients. In Study 1, 4 patients (1.3%) discontinued Exjade because of hepatic toxicity (drug-induced hepatitis in 2 patients and increased serum transaminases in 2 additional patients). Hepatic toxicity appears to be more common in patients greater than 55 years of age. Hepatic failure was more common in patients with significant comorbidities, including liver cirrhosis and multiorgan failure. | |||
:*Measure transaminases (AST and ALT) and bilirubin in all patients before the initiation of treatment and every 2 weeks during the first month and at least monthly thereafter. Consider dose modifications or interruption of treatment for severe or persistent elevations. | |||
:*Avoid the use of Exjade in patients with severe (Child-Pugh C) hepatic impairment. Reduce the starting dose in patients with moderate (Child-Pugh B) hepatic impairment. Patients with mild (Child-Pugh A) or moderate (Child-Pugh B) hepatic impairment may be at higher risk for hepatic toxicity. | |||
*Gastrointestinal (GI) Hemorrhage | |||
:*GI hemorrhage, including deaths, has been reported, especially in elderly patients who had advanced hematologic malignancies and/or low platelet counts. Nonfatal upper GI irritation, ulceration and hemorrhage have been reported in patients, including children and adolescents, receiving Exjade [see Adverse Reactions (6.1)]. Monitor for signs and symptoms of GI ulceration and hemorrhage during Exjade therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. The risk of gastrointestinal hemorrhage may be increased when administering Exjade in combination with drugs that have ulcerogenic or hemorrhagic potential, such as nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, oral bisphosphonates, or anticoagulants. | |||
*Bone Marrow Suppression | |||
:*Neutropenia, agranulocytosis, worsening anemia, and thrombocytopenia, including fatal events, have been reported in patients treated with Exjade. Preexisting hematologic disorders may increase this risk. Monitor blood counts in all patients. Interrupt treatment with Exjade in patients who develop cytopenias until the cause of the cytopenia has been determined. Exjade is contraindicated in patients with platelet counts below 50 x 109/L. | |||
*Increased Risk of Toxicity in the Elderly | |||
:*Exjade has been associated with serious and fatal adverse reactions in the postmarketing setting, predominantly in elderly patients. Monitor elderly patients treated with Exjade more frequently for toxicity. | |||
*Hypersensitivity | |||
:*Exjade may cause serious hypersensitivity reactions (such as anaphylaxis and angioedema), with the onset of the reaction usually occurring within the first month of treatment [see Adverse Reactions (6.2)]. If reactions are severe, discontinue Exjade and institute appropriate medical intervention. Exjade is contraindicated in patients with known hypersensitivity to Exjade. | |||
*Severe Skin Reactions | |||
:*Severe skin reactions, including Stevens-Johnson syndrome (SJS) and erythema multiforme, have been reported during Exjade therapy [see Adverse Reactions (6.2)]. If SJS or erythema multiforme is suspected, discontinue Exjade and evaluate. | |||
*Skin Rash | |||
:*Rashes may occur during Exjade treatment [see Adverse Reactions (6.1)]. For rashes of mild to moderate severity, Exjade may be continued without dose adjustment, since the rash often resolves spontaneously. In severe cases, interrupt treatment with Exjade. Reintroduction at a lower dose with escalation may be considered in combination with a short period of oral steroid administration. | |||
* Auditory and Ocular Abnormalities | |||
:*Auditory disturbances (high frequency hearing loss, decreased hearing), and ocular disturbances (lens opacities, cataracts, elevations in intraocular pressure, and retinal disorders) were reported at a frequency of <1% with Exjade therapy in the clinical studies. Perform auditory and ophthalmic testing (including slit lamp examinations and dilated fundoscopy) before starting Exjade treatment and thereafter at regular intervals (every 12 months). If disturbances are noted, monitor more frequently. Consider dose reduction or interruption. | |||
*Overchelation | |||
:*For patients with transfusional iron overload, measure serum ferritin monthly to assess for possible overchelation of iron. If the serum ferritin falls below 500 mcg/L, consider interrupting therapy with Exjade, since overchelation may increase Exjade toxicity [see Dosage and Administration (2.1)]. | |||
:*For patients with NTDT, measure LIC by liver biopsy or by using an FDA-cleared or approved method for monitoring patients receiving deferasirox therapy every 6 months on treatment. Interrupt Exjade administration when the LIC is less than 3 mg Fe/g dw. Measure serum ferritin monthly, and if the serum ferritin falls below 300 mcg/L, interrupt Exjade and obtain a confirmatory LIC. | |||
<!--Adverse Reactions--> | |||
<!--Clinical Trials Experience--> | |||
|clinicalTrials= | |||
*Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. | |||
=====Transfusional Iron Overload===== | |||
*A total of 700 adult and pediatric patients were treated with Exjade (deferasirox) for 48 weeks in premarketing studies. ;<hese included 469 patients with beta-thalassemia, 99 with rare anemias, and 132 with sickle cell disease. Of these patients, 45% were male, 70% were Caucasian and 292 patients were <l16 years of age. In the sickle cell disease population, 89% of patients were black. Median treatment duration among the sickle cell patients was 51 weeks. Of the 700 patients treated, 469 (403 beta-thalassemia and 66 rare anemias) were entered into extensions of the original clinical protocols. In ongoing extension studies, median durations of treatment were 88-205 weeks. | |||
*Six hundred twenty-seven patients with MDS were enrolled across 5 uncontrolled trials. These studies varied in duration from 1 to 5 years. The discontinuation rate across studies in the first year was 46% (AEs 20%, withdrawal of consent 10%, death 8%, other 4%, lab abnormalities 3%, and lack of efficacy 1%). Among 47 patients enrolled in the study of 5-year duration, 10 remained on Exjade at the completion of the study. | |||
*Table 1 displays adverse reactions occurring in >5% of Exjade-treated beta-thalassemia patients (Study 1), sickle cell disease patients (Study 3), and patients with MDS (MDS pool). Abdominal pain, nausea, vomiting, diarrhea, skin rashes, and increases in serum creatinine were the most frequent adverse reactions reported with a suspected relationship to Exjade. Gastrointestinal symptoms, increases in serum creatinine, and skin rash were dose related. | |||
T1 | |||
*In Study 1, a total of 113 (38%) patients treated with Exjade had increases in serum creatinine >33% above baseline on 2 separate occasions (Table 2) and 25 (8%) patients required dose reductions. Increases in serum creatinine appeared to be dose related [see Warnings and Precautions (5.1)]. In this study, 17 (6%) patients treated with Exjade developed elevations in SGPT/ALT levels >5 times the upper limit of normal at 2 consecutive visits. Of these, 2 patients had liver biopsy proven drug-induced hepatitis and both discontinued Exjade therapy [see Warnings and Precautions (5.2)]. An additional 2 patients, who did not have elevations in SGPT/ALT >5 times the upper limit of normal, discontinued Exjade because of increased SGPT/ALT. Increases in transaminases did not appear to be dose related. Adverse reactions that led to discontinuations included abnormal liver function tests (2 patients) and drug-induced hepatitis (2 patients), skin rash, glycosuria/proteinuria, Henoch Schönlein purpura, hyperactivity/insomnia, drug fever, and cataract (1 patient each). | |||
*In Study 3, a total of 48 (36%) patients treated with Exjade had increases in serum creatinine >33% above baseline on 2 separate occasions (Table 2) [see Warnings and Precautions (5.1)]. Of the patients who experienced creatinine increases in Study 3, 8 Exjade-treated patients required dose reductions. In this study, 5 patients in the Exjade group developed elevations in SGPT/ALT levels >5 times the upper limit of normal at 2 consecutive visits and 1 patient subsequently had Exjade permanently discontinued. Four additional patients discontinued Exjade due to adverse reactions with a suspected relationship to study drug, including diarrhea, pancreatitis associated with gallstones, atypical tuberculosis, and skin rash. | |||
*In the MDS pool, in the first year, a total of 229 (37%) patients treated with Exjade had increases in serum creatinine >33% above baseline on 2 consecutive occasions (Table 2) and 8 (3.5%) patients permanently discontinued [see Warnings and Precautions (5.1)]. A total of 5 (0.8%) patients developed SGPT/ALT levels >5 times the upper limit of normal at 2 consecutive visits. The most frequent adverse reactions that led to discontinuation included increases in serum creatinine, diarrhea, nausea, rash, and vomiting. Death was reported in the first year in 52 (8%) of patients [see Clinical Studies (14)]. | |||
T2 | |||
===== | =====Non-Transfusion-Dependent Thalassemia Syndromes===== | ||
*In Study 4, 110 patients with NTDT received 1 year of treatment with Exjade 5 or 10 mg/kg/day and 56 patients received placebo in a double-blind, randomized trial. In Study 5, 130 of the patients who completed Study 4 were treated with open-label Exjade at 5, 10, or 20 mg/kg/day (depending on the baseline LIC) for 1 year [see Clinical Studies (14)]. Table 3 displays adverse reactions occurring in >5% in any group. The most frequent adverse reactions with a suspected relationship to study drug were nausea, rash, and diarrhea. | |||
T3 | |||
In Study 4, 1 patient in the placebo 10 mg/kg/day group experienced an ALT increase to >5 times ULN and >2 times baseline (Table 4). Three Exjade-treated patients (all in the 10 mg/kg/day group) had 2 consecutive serum creatinine level increases >33% from baseline and >ULN. Serum creatinine returned to normal in all 3 patients (in 1 spontaneously and in the other 2 after drug interruption). Two additional cases of ALT increase and 2 additional cases of serum creatinine increase were observed in the 1-year extension of Study 4. | |||
T4 | |||
*Proteinuria | |||
:*In clinical studies, urine protein was measured monthly. Intermittent proteinuria (urine protein/creatinine ratio >0.6 mg/mg) occurred in 18.6% of Exjade-treated patients compared to 7.2% of deferoxamine-treated patients in Study 1 [see Warnings and Precautions (5.1)]. | |||
*Other Adverse Reactions | |||
:*In the population of more than 5,000 patients with transfusional iron overload who have been treated with Exjade during clinical trials, adverse reactions occurring in 0.1% to 1% of patients included gastritis, edema, sleep disorder, pigmentation disorder, dizziness, anxiety, maculopathy, cholelithiasis, pyrexia, fatigue, pharyngolaryngeal pain, early cataract, hearing loss, gastrointestinal hemorrhage, gastric ulcer (including multiple ulcers), duodenal ulcer, and renal tubulopathy (Fanconi’s Syndrome). Adverse reactions occurring in 0.01% to 0.1% of patients included optic neuritis, esophagitis, and erythema multiforme. Adverse reactions which most frequently led to dose interruption or dose adjustment during clinical trials were rash, gastrointestinal disorders, infections, increased serum creatinine, and increased serum transaminases. | |||
<!--Postmarketing Experience--> | <!--Postmarketing Experience--> | ||
| Line 262: | Line 265: | ||
|postmarketing= | |postmarketing= | ||
*The following adverse reactions have been spontaneously reported during post-approval use of Exjade in the transfusional iron overload setting. Because these reactions are reported voluntarily from a population of uncertain size, in which patients may have received concomitant medication, it is not always possible to reliably estimate frequency or establish a causal relationship to drug exposure. | |||
===== | =====Skin and subcutaneous tissue disorders===== | ||
Stevens-Johnson syndrome (SJS), leukocytoclastic vasculitis, urticaria, alopecia | |||
=====Immune system disorders===== | |||
Hypersensitivity reactions (including anaphylaxis and angioedema) | |||
=====Renal and urinary disorders===== | |||
Acute renal failure, tubulointerstitial nephritis | |||
===== | =====Hepatobiliary disorders===== | ||
Hepatic failure | |||
=====Gastrointestinal disorders===== | |||
Gastrointestinal hemorrhage | |||
=====Blood and lymphatic system disorders===== | |||
Worsening anemia | |||
<!--Drug Interactions--> | |||
|drugInteractions= | |||
* Aluminum Containing Antacid Preparations | |||
The concomitant administration of Exjade and aluminum-containing antacid preparations has not been formally studied. Although deferasirox has a lower affinity for aluminum than for iron, avoid use of Exjade with aluminum-containing antacid preparations due to the mechanism of action of Exjade. | |||
7.2 Agents Metabolized by CYP3A4 | |||
Deferasirox may induce CYP3A4 resulting in a decrease in CYP3A4 substrate concentration when these drugs are coadministered. Closely monitor patients for signs of reduced effectiveness when deferasirox is administered with drugs metabolized by CYP3A4 (e.g., alfentanil, aprepitant, budesonide, buspirone, conivaptan, cyclosporine, darifenacin, darunavir, dasatinib, dihydroergotamine, dronedarone, eletriptan, eplerenone, ergotamine, everolimus, felodipine, fentanyl, hormonal contraceptive agents, indinavir, fluticasone, lopinavir, lovastatin, lurasidone, maraviroc, midazolam, nisoldipine, pimozide, quetiapine, quinidine, saquinavir, sildenafil, simvastatin, sirolimus, tacrolimus, tolvaptan, tipranavir, triazolam, ticagrelor, and vardenafil) [see Clinical Pharmacology (12.3)]. | |||
7.3 Agents Metabolized by CYP2C8 | |||
Deferasirox inhibits CYP2C8 resulting in an increase in CYP2C8 substrate (e.g., repaglinide and paclitaxel) concentration when these drugs are coadministered. If Exjade and repaglinide are used concomitantly, consider decreasing the dose of repaglinide and perform careful monitoring of blood glucose levels. Closely monitor patients for signs of exposure related toxicity when Exjade is coadministered with other CYP2C8 substrates [see Clinical Pharmacology (12.3)]. | |||
7.4 Agents Metabolized by CYP1A2 | |||
Deferasirox inhibits CYP1A2 resulting in an increase in CYP1A2 substrate (e.g., alosetron, caffeine, duloxetine, melatonin, ramelteon, tacrine, theophylline, tizanidine) concentration when these drugs are coadministered. An increase in theophylline plasma concentrations could lead to clinically significant theophylline induced CNS or other adverse reactions. Avoid the concomitant use of theophylline or other CYP1A2 substrates with a narrow therapeutic index (e.g., tizanidine) with Exjade. Monitor theophylline concentrations and consider theophylline dose modification if you must coadminister theophylline with Exjade. Closely monitor patients for signs of exposure related toxicity when Exjade is coadministered with other drugs metabolized by CYP1A2 [see Clinical Pharmacology (12.3)]. | |||
7.5 Agents Inducing UDP-glucuronosyltransferase (UGT) Metabolism | |||
Deferasirox is a substrate of UGT1A1 and to a lesser extent UGT1A3. The concomitant use of Exjade with potent UGT inducers (e.g., rifampicin, phenytoin, phenobarbital, ritonavir) may result in a decrease in Exjade efficacy due to a possible decrease in deferasirox concentration. Avoid the concomitant use of potent UGT inducers with Exjade. Consider increasing the initial dose of Exjade if you must coadminister these agents together [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)]. | |||
7.6 Bile Acid Sequestrants | |||
Avoid the concomitant use of bile acid sequestrants (e.g., cholestyramine, colesevelam, colestipol) with Exjade due to a possible decrease in deferasirox concentration. If you must coadminister these agents together, consider increasing the initial dose of Exjade [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)]. | |||
<!--Use in Specific Populations--> | <!--Use in Specific Populations--> | ||
Revision as of 13:59, 4 February 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: RENAL FAILURE, HEPATIC FAILURE, AND GASTROINTESTINAL HEMORRHAGE
See full prescribing information for complete Boxed Warning.
ConditionName:
|
Overview
Deferasirox is a that is FDA approved for the {{{indicationType}}} of . There is a Black Box Warning for this drug as shown here. Common adverse reactions include .
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Condition1
- Dosing Information
- Dosage
Condition2
- Dosing Information
- Dosage
Condition3
- Dosing Information
- Dosage
Condition4
- Dosing Information
- Dosage
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Condition1
- Developed by:
- Class of Recommendation:
- Strength of Evidence:
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Guideline-Supported Use of Deferasirox in adult patients.
Non–Guideline-Supported Use
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Non–Guideline-Supported Use of Deferasirox in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding FDA-Labeled Use of Deferasirox in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition1
- Developed by:
- Class of Recommendation:
- Strength of Evidence:
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Guideline-Supported Use of Deferasirox in pediatric patients.
Non–Guideline-Supported Use
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Non–Guideline-Supported Use of Deferasirox in pediatric patients.
Contraindications
- Condition1
Warnings
|
WARNING: RENAL FAILURE, HEPATIC FAILURE, AND GASTROINTESTINAL HEMORRHAGE
See full prescribing information for complete Boxed Warning.
ConditionName:
|
Precautions
- Renal Toxicity, Renal Failure, and Proteinuria
- Exjade can cause acute renal failure, fatal in some patients and requiring dialysis in others. Postmarketing experience showed that most fatalities occurred in patients with multiple comorbidities and who were in advanced stages of their hematological disorders. In the clinical trials, Exjade-treated patients experienced dose-dependent increases in serum creatinine. In patients with transfusional iron overload, these increases in creatinine occurred at a greater frequency compared to deferoxamine-treated patients (38% versus 14%, respectively, in Study 1 and 36% versus 22%, respectively, in Study 3).
- Measure serum creatinine in duplicate (due to variations in measurements) and determine the creatinine clearance (estimated by the Cockcroft-Gault method) before initiating therapy in all patients in order to establish a reliable pretreatment baseline. Monitor serum creatinine weekly during the first month after initiation or modification of therapy and at least monthly thereafter. Monitor serum creatinine and/or creatinine clearance more frequently if creatinine levels are increasing. Dose reduction, interruption, or discontinuation based on increases in serum creatinine may be necessary.
- Exjade is contraindicated in patients with creatinine clearance less than 40 mL/minute or serum creatinine greater than 2 times the age appropriate upper limit of normal.
- Renal tubular damage, including Fanconi’s Syndrome, has been reported in patients treated with Exjade, most commonly in children and adolescents with beta-thalassemia and serum ferritin levels <;<1500 mcg/L.
- Intermittent proteinuria (urine protein/creatinine ratio >0.6 mg/mg) occurred in 18.6% of Exjade-treated patients compared to 7.2% of deferoxamine-treated patients in Study 1. In clinical trials in patients with transfusional iron overload, Exjade was temporarily withheld until the urine protein/creatinine ratio fell below 0.6 mg/mg. Monthly monitoring for proteinuria is recommended. The mechanism and clinical significance of the proteinuria are uncertain.
- Hepatic Toxicity and Failure
- Exjade can cause hepatic injury, fatal in some patients. In Study 1, 4 patients (1.3%) discontinued Exjade because of hepatic toxicity (drug-induced hepatitis in 2 patients and increased serum transaminases in 2 additional patients). Hepatic toxicity appears to be more common in patients greater than 55 years of age. Hepatic failure was more common in patients with significant comorbidities, including liver cirrhosis and multiorgan failure.
- Measure transaminases (AST and ALT) and bilirubin in all patients before the initiation of treatment and every 2 weeks during the first month and at least monthly thereafter. Consider dose modifications or interruption of treatment for severe or persistent elevations.
- Avoid the use of Exjade in patients with severe (Child-Pugh C) hepatic impairment. Reduce the starting dose in patients with moderate (Child-Pugh B) hepatic impairment. Patients with mild (Child-Pugh A) or moderate (Child-Pugh B) hepatic impairment may be at higher risk for hepatic toxicity.
- Gastrointestinal (GI) Hemorrhage
- GI hemorrhage, including deaths, has been reported, especially in elderly patients who had advanced hematologic malignancies and/or low platelet counts. Nonfatal upper GI irritation, ulceration and hemorrhage have been reported in patients, including children and adolescents, receiving Exjade [see Adverse Reactions (6.1)]. Monitor for signs and symptoms of GI ulceration and hemorrhage during Exjade therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. The risk of gastrointestinal hemorrhage may be increased when administering Exjade in combination with drugs that have ulcerogenic or hemorrhagic potential, such as nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, oral bisphosphonates, or anticoagulants.
- Bone Marrow Suppression
- Neutropenia, agranulocytosis, worsening anemia, and thrombocytopenia, including fatal events, have been reported in patients treated with Exjade. Preexisting hematologic disorders may increase this risk. Monitor blood counts in all patients. Interrupt treatment with Exjade in patients who develop cytopenias until the cause of the cytopenia has been determined. Exjade is contraindicated in patients with platelet counts below 50 x 109/L.
- Increased Risk of Toxicity in the Elderly
- Exjade has been associated with serious and fatal adverse reactions in the postmarketing setting, predominantly in elderly patients. Monitor elderly patients treated with Exjade more frequently for toxicity.
- Hypersensitivity
- Exjade may cause serious hypersensitivity reactions (such as anaphylaxis and angioedema), with the onset of the reaction usually occurring within the first month of treatment [see Adverse Reactions (6.2)]. If reactions are severe, discontinue Exjade and institute appropriate medical intervention. Exjade is contraindicated in patients with known hypersensitivity to Exjade.
- Severe Skin Reactions
- Severe skin reactions, including Stevens-Johnson syndrome (SJS) and erythema multiforme, have been reported during Exjade therapy [see Adverse Reactions (6.2)]. If SJS or erythema multiforme is suspected, discontinue Exjade and evaluate.
- Skin Rash
- Rashes may occur during Exjade treatment [see Adverse Reactions (6.1)]. For rashes of mild to moderate severity, Exjade may be continued without dose adjustment, since the rash often resolves spontaneously. In severe cases, interrupt treatment with Exjade. Reintroduction at a lower dose with escalation may be considered in combination with a short period of oral steroid administration.
- Auditory and Ocular Abnormalities
- Auditory disturbances (high frequency hearing loss, decreased hearing), and ocular disturbances (lens opacities, cataracts, elevations in intraocular pressure, and retinal disorders) were reported at a frequency of <1% with Exjade therapy in the clinical studies. Perform auditory and ophthalmic testing (including slit lamp examinations and dilated fundoscopy) before starting Exjade treatment and thereafter at regular intervals (every 12 months). If disturbances are noted, monitor more frequently. Consider dose reduction or interruption.
- Overchelation
- For patients with transfusional iron overload, measure serum ferritin monthly to assess for possible overchelation of iron. If the serum ferritin falls below 500 mcg/L, consider interrupting therapy with Exjade, since overchelation may increase Exjade toxicity [see Dosage and Administration (2.1)].
- For patients with NTDT, measure LIC by liver biopsy or by using an FDA-cleared or approved method for monitoring patients receiving deferasirox therapy every 6 months on treatment. Interrupt Exjade administration when the LIC is less than 3 mg Fe/g dw. Measure serum ferritin monthly, and if the serum ferritin falls below 300 mcg/L, interrupt Exjade and obtain a confirmatory LIC.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Transfusional Iron Overload
- A total of 700 adult and pediatric patients were treated with Exjade (deferasirox) for 48 weeks in premarketing studies. ;<hese included 469 patients with beta-thalassemia, 99 with rare anemias, and 132 with sickle cell disease. Of these patients, 45% were male, 70% were Caucasian and 292 patients were <l16 years of age. In the sickle cell disease population, 89% of patients were black. Median treatment duration among the sickle cell patients was 51 weeks. Of the 700 patients treated, 469 (403 beta-thalassemia and 66 rare anemias) were entered into extensions of the original clinical protocols. In ongoing extension studies, median durations of treatment were 88-205 weeks.
- Six hundred twenty-seven patients with MDS were enrolled across 5 uncontrolled trials. These studies varied in duration from 1 to 5 years. The discontinuation rate across studies in the first year was 46% (AEs 20%, withdrawal of consent 10%, death 8%, other 4%, lab abnormalities 3%, and lack of efficacy 1%). Among 47 patients enrolled in the study of 5-year duration, 10 remained on Exjade at the completion of the study.
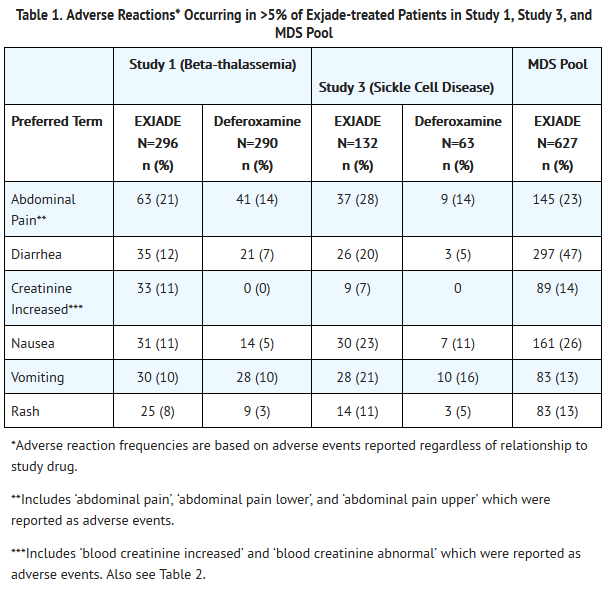
- Table 1 displays adverse reactions occurring in >5% of Exjade-treated beta-thalassemia patients (Study 1), sickle cell disease patients (Study 3), and patients with MDS (MDS pool). Abdominal pain, nausea, vomiting, diarrhea, skin rashes, and increases in serum creatinine were the most frequent adverse reactions reported with a suspected relationship to Exjade. Gastrointestinal symptoms, increases in serum creatinine, and skin rash were dose related.
T1
- In Study 1, a total of 113 (38%) patients treated with Exjade had increases in serum creatinine >33% above baseline on 2 separate occasions (Table 2) and 25 (8%) patients required dose reductions. Increases in serum creatinine appeared to be dose related [see Warnings and Precautions (5.1)]. In this study, 17 (6%) patients treated with Exjade developed elevations in SGPT/ALT levels >5 times the upper limit of normal at 2 consecutive visits. Of these, 2 patients had liver biopsy proven drug-induced hepatitis and both discontinued Exjade therapy [see Warnings and Precautions (5.2)]. An additional 2 patients, who did not have elevations in SGPT/ALT >5 times the upper limit of normal, discontinued Exjade because of increased SGPT/ALT. Increases in transaminases did not appear to be dose related. Adverse reactions that led to discontinuations included abnormal liver function tests (2 patients) and drug-induced hepatitis (2 patients), skin rash, glycosuria/proteinuria, Henoch Schönlein purpura, hyperactivity/insomnia, drug fever, and cataract (1 patient each).
- In Study 3, a total of 48 (36%) patients treated with Exjade had increases in serum creatinine >33% above baseline on 2 separate occasions (Table 2) [see Warnings and Precautions (5.1)]. Of the patients who experienced creatinine increases in Study 3, 8 Exjade-treated patients required dose reductions. In this study, 5 patients in the Exjade group developed elevations in SGPT/ALT levels >5 times the upper limit of normal at 2 consecutive visits and 1 patient subsequently had Exjade permanently discontinued. Four additional patients discontinued Exjade due to adverse reactions with a suspected relationship to study drug, including diarrhea, pancreatitis associated with gallstones, atypical tuberculosis, and skin rash.
- In the MDS pool, in the first year, a total of 229 (37%) patients treated with Exjade had increases in serum creatinine >33% above baseline on 2 consecutive occasions (Table 2) and 8 (3.5%) patients permanently discontinued [see Warnings and Precautions (5.1)]. A total of 5 (0.8%) patients developed SGPT/ALT levels >5 times the upper limit of normal at 2 consecutive visits. The most frequent adverse reactions that led to discontinuation included increases in serum creatinine, diarrhea, nausea, rash, and vomiting. Death was reported in the first year in 52 (8%) of patients [see Clinical Studies (14)].
T2
Non-Transfusion-Dependent Thalassemia Syndromes
- In Study 4, 110 patients with NTDT received 1 year of treatment with Exjade 5 or 10 mg/kg/day and 56 patients received placebo in a double-blind, randomized trial. In Study 5, 130 of the patients who completed Study 4 were treated with open-label Exjade at 5, 10, or 20 mg/kg/day (depending on the baseline LIC) for 1 year [see Clinical Studies (14)]. Table 3 displays adverse reactions occurring in >5% in any group. The most frequent adverse reactions with a suspected relationship to study drug were nausea, rash, and diarrhea.
T3
In Study 4, 1 patient in the placebo 10 mg/kg/day group experienced an ALT increase to >5 times ULN and >2 times baseline (Table 4). Three Exjade-treated patients (all in the 10 mg/kg/day group) had 2 consecutive serum creatinine level increases >33% from baseline and >ULN. Serum creatinine returned to normal in all 3 patients (in 1 spontaneously and in the other 2 after drug interruption). Two additional cases of ALT increase and 2 additional cases of serum creatinine increase were observed in the 1-year extension of Study 4.
T4
- Proteinuria
- In clinical studies, urine protein was measured monthly. Intermittent proteinuria (urine protein/creatinine ratio >0.6 mg/mg) occurred in 18.6% of Exjade-treated patients compared to 7.2% of deferoxamine-treated patients in Study 1 [see Warnings and Precautions (5.1)].
- Other Adverse Reactions
- In the population of more than 5,000 patients with transfusional iron overload who have been treated with Exjade during clinical trials, adverse reactions occurring in 0.1% to 1% of patients included gastritis, edema, sleep disorder, pigmentation disorder, dizziness, anxiety, maculopathy, cholelithiasis, pyrexia, fatigue, pharyngolaryngeal pain, early cataract, hearing loss, gastrointestinal hemorrhage, gastric ulcer (including multiple ulcers), duodenal ulcer, and renal tubulopathy (Fanconi’s Syndrome). Adverse reactions occurring in 0.01% to 0.1% of patients included optic neuritis, esophagitis, and erythema multiforme. Adverse reactions which most frequently led to dose interruption or dose adjustment during clinical trials were rash, gastrointestinal disorders, infections, increased serum creatinine, and increased serum transaminases.
Postmarketing Experience
- The following adverse reactions have been spontaneously reported during post-approval use of Exjade in the transfusional iron overload setting. Because these reactions are reported voluntarily from a population of uncertain size, in which patients may have received concomitant medication, it is not always possible to reliably estimate frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissue disorders
Stevens-Johnson syndrome (SJS), leukocytoclastic vasculitis, urticaria, alopecia
Immune system disorders
Hypersensitivity reactions (including anaphylaxis and angioedema)
Renal and urinary disorders
Acute renal failure, tubulointerstitial nephritis
Hepatobiliary disorders
Hepatic failure
Gastrointestinal disorders
Gastrointestinal hemorrhage
Blood and lymphatic system disorders
Worsening anemia
Drug Interactions
- Aluminum Containing Antacid Preparations
The concomitant administration of Exjade and aluminum-containing antacid preparations has not been formally studied. Although deferasirox has a lower affinity for aluminum than for iron, avoid use of Exjade with aluminum-containing antacid preparations due to the mechanism of action of Exjade.
7.2 Agents Metabolized by CYP3A4
Deferasirox may induce CYP3A4 resulting in a decrease in CYP3A4 substrate concentration when these drugs are coadministered. Closely monitor patients for signs of reduced effectiveness when deferasirox is administered with drugs metabolized by CYP3A4 (e.g., alfentanil, aprepitant, budesonide, buspirone, conivaptan, cyclosporine, darifenacin, darunavir, dasatinib, dihydroergotamine, dronedarone, eletriptan, eplerenone, ergotamine, everolimus, felodipine, fentanyl, hormonal contraceptive agents, indinavir, fluticasone, lopinavir, lovastatin, lurasidone, maraviroc, midazolam, nisoldipine, pimozide, quetiapine, quinidine, saquinavir, sildenafil, simvastatin, sirolimus, tacrolimus, tolvaptan, tipranavir, triazolam, ticagrelor, and vardenafil) [see Clinical Pharmacology (12.3)].
7.3 Agents Metabolized by CYP2C8
Deferasirox inhibits CYP2C8 resulting in an increase in CYP2C8 substrate (e.g., repaglinide and paclitaxel) concentration when these drugs are coadministered. If Exjade and repaglinide are used concomitantly, consider decreasing the dose of repaglinide and perform careful monitoring of blood glucose levels. Closely monitor patients for signs of exposure related toxicity when Exjade is coadministered with other CYP2C8 substrates [see Clinical Pharmacology (12.3)].
7.4 Agents Metabolized by CYP1A2
Deferasirox inhibits CYP1A2 resulting in an increase in CYP1A2 substrate (e.g., alosetron, caffeine, duloxetine, melatonin, ramelteon, tacrine, theophylline, tizanidine) concentration when these drugs are coadministered. An increase in theophylline plasma concentrations could lead to clinically significant theophylline induced CNS or other adverse reactions. Avoid the concomitant use of theophylline or other CYP1A2 substrates with a narrow therapeutic index (e.g., tizanidine) with Exjade. Monitor theophylline concentrations and consider theophylline dose modification if you must coadminister theophylline with Exjade. Closely monitor patients for signs of exposure related toxicity when Exjade is coadministered with other drugs metabolized by CYP1A2 [see Clinical Pharmacology (12.3)].
7.5 Agents Inducing UDP-glucuronosyltransferase (UGT) Metabolism
Deferasirox is a substrate of UGT1A1 and to a lesser extent UGT1A3. The concomitant use of Exjade with potent UGT inducers (e.g., rifampicin, phenytoin, phenobarbital, ritonavir) may result in a decrease in Exjade efficacy due to a possible decrease in deferasirox concentration. Avoid the concomitant use of potent UGT inducers with Exjade. Consider increasing the initial dose of Exjade if you must coadminister these agents together [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
7.6 Bile Acid Sequestrants
Avoid the concomitant use of bile acid sequestrants (e.g., cholestyramine, colesevelam, colestipol) with Exjade due to a possible decrease in deferasirox concentration. If you must coadminister these agents together, consider increasing the initial dose of Exjade [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
Use in Specific Populations
Pregnancy
- Pregnancy Category
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Deferasirox in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Deferasirox during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Deferasirox with respect to nursing mothers.
Pediatric Use
There is no FDA guidance on the use of Deferasirox with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of Deferasirox with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Deferasirox with respect to specific gender populations.
Race
There is no FDA guidance on the use of Deferasirox with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Deferasirox in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Deferasirox in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Deferasirox in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Deferasirox in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- Intravenous
Monitoring
There is limited information regarding Monitoring of Deferasirox in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Deferasirox in the drug label.
Overdosage
Acute Overdose
Signs and Symptoms
- Description
Management
- Description
Chronic Overdose
There is limited information regarding Chronic Overdose of Deferasirox in the drug label.
Pharmacology
There is limited information regarding Deferasirox Pharmacology in the drug label.
Mechanism of Action
Structure
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Deferasirox in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Deferasirox in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Deferasirox in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Deferasirox in the drug label.
How Supplied
Storage
There is limited information regarding Deferasirox Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Deferasirox |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Deferasirox |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Deferasirox in the drug label.
Precautions with Alcohol
- Alcohol-Deferasirox interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- ®[1]
Look-Alike Drug Names
- A® — B®[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Empty citation (help)
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Deferasirox |Pill Name=No image.jpg |Drug Name= |Pill Ingred=|+sep=; |Pill Imprint= |Pill Dosage= |Pill Color=|+sep=; |Pill Shape= |Pill Size (mm)= |Pill Scoring= |Pill Image= |Drug Author= |NDC=
}}
{{#subobject:
|Label Page=Deferasirox |Label Name=Deferasirox11.png
}}
{{#subobject:
|Label Page=Deferasirox |Label Name=Deferasirox11.png
}}