Clomipramine: Difference between revisions
No edit summary |
No edit summary |
||
| Line 62: | Line 62: | ||
|warnings=<b>Conidition 1</b> | |warnings=<b>Conidition 1</b> | ||
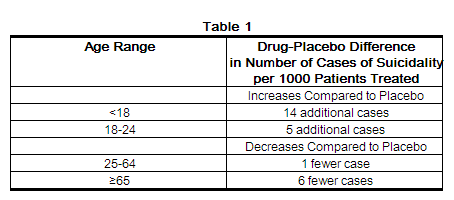
Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the strongest predictors of suicide. There has been a long-standing concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment. Pooled analyses of short-term placebo-controlled trials of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18-24) with major depressive disorder (MDD) and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older. | |||
The pooled analyses of placebo-controlled trials in children and adolescents with MDD, obsessive compulsive disorder (OCD), or other psychiatric disorders included a total of 24 short-term trials of 9 antidepressant drugs in over 4400 patients. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs placebo), however, were relatively stable within age strata and across indications. | |||
[[File:Clomipramine6.png|thumb|none|400px]] | [[File:Clomipramine6.png|thumb|none|400px]] | ||
|clinicalTrials=[[File:CLOMIPRAMINE1.PNG]] | |clinicalTrials=[[File:CLOMIPRAMINE1.PNG]] | ||
| Line 215: | Line 217: | ||
}} | }} | ||
|fileName=}} | |||
|fileName=}} | |fileName=}} | ||
|fileName=}} | |fileName=}} | ||
Revision as of 07:09, 16 May 2014
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Pratik Bahekar, MBBS [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Suicidality and Antidepressant Drug
See full prescribing information for complete Boxed Warning.
Condition Name:
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of clomipramine hydrochloride capsules or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Clomipramine hydrochloride capsules are not approved for use in pediatric patients except for patients with obsessive compulsive disorder (OCD) (see WARNINGS: Clinical Worsening and Suicide Risk, PRECAUTIONS: Information for Patients, and PRECAUTIONS: Pediatric Use).
|
Overview
Clomipramine is a Tricyclic antidepressant that is FDA approved for the {{{indicationType}}} of obsessive-compulsive disorder. There is a Black Box Warning for this drug as shown here. Common adverse reactions include diaphoresis, weight increased, constipation, diarrhea, indigestion, loss of appetite, nausea, xerostomia, myalgia, dizziness, feeling nervous, headache, insomnia, myoclonus, somnolence, tremor, abnormal vision, disorder of the urinary system, disorder of ejaculation, impotence, pharyngitis fatigue..
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Obsessive-compulsive disorder
- Monotherapy
- Maximum, 250 mg/day[1]
- Combination Therapy
- Initial dose, 25 mg/day; weekly titration up to 75 mg/day; mean dose, 55 mg/day) plus fluoxetine (maximum dose, 40 mg/day), quetiapine initial dose, 50 mg/day; weekly titration up to 200 mg/day; mean dose, 142 mg/day
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Condition 1
- Developed by: (Organization)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Delusional disorder
- Initial, 25 mg/day ORALLY, may increase dosage to 100 mg/day during the first 2 weeks (MAX dose 250 mg/day, mean dose 140 mg/day)
Depression
- Initial, 75 mg/day ORALLY (3 divided doses); may increase dosage slowly as needed and tolerated to a range of 100-250 mg/day (3 divided doses)
Disorder of ejaculation
Obsessive-compulsive disorder, Intravenous therapy
- Initial, 25 mg/day ORALLY, may increase dosage to 100 mg/day ORALLY during the first 2 weeks; MAX dose 250 mg/day
Pain, chronic
Panic disorder
- 25-75 mg/day ORALLY
Pervasive developmental disorder
- There is limited information about Off-Label Non–Guideline-Supported Use of Clomipramine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Obsessive-compulsive disorder
- 10 years and older, initial, 25 mg/day ORALLY, may increase dosage to 3 mg/kg or 100 mg/day (whichever is less) ORALLY during the first 2 weeks; MAX dose 200 mg/day OR 3 mg/kg/day (whichever is less)
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition 1
- Developed by: (Organization)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Depression
- Safety and effectiveness in children less than 10 years of age have not been established
- 20-30 mg/day ORALLY; may increase dosage by 10 mg/day at 4-5 day intervals as needed and tolerated
- There is limited information about Off-Label Non–Guideline-Supported Use of Clomipramine in pediatric patients.
Contraindications
- Contraindicated in patients with a history of hypersensitivity to clomipramine hydrochloride or other tricyclic antidepressants
- Monoamine Oxidase Inhibitors (MAOIs)-The use of MAOIs intended to treat psychiatric disorders with clomipramine or within 14 days of stopping treatment with clomipramine is contraindicated because of an increased risk of serotonin syndrome.
- The use of clomipramine within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated.
- Starting clomipramine in a patient who is being treated with linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome.
- Myocardial Infarction- Clomipramine is contraindicated during the acute recovery period after a myocardial infarction.
Warnings
|
Suicidality and Antidepressant Drug
See full prescribing information for complete Boxed Warning.
Condition Name:
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of clomipramine hydrochloride capsules or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Clomipramine hydrochloride capsules are not approved for use in pediatric patients except for patients with obsessive compulsive disorder (OCD) (see WARNINGS: Clinical Worsening and Suicide Risk, PRECAUTIONS: Information for Patients, and PRECAUTIONS: Pediatric Use).
|
Conidition 1
Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the strongest predictors of suicide. There has been a long-standing concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment. Pooled analyses of short-term placebo-controlled trials of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18-24) with major depressive disorder (MDD) and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older. The pooled analyses of placebo-controlled trials in children and adolescents with MDD, obsessive compulsive disorder (OCD), or other psychiatric disorders included a total of 24 short-term trials of 9 antidepressant drugs in over 4400 patients. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs placebo), however, were relatively stable within age strata and across indications.
Adverse Reactions
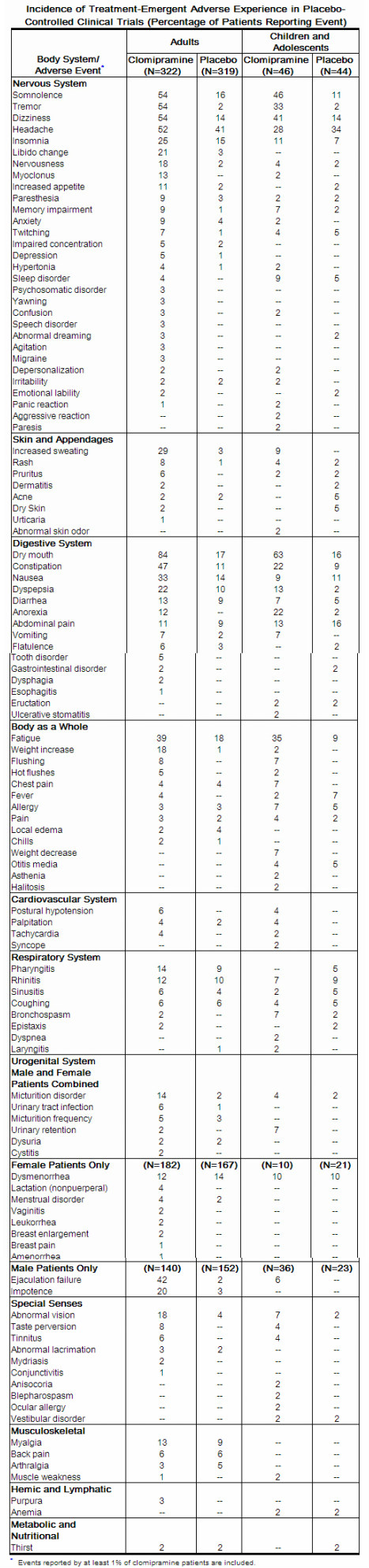
Clinical Trials Experience
Central Nervous System
- Frequent – abnormal thinking, vertigo. Infrequent – abnormal coordination, abnormal EEG, abnormal gait, apathy, ataxia, coma, convulsions, delirium, delusion, dyskinesia, dysphonia, encephalopathy, euphoria, extrapyramidal disorder, hallucinations, hostility, hyperkinesia, hypnagogic hallucinations, hypokinesia, leg cramps, manic reaction, neuralgia, paranoia, phobic disorder, psychosis, sensory disturbance, somnambulism, stimulation, suicidal ideation, suicide attempt, teeth-grinding. Rare – anticholinergic syndrome, aphasia, apraxia, catalepsy, cholinergic syndrome, choreoathetosis, generalized spasm, hemiparesis, hyperesthesia, hyperreflexia, hypoesthesia, illusion, impaired impulse control, indecisiveness, mutism, neuropathy, nystagmus, oculogyric crisis, oculomotor nerve paralysis, schizophrenic reaction, stupor, suicide.
Cardiovascular
- Infrequent – abnormal ECG, arrhythmia, bradycardia, cardiac arrest, extrasystoles, pallor. Rare – aneurysm, atrial flutter, bundle branch block, cardiac failure, cerebral hemorrhage, heart block, myocardial infarction, myocardial ischemia, peripheral ischemia, thrombophlebitis, vasospasm, ventricular tachycardia.
Respiratory
- Infrequent – bronchitis, hyperventilation, increased sputum, pneumonia. Rare – cyanosis, hemoptysis, hypoventilation, laryngismus.
Digestive System
- Infrequent – abnormal hepatic function, blood in stool, colitis, duodenitis, gastric ulcer, gastritis, gastroesophageal reflux, gingivitis, glossitis, hemorrhoids, hepatitis, increased saliva, irritable bowel syndrome, peptic ulcer, rectal hemorrhage, tongue ulceration, tooth caries. Rare – cheilitis, chronic enteritis, discolored feces, gastric dilatation, gingival bleeding, hiccup, intestinal obstruction, oral/pharyngeal edema, paralytic ileus, salivary gland enlargement.
Hypersensitive Reactions
- Infrequent – alopecia, cellulitis, cyst, eczema, erythematous rash, genital pruritus, maculopapular rash, photosensitivity reaction, psoriasis, pustular rash, skin discoloration. Rare – chloasma, folliculitis, hypertrichosis, piloerection, seborrhea, skin hypertrophy, skin ulceration.
Urogenital System
- Infrequent – endometriosis, epididymitis, hematuria, nocturia, oliguria, ovarian cyst, perineal pain, polyuria, prostatic disorder, renal calculus, renal pain, urethral disorder, urinary incontinence, uterine hemorrhage, vaginal hemorrhage. Rare – albuminuria, anorgasmy, breast engorgement, breast fibroadenosis, cervical dysplasia, endometrial hyperplasia, premature ejaculation, pyelonephritis, pyuria, renal cyst, uterine inflammation, vulvar disorder.
Endocrine System
- Infrequent – hypothyroidism. Rare – goiter, gynecomastia, hyperthyroidism.
Hemic and Lymphatic System
- Infrequent – lymphadenopathy. Rare – leukemoid reaction, lymphoma-like disorder, marrow depression.
Metabolic and Nutritional Disorder
- Infrequent – dehydration, diabetes mellitus, gout, hypercholesterolemia, hyperglycemia, hyperuricemia, hypokalemia. Rare – fat intolerance, glycosuria.
Musculoskeletal System
- Infrequent – arthrosis. Rare – dystonia, exostosis, lupus erythematosus rash, bruising, myopathy, myositis, polyarteritis nodosa, torticollis.
Miscellaneous
- The most commonly observed adverse events associated with the use of clomipramine and not seen at an equivalent incidence among placebo-treated patients were gastrointestinal complaints, including dry mouth, constipation, nausea, dyspepsia, and anorexia; nervous system complaints, including somnolence, tremor, dizziness, nervousness, and myoclonus; genitourinary complaints, including changed libido, ejaculatory failure, impotence, and micturition disorder; and other miscellaneous complaints, including fatigue, sweating, increased appetite, weight gain, and visual changes.
Postmarketing Experience
Central Nervous System
- (list/description of adverse reactions)
Cardiovascular
- (list/description of adverse reactions)
Respiratory
- (list/description of adverse reactions)
Gastrointestinal
- (list/description of adverse reactions)
Hypersensitive Reactions
- (list/description of adverse reactions)
Miscellaneous
- (list/description of adverse reactions)
Drug Interactions
- The risks of using clomipramine in combination with other drugs have not been systematically evaluated. Given the primary CNS effects of clomipramine, caution is advised in using it concomitantly with other CNS-active drugs. Clomipramine should not be used with MAO inhibitors.
- Close supervision and careful adjustment of dosage are required when clomipramine is administered with anticholinergic or sympathomimetic drugs.
- Several tricyclic antidepressants have been reported to block the pharmacologic effects of guanethidine, clonidine, or similar agents, and such an effect may be anticipated with clomipramine because of its structural similarity to other tricyclic antidepressants.
- The plasma concentration of clomipramine has been reported to be increased by the concomitant administration of haloperidol; plasma levels of several closely related tricyclic antidepressants have been reported to be increased by the concomitant administration of methylphenidate or hepatic enzyme inhibitors (e.g., cimetidine, fluoxetine) and decreased by the concomitant administration of hepatic enzyme inducers (e.g., barbiturates, phenytoin), and such an effect may be anticipated with clomipramine as well. Administration of clomipramine has been reported to increase the plasma levels of phenobarbital, if given concomitantly.
- Drugs Metabolized by P450 2D6-The biochemical activity of the drug metabolizing isozyme cytochrome P450 2D6 (debrisoquin hydroxylase) is reduced in a subset of the Caucasian population (about 7% to 10% of Caucasians are so-called “poor metabolizers”); reliable estimates of the prevalence of reduced P450 2D6 isozyme activity among Asian, African and other populations are not yet available. Poor metabolizers have higher than expected plasma concentrations of tricyclic antidepressants (TCAs) when given usual doses. Depending on the fraction of drug metabolized by P450 2D6, the increase in plasma concentration may be small, or quite large (8 fold increase in plasma AUC of the TCA). In addition, certain drugs inhibit the activity of this isozyme and make normal metabolizers resemble poor metabolizers. An individual who is stable on a given dose of TCA may become abruptly toxic when given one of these inhibiting drugs as concomitant therapy. The drugs that inhibit cytochrome P450 2D6 include some that are not metabolized by the enzyme (quinidine; cimetidine) and many that are substrates for P450 2D6 (many other antidepressants, phenothiazines, and the Type 1C antiarrhythmics propafenone and flecainide). While all the selective serotonin reuptake inhibitors (SSRIs), e.g., fluoxetine, sertraline, paroxetine, and fluvoxamine, inhibit P450 2D6, they may vary in the extent of inhibition. Fluvoxamine has also been shown to inhibit P450 1A2, an isoform also involved in TCA metabolism. The extent to which SSRI-TCA interactions may pose clinical problems will depend on the degree of inhibition and the pharmacokinetics of the SSRI involved. Nevertheless, caution is indicated in the co-administration of TCAs with any of the SSRIs and also in switching from one class to the other. Of particular importance, sufficient time must elapse before initiating TCA treatment in a patient being withdrawn from fluoxetine, given the long half-life of the parent and active metabolite (at least 5 weeks may be necessary). Concomitant use of agents in the tricyclic antidepressant class (which includes clomipramine hydrochloride) with drugs that can inhibit cytochrome P450 2D6 may require lower doses than usually prescribed for either the tricyclic antidepressant agent or the other drug. Furthermore, whenever one of these drugs is withdrawn from co-therapy, an increased dose of tricyclic antidepressant agent may be required. It is desirable to monitor TCA plasma levels whenever an agent of the tricyclic antidepressant class including clomipramine hydrochloride is going to be co-administered with another drug known to be an inhibitor of P450 2D6 (and/or P450 1A2).
- Because clomipramine hydrochloride is highly bound to serum protein, the administration of clomipramine hydrochloride to patients taking other drugs that are highly bound to protein (e.g., warfarin, digoxin) may cause an increase in plasma concentrations of these drugs, potentially resulting in adverse effects. Conversely, adverse effects may result from displacement of protein-bound clomipramine by other highly bound drugs.
Use in Specific Populations
Pregnancy
- No teratogenic effects were observed in studies performed in rats and mice at doses up to 100 mg/kg, which is 24 times the maximum recommended human daily dose (MRHD) on a mg/kg basis and 4 times (rats) and 2 times (mice) the MRHD on a mg/m2 basis. Slight nonspecific embryo/fetotoxic effects were seen in the offspring of treated rats given 50 and 100 mg/kg and of treated mice given 100 mg/kg.
- There are no adequate or well-controlled studies in pregnant women. Withdrawal symptoms, including jitteriness, tremor, and seizures, have been reported in neonates whose mothers had taken clomipramine hydrochloride until delivery. Clomipramine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Clomipramine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Clomipramine during labor and delivery.
Nursing Mothers
- Clomipramine has been found in human milk. Because of the potential for adverse reactions, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- Safety and effectiveness in the pediatric population other than pediatric patients with OCD have not been established . Anyone considering the use of clomipramine hydrochloride in a child or adolescent must balance the potential risks with the clinical need.
- In a controlled clinical trial in children and adolescents (10 to 17 years of age), 46 outpatients received clomipramine for up to 8 weeks. In addition, 150 adolescent patients have received clomipramine in open-label protocols for periods of several months to several years. Of the 196 adolescents studied, 50 were 13 years of age or less and 146 were 14 to 17 years of age. The adverse reaction profile in this age group is similar to that observed in adults.
- The risks, if any, that may be associated with clomipramine’s extended use in children and adolescents with OCD have not been systematically assessed. The evidence supporting the conclusion that clomipramine is safe for use in children and adolescents is derived from relatively short term clinical studies and from extrapolation of experience gained with adult patients. In particular, there are no studies that directly evaluate the effects of long term clomipramine use on the growth, development, and maturation of children and adolescents. Although there is no evidence to suggest that clomipramine adversely affects growth, development or maturation, the absence of such findings is not adequate to rule out a potential for such effects in chronic use.
- The safety and effectiveness in pediatric patients below the age of 10 have not been established. Therefore, specific recommendations cannot be made for the use of clomipramine in pediatric patients under the age of 10.
Geriatic Use
- Clinical studies of clomipramine hydrochloride did not include sufficient numbers of subjects age 65 and over to determine whether they respond differently from younger subjects; 152 patients at least 60 years of age participating in various U.S. clinical trials received clomipramine hydrochloride for periods of several months to several years. No unusual age-related adverse events were identified in this population. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
Gender
There is no FDA guidance on the use of Clomipramine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Clomipramine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Clomipramine in patients with renal impairment.
Hepatic Impairment
- During premarketing testing, clomipramine hydrochloride was occasionally associated with elevations in SGOT and SGPT (pooled incidence of approximately 1% and 3%, respectively) of potential clinical importance (i.e., values greater than 3 times the upper limit of normal). In the vast majority of instances, these enzyme increases were not associated with other clinical findings suggestive of hepatic injury; moreover, none were jaundiced. Rare reports of more severe liver injury, some fatal, have been recorded in foreign postmarketing experience. Caution is indicated in treating patients with known liver disease, and periodic monitoring of hepatic enzyme levels is recommended in such patients.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Clomipramine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Clomipramine in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Clomipramine Administration in the drug label.
Monitoring
There is limited information regarding Clomipramine Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Clomipramine and IV administrations.
Overdosage
There is limited information regarding Clomipramine overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology
Mechanism of Action
There is limited information regarding Clomipramine Mechanism of Action in the drug label.
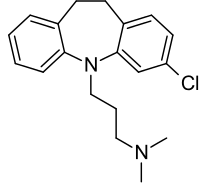
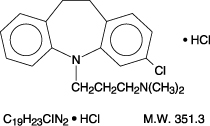
Structure
Pharmacodynamics
There is limited information regarding Clomipramine Pharmacodynamics in the drug label.
Pharmacokinetics
There is limited information regarding Clomipramine Pharmacokinetics in the drug label.
Nonclinical Toxicology
There is limited information regarding Clomipramine Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Clomipramine Clinical Studies in the drug label.
How Supplied
There is limited information regarding Clomipramine How Supplied in the drug label.
Storage
There is limited information regarding Clomipramine Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Clomipramine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Clomipramine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Clomipramine Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Clomipramine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Clomipramine Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Clomipramine Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Flament MF, Rapoport JL, Berg CJ, Sceery W, Kilts C, Mellström B et al. (1985) Clomipramine treatment of childhood obsessive-compulsive disorder. A double-blind controlled study. Arch Gen Psychiatry 42 (10):977-83. PMID: 3899048
- ↑ Template:Cite isbn
{{#subobject:
|Label Page=Clomipramine |Label Name=clomipramine2.png.jpg
}}
{{#subobject:
|Label Page=Clomipramine |Label Name=clomipramine3.png.jpg
}}
{{#subobject:
|Label Page=Clomipramine |Label Name=Clomipramine.png.jpg
}}
{{#subobject:
|Label Page=Clomipramine |Label Name=CLOMIPRAMINE33.PNG
}}
|fileName=}}
|fileName=}}
|fileName=}}
|fileName=}}
|fileName=}}
|fileName=}}
|fileName=}}
|fileName=}}
|fileName=}}