Bromopyruvic acid: Difference between revisions
Gerald Chi (talk | contribs) mNo edit summary |
Gerald Chi (talk | contribs) mNo edit summary |
||
| Line 1: | Line 1: | ||
{{Chembox | {{Chembox | ||
|ImageFile=Bromopyruvic_acid.png | |ImageFile=Bromopyruvic_acid.png | ||
|ImageSize=180px | |ImageSize=180px | ||
Revision as of 14:47, 8 April 2015
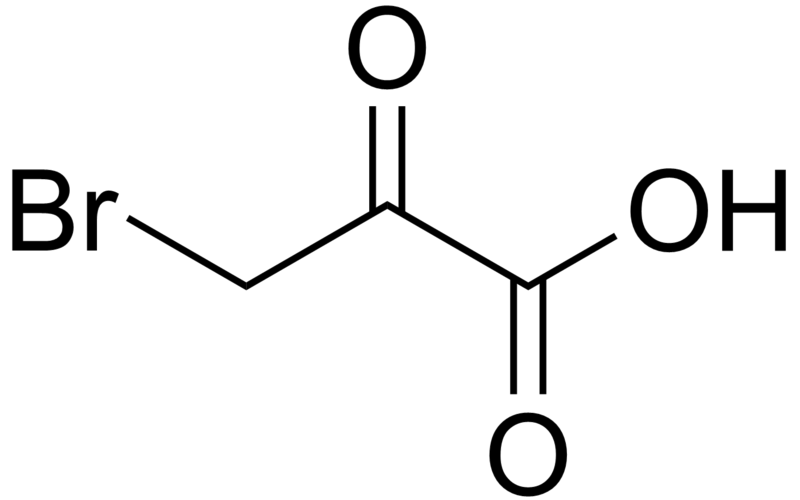
| |
| Names | |
|---|---|
| IUPAC name
3-Bromo-2-oxopropanoic acid
| |
| Other names
Bromopyruvate
3-Bromopyruvic acid 3-Bromopyruvate | |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | Lua error in Module:Wikidata at line 879: attempt to index field 'wikibase' (a nil value). Lua error in Module:Wikidata at line 879: attempt to index field 'wikibase' (a nil value). |
PubChem CID
|
|
| |
| Properties | |
| C3H3BrO3 | |
| Molar mass | 166.95812 |
| Hazards | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Bromopyruvic acid, or bromopyruvate, is a synthetic brominated derivative of pyruvic acid. It is being studied as a potential treatment for certain types of cancer. Initial studies in laboratory animals by Young Hee Ko at Johns Hopkins University showed that bromopyruvic acid is effective at eliminating aggressive liver tumors.[1]
According to the Warburg hypothesis, unlike normal tissues that derive most of their energy in the form of adenosine triphosphate (ATP) by metabolizing glucose in the mitochondria, aggressive cancers obtain much of their ATP by metabolizing glucose directly to lactic acid.[2] The mechanism of action of bromopyruvic acid involves interruption of this latter process by the inhibition of the enzyme hexokinase II, due to the fact that the bromopyruvic acid is similar in chemical structure to lactic acid.[1]
Further study demonstated that when 19 rats with advanced cancers (generated by seeding with hepatocellular carcinoma cells) were treated with bromopyruvic acid, the cancers were eradicated without apparent toxicity or recurrence in all treated animals.[3] Another study reported that intraarterial delivery of bromopyruvic acid directly to the site of a tumor, followed by systemic delivery only when necessary, represents a new strategy for stopping the growth of liver and other cancers, while minimizing toxic side effects.[4]
While pre-clinical studies have been promising, human clinical trials to study the effectiveness of bromopyruvic acid have not yet begun. One obstacle to further development of bromopyruvic acid as a pharmaceutical treatment for cancer is that since bromopyruvic acid is not a novel chemical compound, it is not patentable.[5] Without the exclusivity allowed by patent protection, there is little prospect for recovery of the high financial investment needed for conducting the clinical trials required by worldwide regulatory agencies for approval of pharmaceutical drugs.
References
- ↑ 1.0 1.1 Ko YH, Pedersen PL, Geschwind JF (2001). "Glucose catabolism in the rabbit VX2 tumor model for liver cancer: characterization and targeting hexokinase". Cancer Lett. 173 (1): 83–91. PMID 11578813.
- ↑ Pedersen PL (2007). "Warburg, me and Hexokinase 2: Multiple discoveries of key molecular events underlying one of cancers' most common phenotypes, the "Warburg Effect", i.e., elevated glycolysis in the presence of oxygen". doi:10.1007/s10863-007-9094-x. PMID 17879147.
- ↑ Ko YH, Smith BL, Wang Y; et al. (2004). "Advanced cancers: eradication in all cases using 3-bromopyruvate therapy to deplete ATP". Biochem. Biophys. Res. Commun. 324 (1): 269–75. doi:10.1016/j.bbrc.2004.09.047. PMID 15465013.
- ↑ Geschwind JF, Ko YH, Torbenson MS, Magee C, Pedersen PL (2002). "Novel therapy for liver cancer: direct intraarterial injection of a potent inhibitor of ATP production". Cancer Res. 62 (14): 3909–13. PMID 12124317.
- ↑ Young researcher stalks cancer
Vali M, Liapi E, Kowalski J, Hong K, Khwaja A, Torbenson MS, Georgiades C, Geschwind JF.
Intraarterial therapy with a new potent inhibitor of tumor metabolism (3-bromopyruvate): identification of therapeutic dose and method of injection in an animal model of liver cancer.
J Vasc Interv Radiol. 2007 Jan;18(1 Pt 1):95-101. PMID: 17296709
[[1]]
External links
- The cancer cell's "power plants" as promising therapeutic targets: An overview, by Peter Pederson
- Glycolytic enzyme inhibitors as novel anti-cancer drugs, James C.K. Lai et al., Idaho State, June 2007
- Pyruvate in Cancer Prevention and Treatment
- Energy Blocker Kills Big Tumors in Rats
- 3-Bromopyruvic Acid As a Potent Anticancer Agent Delivered Intraarterially
- Pages with script errors
- CS1 maint: Multiple names: authors list
- CS1 maint: Explicit use of et al.
- Articles without InChI source
- Chemical pages without ChemSpiderID
- Articles without EBI source
- Articles without KEGG source
- Articles without UNII source
- ECHA InfoCard ID from Wikidata
- Chembox having DSD data
- Chemical articles with unknown parameter in Chembox
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Alkylating agents
- Carboxylic acids
- Organobromides