Clevidipine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sheng Shi, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Clevidipine is a Calcium Channel Blocker that is FDA approved for the {{{indicationType}}} of reduction of blood pressure when oral therapy is not feasible or not desirabl. Common adverse reactions include Nausea, Vomiting, Headache, Acute renal failure.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Hypertension
- Dosing information
- Initial dosage: 1-2 mg/hour
- Dose titration: The dose may be doubled at short (90 second) intervals initially. As the blood pressure approaches goal, the increase in doses should be less than doubling and the time between dose adjustments should be lengthened to every 5-10 minutes. An approximately 1-2 mg/hour increase will generally produce an additional 2-4 mmHg decrease in systolic pressure.
- Maintenance dose: The desired therapeutic response for most patients occurs at doses of 4-6 mg/hour. Patients with severe hypertension may require doses up to 32 mg/hour, but there is limited experience at this dose rate.
- Maximum dose: Most patients were treated with maximum doses of 16 mg/hour or less. There is limited short-term experience with doses up to 32 mg/hour. Because of lipid load restrictions, no more than 1000 mL or an average of 21 mg/hour of Clevidipine infusion is recommended per 24 hour period.
- Transition to an oral antihypertensive agent: Discontinue Clevidipine or titrate downward while appropriate oral therapy is established. When an oral antihypertensive agent is being instituted, consider the lag time of onset of the oral agent’s effect. Continue blood pressure monitoring until desired effect is achieved.
- Special populations: Special populations were not specifically studied. In clinical trials, 78 patients with abnormal hepatic function (one or more of the following: elevated serum bilirubin, AST/SGOT, ALT/SGPT) and 121 patients with moderate to severe renal impairment were treated with Clevidipine. An initial Clevidipine infusion rate of 1-2 mg/hour is appropriate in these patients.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Clevidipine sandbox in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Clevidipine sandbox in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information about the FDA-labeled indications and dosage information for children.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Clevidipine sandbox in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Clevidipine sandbox in pediatric patients.
Contraindications
Known Allergy
Clevidipine is contraindicated in patients with allergies to soybeans, soy products, eggs, or egg products.
Defective Lipid Metabolism
Clevidipine is contraindicated in patients with defective lipid metabolism such as pathologic hyperlipemia, lipoid nephrosis, or acute pancreatitis if it is accompanied by hyperlipidemia.
Severe Aortic Stenosis
Clevidipine is contraindicated in patients with severe aortic stenosis because afterload reduction can be expected to reduce myocardial oxygen delivery.
Warnings
Need for Aseptic Technique
Use aseptic technique and discard any unused product within 12 hours of stopper puncture [see Dosage and Administration (2.3)].
Hypotension and Reflex Tachycardia
Clevidipine may produce systemic hypotension and reflex tachycardia. If either occurs, decrease the dose of Clevidipine. There is limited experience with short-duration therapy with beta-blockers as a treatment for Clevidipine-induced tachycardia. Beta-blocker use for this purpose is not recommended.
Lipid Intake
Clevidipine contains approximately 0.2 g of lipid per mL (2.0 kcal). Lipid intake restrictions may be necessary for patients with significant disorders of lipid metabolism. For these patients, a reduction in the quantity of concurrently administered lipids may be necessary to compensate for the amount of lipid infused as part of the Clevidipine formulation.
Negative Inotropy
Dihydropyridine calcium channel blockers can produce negative inotropic effects and exacerbate heart failure. Monitor heart failure patients carefully.
Beta-Blocker Withdrawal
Clevidipine is not a beta-blocker, does not reduce heart rate, and gives no protection against the effects of abrupt beta-blocker withdrawal. Beta-blockers should be withdrawn only after a gradual reduction in dose.
Rebound Hypertension
Patients who receive prolonged Clevidipine infusions and are not transitioned to other antihypertensive therapies should be monitored for the possibility of rebound hypertension for at least 8 hours after the infusion is stopped.
Pheochromocytoma
There is no information to guide use of Clevidipine in treating hypertension associated with pheochromocytoma.
Adverse Reactions
Clinical Trials Experience
Clinical Trials Experience
Clevidipine clinical development included 19 studies, with 99 healthy subjects and 1307 hypertensive patients who received at least one dose of clevidipine (1406 total exposures). Clevidipine was evaluated in 15 studies in hypertensive patients: 1099 patients with perioperative hypertension, 126 with severe hypertension and 82 patients with essential hypertension.
The desired therapeutic response was achieved at doses of 4-6 mg/hour. Clevidipine was infused for <24 hours in the majority of patients (n=1199); it was infused as a continuous infusion in an additional 93 patients for durations between 24 and 72 hours.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Perioperative Hypertension
The placebo-controlled experience with Clevidipine in the perioperative setting was both small and brief (about 30 minutes). Table 2 shows treatment-emergent adverse reactions and the category of “any common adverse event” in ESCAPE-1 and ESCAPE-2 where the rate on Clevidipine exceeded the rate on placebo by at least 5% (common adverse reactions).
Three randomized, parallel, open-label studies called ECLIPSE, with longer exposure in cardiac surgery patients define the adverse reactions for patients with perioperative hypertension. Each ECLIPSE study compared Cleviprex (n=752) to an active comparator: nitroglycerin (NTG, n=278), sodium nitroprusside (SNP, n=283), or nicardipine (NIC, n=193). The pooled mean maximum dose in these studies was 10 mg/hour and the mean duration of treatment was 8 hours.
There were many adverse events associated with the operative procedure in the clinical studies of Cleviprex and relatively few plausibly related to the drugs used to lower blood pressure. Thus, the ability to differentiate the adverse event profile between treatments is limited. The adverse events observed within one hour of the end of the infusion were similar in patients who received Cleviprex and in those who received comparator agents. There was no adverse reaction that was more than 2% more common on Cleviprex than on the average of all comparators.
Serious Adverse Events and Discontinuation – Perioperative hypertension Studies The incidence of adverse events leading to study drug discontinuation in patients with perioperative hypertension receiving Cleviprex was 5.9% versus 3.2% for all active comparators. For patients receiving Cleviprex and all active comparators the incidence of serious adverse events within one hour of drug infusion discontinuation was similar.
Severe Hypertension
The adverse events for patients with severe hypertension are based on an uncontrolled study in patients with severe hypertension (VELOCITY, n=126).
The common adverse reactions for Cleviprex in severe hypertension included headache (6.3%), nausea (4.8%), and vomiting (3.2%). The incidence of adverse events leading to study drug discontinuation for Cleviprex in severe hypertension was 4.8%.
Less Common Adverse Reactions in Patients with Severe or Essential Hypertension
Adverse reactions that were reported in <1% of patients with severe or essential hypertension included: Cardiac: myocardial infarction, cardiac arrest Nervous system: syncope Respiratory: dyspnea
Postmarketing Experience
Because adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate reliably their frequency or to establish a causal relationship to drug exposure. The following adverse reactions have been identified during post-approval use of Clevidipine: increased blood triglycerides, ileus, hypersensitivity, hypotension, nausea, decreased oxygen saturation (possible pulmonary shunting) and reflex tachycardia.
Drug Interactions
No clinical drug interaction studies were conducted. Clevidipine and its major dihydropyridine metabolite do not have the potential for blocking or inducing any CYP enzyme.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C There are no adequate and well-controlled studies of Clevidipine use in pregnant women. In animal studies, clevidipine caused increases in maternal and fetal mortality and length of gestation. Clevidipine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. There was decreased fetal survival when pregnant rats and rabbits were treated with clevidipine during organogenesis at doses 0.7 times (on a body surface area basis) the maximum recommended human dose (MRHD) in rats and 2 times the MRHD in rabbits.
In pregnant rats dosed with clevidipine during late gestation and lactation, there were dose-related increases in maternal mortality, length of gestation and prolonged parturition at doses greater than or equal to 1/6 of the MRHD based on body surface area. When offspring of these dams were mated, they had a conception rate lower than that of controls. Clevidipine has been shown to cross the placenta in rats [see Nonclinical Toxicology (13.3)].
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Clevidipine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Clevidipine during labor and delivery.
Nursing Mothers
It is not known whether clevidipine is excreted in human milk. Because many drugs are excreted in human milk, consider possible infant exposure when Clevidipine is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of Clevidipine in children under 18 years of age have not been established.
Geriatic Use
Of the 1406 subjects (1307 with hypertension) treated with Clevidipine in clinical studies, 620 were ≥65 years of age and 232 were ≥75 years of age. No overall differences in safety or effectiveness were observed between these and younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, for an elderly patient doses should be titrated cautiously, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
Gender
There is no FDA guidance on the use of Clevidipine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Clevidipine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Clevidipine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Clevidipine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Clevidipine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Clevidipine in patients who are immunocompromised.
Administration and Monitoring
Administration
Clevidipine is intended for intravenous use. Titrate drug to achieve the desired blood pressure reduction. Individualize dosage depending on the blood pressure to be obtained and the response of the patient.
Instructions for Administration
Maintain aseptic technique while handling Clevidipine. Clevidipine is a single-use parenteral product. Do not use if contamination is suspected. Once the stopper is punctured, use within 12 hours and discard any unused portion. Clevidipine is supplied in sterile, pre-mixed, ready-to-use 50 mL or 100 mL vials. Invert vial gently several times before use to ensure uniformity of the emulsion prior to administration. Inspect parenteral drug products for particulate matter and discoloration prior to administration whenever solution and container permit. Administer Clevidipine using an infusion device allowing calibrated infusion rates. Commercially available standard plastic cannulae may be used to administer the infusion.Administer Clevidipine by a central line or a peripheral line. Clevidipine should not be administered in the same line as other medications. Clevidipine should not be diluted, but it can be administered with the following: Water for Injection, USP Sodium Chloride (0.9%) Injection, USP Dextrose (5%) Injection, USP Dextrose (5%) in Sodium Chloride (0.9%) Injection, USP Dextrose (5%) in Ringers Lactate Injection, USP Lactated Ringers Injection, USP 10% amino acid
Monitoring
Monitor blood pressure and heart rate continually during infusion, and then until vital signs are stable. Patients who receive prolonged Clevidipine infusions and are not transitioned to other antihypertensive therapies should be monitored for the possibility of rebound hypertension for at least 8 hours after the infusion is stopped. These patients may need follow-up adjustments in blood pressure control.
IV Compatibility
There is limited information about the IV Compatibility.
Overdosage
There has been no experience of overdosage in human clinical trials. In clinical trials, doses of Clevidipine up to 106 mg/hour or 1153 mg maximum total dose were administered. The expected major effects of overdose would be hypotension and reflex tachycardia.
Discontinuation of Clevidipine leads to a reduction in antihypertensive effects within 5 to 15 minutes [see Clinical Pharmacology (12.2)]. In case of suspected overdosage, Clevidipine should be discontinued immediately and the patient’s blood pressure should be supported.
Pharmacology
Mechanism of Action
Clevidipine is a dihydropyridine L-type calcium channel blocker. L-type calcium channels mediate the influx of calcium during depolarization in arterial smooth muscle. Experiments in anesthetized rats and dogs show that clevidipine reduces mean arterial blood pressure by decreasing systemic vascular resistance. Clevidipine does not reduce cardiac filling pressure (pre-load), confirming lack of effects on the venous capacitance vessels.
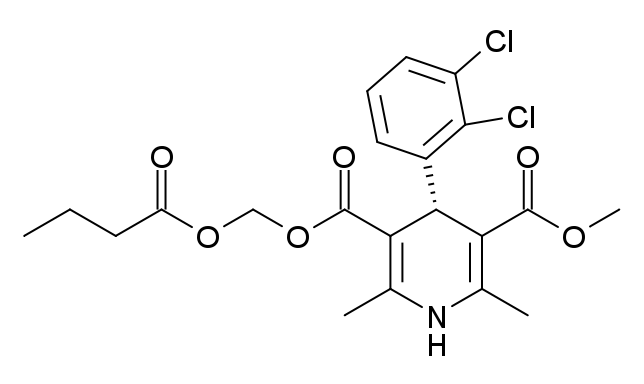
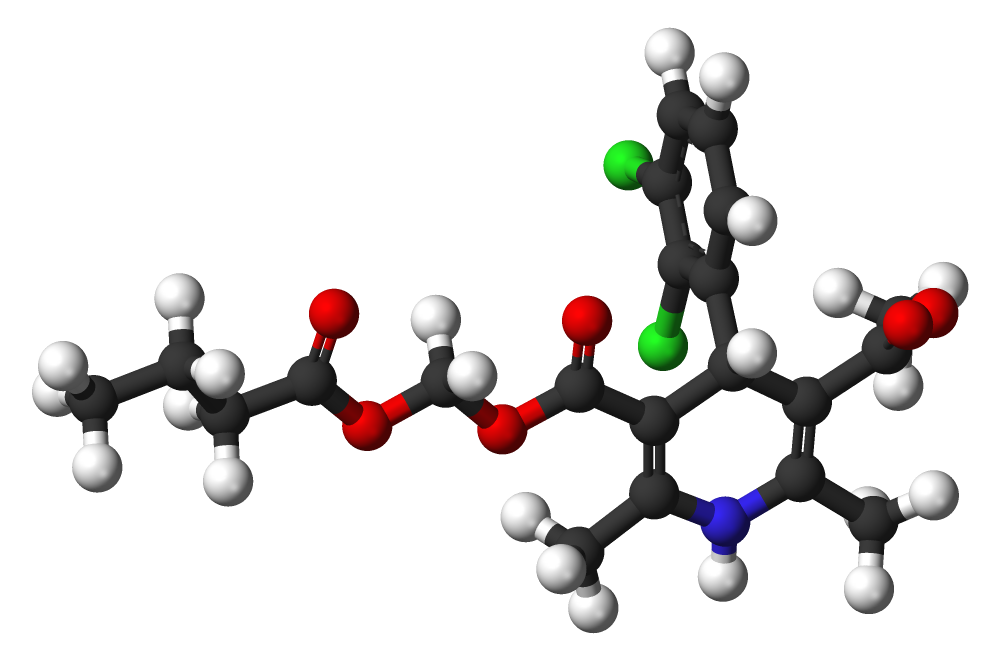
Structure
Clevidipine is a sterile, milky-white emulsion containing 0.5 mg/mL of clevidipine suitable for intravenous administration. Clevidipine is a dihydropyridine calcium channel blocker. Chemically, the active substance, clevidipine, is butyroxymethyl methyl 4-(2´,3´-dichlorophenyl)-1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate. It is a racemic mixture with a molecular weight of 456.3 g/mol. Each enantiomer has equipotent antihypertensive activity. The structure and formula are:
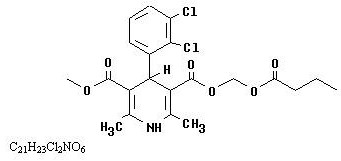
Clevidipine is practically insoluble in water and is formulated in an oil-in-water emulsion. In addition to the active ingredient, clevidipine, Clevidipine contains soybean oil (200 mg/mL), glycerin (22.5 mg/mL), purified egg yolk phospholipids (12 mg/mL), oleic acid (0.3 mg/mL), disodium edetate (0.05 mg/mL), and sodium hydroxide to adjust pH. Clevidipine has a pH of 6.0 – 8.0 and is a ready-to-use emulsion.
Pharmacodynamics
Clevidipine is titrated to the desired reduction in blood pressure. The effect of Clevidipine appears to plateau at approximately 25% of baseline systolic pressure. The infusion rate for which half the maximal effect is observed is approximately 10 mg/hour.
Onset of Effect: In the perioperative patient population, Clevidipine produces a 4-5% reduction in systolic blood pressure within 2-4 minutes after starting a 0.4 mcg/kg/min infusion (approximately 1-2 mg/hr).
Maintenance of Effect: In studies up to 72 hours of continuous infusion, there was no evidence of tolerance or hysteresis.
Offset of Effect: In most patients, full recovery of blood pressure is achieved in 5-15 minutes after the infusion is stopped.
In studies up to 72 hours of continuous infusion, in patients that were not transitioned to other antihypertensive therapies, there was some evidence of rebound hypertension following Clevidipine discontinuation.
Hemodynamics: Clevidipine causes a dose-dependent decrease in systemic vascular resistance.
Heart Rate: An increase in heart rate is a normal response to vasodilation and decrease in blood pressure; in some patients these increases in heart rate may be pronounced[see Warnings and Precautions (5.2)].
Electrophysiologic Effects: In healthy volunteers, clevidipine or its major carboxylic acid metabolite, at therapeutic and supratherapeutic concentrations (approximately 2.8 times steady-state), did not prolong cardiac repolarization.
Pharmacokinetics
Clevidipine is rapidly distributed and metabolized resulting in a very short half life. The arterial blood concentration of clevidipine declines in a multi-phasic pattern following termination of the infusion. The initial phase half-life is approximately 1 minute, and accounts for 85-90% of clevidipine elimination. The terminal half-life is approximately 15 minutes.
Distribution: Clevidipine is >99.5% bound to proteins in plasma at 37°C. The steady-state volume of distribution was determined to be 0.17 L/kg in arterial blood.
Metabolism and Elimination: Clevidipine is rapidly metabolized by hydrolysis of the ester linkage, primarily by esterases in the blood and extravascular tissues, making its elimination unlikely to be affected by hepatic or renal dysfunction. The primary metabolites are the carboxylic acid metabolite and formaldehyde formed by hydrolysis of the ester group. The carboxylic acid metabolite is inactive as an antihypertensive. This metabolite is further metabolized by glucuronidation or oxidation to the corresponding pyridine derivative. The clearance of the primary dihydropyridine metabolite is 0.03 L/h/kg and the terminal half life is approximately 9 hours.
In vitro studies show that clevidipine and its metabolite at the concentrations achieved in clinical practice will not inhibit or induce any CYP enzyme.
In a clinical study with radiolabeled clevidipine, 83% of the drug was excreted in urine and feces. The major fraction, 63-74% is excreted in the urine, 7-22% in the feces. More than 90% of the recovered radioactivity is excreted within the first 72 hours of collection.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
Clevidipine displayed positive genotoxic potential in vitro in the Ames test, mouse lymphoma thymidine kinase locus assay, and chromosomal aberration assay but not in vivo in the mouse micronucleus test. Formaldehyde, a metabolite of clevidipine, a known genotoxicant in vitro and a probable human carcinogen, appears to be at least partially responsible for the positive in vitro results. Long-term studies for evaluation of carcinogenic potential have not been performed with clevidipine due to the intended short-term duration of human use. There were no adverse effects on fertility or mating behavior of male rats at clevidipine doses of up to 55 mg/kg/day, approximately equivalent to the maximum recommended human dose (MRHD) of 504 mg/day (21 mg/hour x 24 hours) on a body surface area basis. Female rats demonstrated pseudopregnancy and changes in estrus cycle at doses as low as 13 mg/kg/day (about 1/4th the MRHD); however, doses of up to 55 mg/kg/day did not affect mating performance or fertility.
Developmental Toxicology
When pregnant rats were dosed with clevidipine during late gestation and lactation, there were dose-related increases in mortality, length of gestation and prolonged parturition at dose levels as low as 13 mg/kg/day (about 1/4th the maximum recommended human dose of 504 mg/day (21 mg/hour x 24 hours) on a body surface area basis). When offspring of these dams were mated, they had a conception rate lower than that of controls. Clevidipine crosses the placental membrane in this species and doses of 35 or more mg/kg/day (about 0.7 times the MRHD) administered during organogenesis adversely affected fetal survival. Fetal survival was also adversely affected when pregnant rabbits were treated during organogenesis with 55 mg/kg/day (about twice the MRHD on a body surface area basis).
Clinical Studies
Perioperative Hypertension
Clevidipine was evaluated in two double-blind, randomized, parallel, placebo-controlled, multicenter trials of cardiac surgery patients—pre-operative use in ESCAPE-1 (n=105) and post-operative use in ESCAPE-2 (n=110). Patients were undergoing coronary artery bypass grafting, with or without valve replacement. Inclusion in ESCAPE-1 required a systolic pressure ≥160 mmHg. In ESCAPE-2, the entry criterion was systolic pressure of ≥140 mmHg within 4 hours of the completed surgery. The mean baseline blood pressure was 178/77 mmHg in ESCAPE -1 and 150/71 mmHg in ESCAPE 2. The population of both studies included 27% females and 47% patients older than age 65.
Clevidipine was infused in ESCAPE-1 preoperatively for 30 minutes, until treatment failure, or until induction of anesthesia, whichever came first. Clevidipine was infused in ESCAPE-2 postoperatively for a minimum of 30 minutes unless alternative therapy was required. The maximum infusion time allowed in the ESCAPE studies was 60 minutes.
In both studies infusion of Clevidipine was started at a dose of 1- 2 mg/hour and was titrated upwards, as tolerated, in doubling increments every 90 seconds up to an infusion rate of 16 mg/hour in order to achieve the desired blood pressure-lowering effect. At doses above 16 mg/hour increments were 7 mg/hour. The average Clevidipine infusion rate in ESCAPE-1 was 15.3 mg/hour and in ESCAPE-2 it was 5.1 mg/hour. The mean duration of exposure in the same ESCAPE studies was 30 minutes for the Clevidipine treated patients.
Approximately 4% of Clevidipine-treated subjects in ESCAPE-1 and 41% in ESCAPE-2 were on concomitant vasodilators during the first 30 minutes of Clevidipine administration.
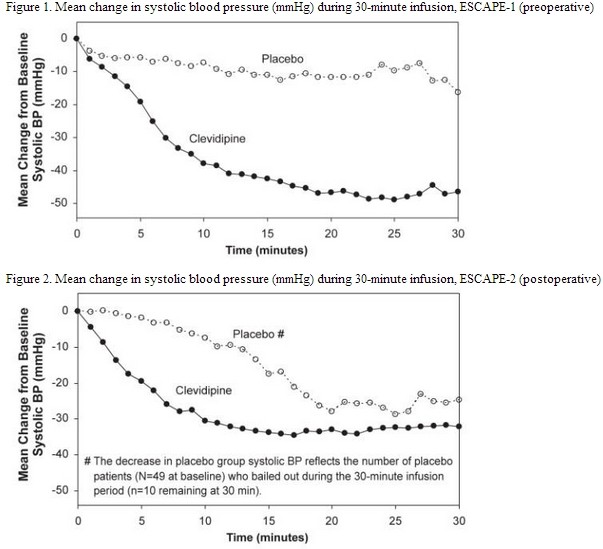
Clevidipine lowered blood pressure within 2-4 minutes. The change in systolic blood pressure over 30 minutes for ESCAPE-1 (preoperative) and ESCAPE-2 (postoperative) are shown in Figure 1 and 2.
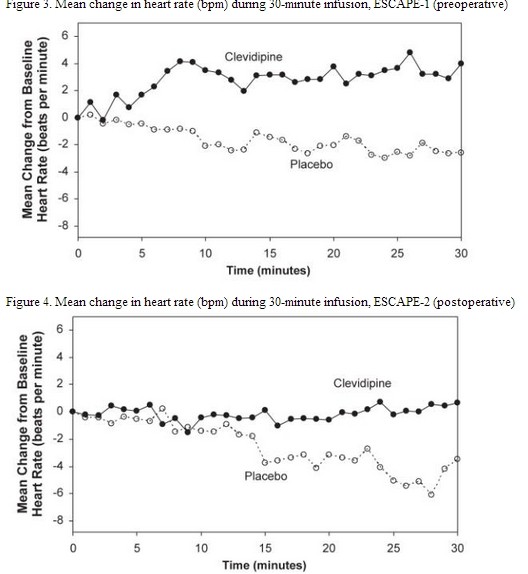
The change in heart rate over 30 minutes for ESCAPE-1 (preoperative) and ESCAPE-2 (postoperative) are shown in Figure 3 and 4.
In three Phase 3 open-label clinical trials (ECLIPSE), 1512 patients were randomized to receive Clevidipine, nitroglycerin (perioperative hypertension), sodium nitroprusside (perioperative hypertension), or nicardipine (postoperative hypertension), for the treatment of hypertension in cardiac surgery. The mean exposure in the ECLIPSE studies was 8 hours at 4.5 mg/hour for the 752 patients who were treated with Clevidipine. Blood pressure control was assessed by measuring the magnitude and duration of SBP excursions outside the predefined pre- and post-operative SBP target range of 75-145 mmHg and the predefined intra-operative SBP range of 65-135 mmHg. In general, blood pressure control was similar with the four treatments.
Severe Hypertension
Clevidipine was evaluated in an open-label, uncontrolled clinical trial (VELOCITY) in 126 patients with severe hypertension (SBP >180 mmHg or diastolic blood pressure [DBP] >115 mmHg). Clevidipine infusion was initiated at 2 mg/hour and up-titrated every 3 minutes, doubling up to a maximum dose of 32 mg/hour as required to achieve a prespecified target blood pressure range within 30 minutes (primary endpoint). The transition to oral antihypertensive therapy was assessed for up to 6 hours following cessation of Clevidipine infusion.
The blood pressure effect in this study is shown in Figure 5. The average infusion rate was 9.5 mg/hour. The mean duration of Clevidipine exposure was 21 hours.
Figure 5. Mean percent change in SBP (%) during the first 30 minutes of infusion, VELOCITY (severe hypertension)
How Supplied
Clevidipine (clevidipine) injectable emulsion is supplied as a sterile, milky white liquid emulsion product in single-use 50 mL or 100 mL glass vials at a concentration of 0.5 mg/mL of clevidipine.
NDC 65293-005-50: 50 mL vial NDC 65293-005-00: 100 mL vial
Storage
Leave vials in cartons until use. Clevidipine is photosensitive and storage in cartons protects against photodegradation. Protection from light during administration is not required.
Store vials refrigerated at 2-8°C (36-46°F). Do not freeze. Vials in cartons may be transferred to 25°C (77°F, USP controlled room temperature) for a period not to exceed 2 months. Upon transfer to room temperature, mark vials in cartons “This product was removed from the refrigerator on _/_/_ date. It must be used or discarded 2 months after this date or the labeled expiration date (whichever date comes first).” Do not return to refrigerated storage after beginning room temperature storage.
Images
Drug Images
{{#ask: Page Name::Clevidipine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Clevidipine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Advise patients with underlying hypertension that they require continued follow up for their medical condition, and, if applicable, encourage patients to continue taking their oral antihypertensive medication(s) as directed.
- Advise patients to contact a healthcare professional immediately for any of the following signs of a new hypertensive emergency: neurological symptoms, visual changes, or evidence of congestive heart failure.
Precautions with Alcohol
Alcohol-Clevidipine interaction has not been established. Talk to your doctor regarding the effects of taking alcohol with this medication.
Brand Names
Cleviprex
Look-Alike Drug Names
There is limited information about the look-alike drugs.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Clevidipine |Label Name=clevidipine08.jpg
}}
{{#subobject:
|Label Page=Clevidipine |Label Name=clevidipine09.jpg
}}
{{#subobject:
|Label Page=Clevidipine |Label Name=clevidipine10.jpg
}}
{{#subobject:
|Label Page=Clevidipine |Label Name=clevidipine11.jpg
}}
{{#subobject:
|Label Page=Clevidipine |Label Name=Cleviprex panel 01.png
}}