Atovaquone and proguanil hydrochloride
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Atovaquone and proguanil hydrochloride is a {{{drugClass}}} that is FDA approved for the treatment of {{{indication}}}. Common adverse reactions include .
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
Prevention of Malaria
- Atovaquone and proguanil hydrochloride tabletsare indicated for the prophylaxis of Plasmodium falciparum malaria, including in areas where chloroquine resistance has been reported.
Treatment of Malaria
- Atovaquone and proguanil hydrochloride tabletsare indicated for the treatment of acute, uncomplicated P. falciparum malaria. Atovaquone and proguanil hydrochloride tablets have been shown to be effective in regions where the drugs chloroquine, halofantrine, mefloquine, and amodiaquine may have unacceptable failure rates,
presumably due to drug resistance.
Dosage
- The daily dose should be taken at the same time each day with food or a milky drink. In the event of vomiting within 1 hour after dosing, a repeat dose should be taken.
Prevention of Malaria
- Start prophylactic treatment withatovaquone and proguanil hydrochloride tablets 1 or 2 days before entering a malaria-endemic area and continue daily during the stay and for 7 days after return.
- Adults: One atovaquone and proguanil hydrochloride tablet(adult strength = 250 mg atovaquone/100 mg proguanil hydrochloride) per day.
Treatment of Acute Malaria
- Adults: Fouratovaquone and proguanil hydrochloride tablets(adult strength; total daily dose 1 g atovaquone/400 mg proguanil hydrochloride) as a single daily dose for 3 consecutive day.
Renal Impairment
- Do not useatovaquone and proguanil hydrochloride tablets for malaria prophylaxis in patients with severe renal impairment (creatinine clearance <30 mL/min). Use with caution for the treatment of malaria in patients with severe renal impairment, only if the benefits of the 3 day treatment regimen outweigh the potential risks associated with increased drug exposure. No dosage adjustments are needed in patients with mild (creatinine clearance 50 to 80 mL/min) or moderate (creatinine clearance 30 to 50 mL/min) renal impairment.
DOSAGE FORMS AND STRENGTHS
- Each atovaquone and proguanil hydrochloride tablets (adult strength) contains 250 mg atovaquone USP and 100 mg proguanil hydrochloride USP. Atovaquone and proguanil hydrochloride tablets are pinkish brown to brown colored, circular, biconvex beveled edge, film-coated tablets with ‘404’ debossed on one side and ‘G’ debossed on the other side.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Atovaquone and proguanil hydrochloride in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Atovaquone and proguanil hydrochloride in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Atovaquone and proguanil hydrochloride in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Atovaquone and proguanil hydrochloride in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Atovaquone and proguanil hydrochloride in pediatric patients.
Contraindications
Hypersensitivity
- Atovaquone and proguanil hydrochloride is contraindicated in individuals with known hypersensitivity reactions (e.g., anaphylaxis, erythema multiforme or Stevens-Johnson syndrome, angioedema, vasculitis) to atovaquone or proguanil hydrochloride or any component of the formulation.
Severe Renal Impairment
- Atovaquone and proguanil hydrochloride is contraindicated for prophylaxis of P. falciparum malaria in patients with severe renal impairment (creatinine clearance <30 mL/min) because of pancytopenia in patients with severe renal impairment treated with proguanil.
Warnings
Vomiting and Diarrhea
- Absorption of atovaquone may be reduced in patients with diarrhea or vomiting. If atovaquone and proguanil hydrochloride is used in patients who are vomiting, parasitemia should be closely monitored and the use of an antiemetic considered. [See Dosage and Administration (2).] Vomiting occurred in up to 19% of pediatric patients given treatment doses of atovaquone and proguanil hydrochloride. In the controlled clinical trials, 15.3% of adults received an antiemetic when they received atovaquone/proguanil and 98.3% of these patients were successfully treated. In patients with severe or persistent diarrhea or vomiting, alternative antimalarial therapy may be required.
Relapse of Infection
- In mixed P. falciparum and Plasmodium vivax infections, P. vivax parasite relapse occurred commonly when patients were treated with atovaquone and proguanil hydrochloride alone.
- In the event of recrudescent P. falciparum infections after treatment with atovaquone and proguanil hydrochloride or failure of chemoprophylaxis with atovaquone and proguanil hydrochloride, patients should be treated with a different blood schizonticide.
Hepatotoxicity
- Elevated liver laboratory tests and cases of hepatitis and hepatic failure requiring liver transplantation have been reported with prophylactic use of atovaquone and proguanil hydrochloride.
Severe or Complicated Malaria
- Atovaquone and proguanil hydrochloride has not been evaluated for the treatment of cerebral malaria or other severe manifestations of complicated malaria, including hyperparasitemia, pulmonary edema, or renal failure. Patients with severe malaria are not candidates for oral therapy
Adverse Reactions
Clinical Trials Experience
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- Because the tablets contain Atovaquone and proguanil hydrochloride, the type and severity of adverse reactions associated with each of the compounds may be expected. The lower prophylactic doses of atovaquone and proguanil hydrochloride were better tolerated than the higher treatment doses.
- Prophylaxis of P. falciparum Malaria: In 3 clinical trials (2 of which were placebo-controlled) 381 adults (mean age 31 years) received atovaquone and proguanil hydrochloride for the prophylaxis of malaria; the majority of adults were black (90%) and 79% were male. In a clinical trial for the prophylaxis of malaria, 125 pediatric patients (mean age 9 years) received atovaquone and proguanil hydrochloride ; all subjects were black and 52% were male. Adverse experiences reported in adults and pediatric patients, considered attributable to therapy, occurred in similar proportions of subjects receiving atovaquone and proguanil hydrochloride or placebo in all studies. Prophylaxis with atovaquone and proguanil hydrochloride was discontinued prematurely due to a treatment-related adverse experience in 3 of 381 (0.8%) adults and 0 of 125 pediatric patients.
- In a placebo-controlled study of malaria prophylaxis with atovaquone and proguanil hydrochloride involving 330 pediatric patients (aged 4 to 14 years) in Gabon, a malaria-endemic area, the safety profile of atovaquone and proguanil hydrochloride was consistent with that observed in the earlier prophylactic studies in adults and pediatric patients. The most common treatment-emergent adverse events with atovaquone and proguanil hydrochloride were abdominal pain (13%), headache (13%), and cough (10%). Abdominal pain (13% vs. 8%) and vomiting (5% vs. 3%) were reported more often with atovaquone and proguanil hydrochloride than with placebo. No patient withdrew from the study due to an adverse experience with atovaquone and proguanil hydrochloride. No routine laboratory data were obtained during this study.
- Non-immune travelers visiting a malaria-endemic area received MALARONE (n = 1,004) for prophylaxis of malaria in 2 active-controlled clinical trials. In one study (n = 493), the mean age of subjects was 33 years and 53% were male; 90% of subjects were white, 6% of subjects were black and the remaining were of other racial/ethnic groups. In the other study (n = 511), the mean age of subjects was 36 years and 51% were female; the majority of subjects (97%) were white. Adverse experiences occurred in a similar or lower proportion of subjects receiving atovaquone and proguanil hydrochloride tablets than an active comparator (Table 1). Fewer neuropsychiatric adverse experiences occurred in subjects who received MALARONE than mefloquine. Fewer gastrointestinal adverse experiences occurred in subjects receiving atovaquone and proguanil hydrochloride tablets than chloroquine/proguanil. Compared with active comparator drugs, subjects receiving atovaquone and proguanil hydrochloride tablets had fewer adverse experiences overall that were attributed to prophylactic therapy (Table 1). Prophylaxis with MALARONE was discontinued prematurely due to a treatment-related adverse experience in 7 of 1,004 travelers.

- In a third active-controlled study, atovaquone and proguanil hydrochloride (n = 110) was compared with chloroquine/proguanil (n = 111) for the prophylaxis of malaria in 221 non-immune pediatric patients (2 to 17 years of age). The mean duration of exposure was 23 days for atovaquone and proguanil hydrochloride , 46 days for chloroquine, and 43 days for proguanil, reflecting the different recommended dosage regimens for these products. Fewer patients treated with atovaquone and proguanil hydrochloride reported abdominal pain (2% vs. 7%) or nausea (<1% vs. 7%) than children who received chloroquine/proguanil. Oral ulceration (2% vs. 2%), vivid dreams (2% vs. <1%), and blurred vision (0% vs. 2%) occurred in similar proportions of patients receiving either atovaquone and proguanil hydrochloride or chloroquine/proguanil, respectively. Two patients discontinued prophylaxis with chloroquine/proguanil due to adverse events, while none of those receiving atovaquone and proguanil hydrochloride discontinued due to adverse events.
- Treatment of Acute, Uncomplicated P. falciparum Malaria: In 7 controlled trials, 436 adolescents and adults received atovaquone and proguanil hydrochloride for treatment of acute, uncomplicated P. falciparum malaria. The range of mean ages of subjects was 26 to 29 years; 79% of subjects were male. In these studies, 48% of subjects were classified as other racial/ethnic groups, primarily Asian; 42% of subjects were black and the remaining subjects were white. Attributable adverse experiences that occurred in ≥5% of patients were abdominal pain (17%), nausea (12%), vomiting (12%), headache (10%), diarrhea (8%), asthenia (8%), anorexia (5%), and dizziness (5%). Treatment was discontinued prematurely due to an adverse experience in 4 of 436 (0.9%) adolescents and adults treated with atovaquone and proguanil hydrochloride .
- In 2 controlled trials, 116 pediatric patients (weighing 11 to 40 kg) (mean age 7 years) received atovaquone and proguanil hydrochloride for the treatment of malaria. The majority of subjects were black (72%); 28% were of other racial/ethnic groups, primarily Asian. Attributable adverse experiences that occurred in ≥5% of patients were vomiting (10%) and pruritus (6%). Vomiting occurred in 43 of 319 (13%) pediatric patients who did not have symptomatic malaria but were given treatment doses of atovaquone and proguanil hydrochloride for 3 days in a clinical trial. The design of this clinical trial required that any patient who vomited be withdrawn from the trial. Among pediatric patients with symptomatic malaria treated with MALARONE, treatment was discontinued prematurely due to an adverse experience in 1 of 116 (0.9%).
- In a study of 100 pediatric patients (5 to <11 kg body weight) who received atovaquone and proguanil hydrochloride for the treatment of uncomplicated P. falciparum malaria, only diarrhea (6%) occurred in ≥5% of patients as an adverse experience attributable to atovaquone and proguanil hydrochloride. In 3 patients (3%), treatment was discontinued prematurely due to an adverse experience.
- Abnormalities in laboratory tests reported in clinical trials were limited to elevations of transaminases in malaria patients being treated with atovaquone and proguanil hydrochloride . The frequency of these abnormalities varied substantially across trials of treatment and were not observed in the randomized portions of the prophylaxis trials.
- One active-controlled trial evaluated the treatment of malaria in Thai adults (n = 182); the mean age of subjects was 26 years (range 15 to 63 years); 80% of subjects were male. Early elevations of ALT and AST occurred more frequently in patients treated with atovaquone and proguanil hydrochloride (n = 91) compared to patients treated with an active control, mefloquine (n = 91). On Day 7, rates of elevated ALT and AST with atovaquone and proguanil hydrochloride and mefloquine (for patients who had normal baseline levels of these clinical laboratory parameters) were ALT 26.7% vs. 15.6%; AST 16.9% vs. 8.6%, respectively. By Day 14 of this 28-day study, the frequency of transaminase elevations equalized across the 2 groups
Postmarketing Experience
- In addition to adverse events reported from clinical trials, the following events have been identified during postmarketing use of atovaquone and proguanil hydrochloride . Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to atovaquone and proguanil hydrochloride.
- Blood and Lymphatic System Disorders: Neutropenia and anemia. Pancytopenia in patients with severe renal impairment treated with proguanil.
- Immune System Disorders: Allergic reactions including anaphylaxis, angioedema, and urticaria, and vasculitis.
- Nervous System Disorders: Seizures and psychotic events (such as hallucinations); however, a causal relationship has not been established.
- Gastrointestinal Disorders: Stomatitis.
- Hepatobiliary Disorders: Elevated liver laboratory tests, hepatitis, cholestasis; hepatic failure requiring transplant has been reported.
- Skin and Subcutaneous Tissue Disorders: Photosensitivity, rash, erythema multiforme, and Stevens-Johnson syndrome
Drug Interactions
Rifampin/Rifabutin
- Concomitant administration of rifampin or rifabutin is known to reduce atovaquone concentrations. The concomitant administration of atovaquone and proguanil hydrochloride and rifampin or rifabutin is not recommended.
Anticoagulants
- Proguanil may potentiate the anticoagulant effect of warfarin and other coumarin-based anticoagulants. The mechanism of this potential drug interaction has not been established. Caution is advised when initiating or withdrawing malaria prophylaxis or treatment with atovaquone and proguanil hydrochloride in patients on continuous treatment with coumarin-based anticoagulants. When these products are administered concomitantly, coagulation tests should be closely monitored.
Tetracycline
- Concomitant treatment with tetracycline has been associated with a reduction in plasma concentrations of atovaquone. Parasitemia should be closely monitored in patients receiving tetracycline.
Metoclopramide
- While antiemetics may be indicated for patients receiving atovaquone and proguanil hydrochloride, metoclopramide may reduce the bioavailability of atovaquone and should be used only if other antiemetics are not available.
Indinavir
- Concomitant administration of atovaquone and indinavir did not result in any change in the steady-state AUC and Cmax of indinavir but resulted in a decrease in the Ctrough of indinavir. Caution should be exercised when prescribing atovaquone with indinavir due to the decrease in trough concentrations of indinavir
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Pregnancy Category C
- Atovaquone: Atovaquone was not teratogenic and did not cause reproductive toxicity in rats at doses up to 1,000 mg/kg/day corresponding to maternal plasma concentrations up to 7.3 times the estimated human exposure during treatment of malaria based on AUC. In rabbits, atovaquone caused adverse fetal effects and maternal toxicity at a dose of 1,200 mg/kg/day corresponding to plasma concentrations that were approximately 1.3 times the estimated human exposure during treatment of malaria based on AUC. Adverse fetal effects in rabbits, including decreased fetal body lengths and increased early resorptions and post-implantation losses, were observed only in the presence of maternal toxicity.
- In a pre- and post-natal study in rats, atovaquone did not produce adverse effects in offspring at doses up to 1,000 mg/kg/day corresponding to AUC exposures of approximately 7.3 times the estimated human exposure during treatment of malaria.
- Proguanil: A pre- and post-natal study in Sprague-Dawley rats revealed no adverse effects at doses up to 16 mg/kg/day of proguanil hydrochloride (up to 0.04-times the average human exposure based on AUC). Pre- and post-natal studies of proguanil in animals at exposures similar to or greater than those observed in humans have not been conducted.
- Atovaquone and Proguanil: The combination of atovaquone and proguanil hydrochloride was not teratogenic in pregnant rats at atovaquone:proguanil hydrochloride (50:20 mg/kg/day) corresponding to plasma concentrations up to 1.7 and 0.1 times, respectively, the estimated human exposure during treatment of malaria based on AUC. In pregnant rabbits, the combination of atovaquone and proguanil hydrochloride was not teratogenic or embryotoxic to rabbit fetuses at atovaquone:proguanil hydrochloride (100:40 mg/kg/day) corresponding to plasma concentrations of approximately 0.3 and 0.5 times, respectively, the estimated human exposure during treatment of malaria based on AUC.
- There are no adequate and well-controlled studies of atovaquone and/or proguanil hydrochloride in pregnant women. Atovaquone and proguanil hydrochloride should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Falciparum malaria carries a higher risk of morbidity and mortality in pregnant women than in the general population. Maternal death and fetal loss are both known complications of falciparum malaria in pregnancy. In pregnant women who must travel to malaria-endemic areas, personal protection against mosquito bites should always be employed in addition to antimalarials. [See Patient Counseling Information (17).]
- The proguanil component of atovaquone and proguanil hydrochloride acts by inhibiting the parasitic dihydrofolate reductase. However, there are no clinical data indicating that folate supplementation diminishes drug efficacy. For women of childbearing age receiving folate supplements to prevent neural tube birth defects, such supplements may be continued while taking atovaquone and proguanil hydrochloride.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Atovaquone and proguanil hydrochloride in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Atovaquone and proguanil hydrochloride during labor and delivery.
Nursing Mothers
- It is not known whether atovaquone is excreted into human milk. In a rat study, atovaquone concentrations in the milk were 30% of the concurrent atovaquone concentrations in the maternal plasma.
- Proguanil is excreted into human milk in small quantities.
- Caution should be exercised when atovaquone and proguanil hydrochloride is administered to a nursing woman.
Pediatric Use
- Prophylaxis of Malaria: Safety and effectiveness have not been established in pediatric patients who weigh less than 11 kg. The efficacy and safety of atovaquone and proguanil hydrochloride have been established for the prophylaxis of malaria in controlled trials involving weighing pediatric patients 11 kg or more .
- Treatment of Malaria: Safety and effectiveness have not been established in pediatric patients who weigh less than 5 kg. The efficacy and safety of atovaquone and proguanil hydrochloride for the treatment of malaria have been established in controlled trials involving pediatric patients weighing 5 kg or more.
Geriatic Use
There is no FDA guidance on the use of Atovaquone and proguanil hydrochloride with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Atovaquone and proguanil hydrochloride with respect to specific gender populations.
Race
There is no FDA guidance on the use of Atovaquone and proguanil hydrochloride with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Atovaquone and proguanil hydrochloride in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Atovaquone and proguanil hydrochloride in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Atovaquone and proguanil hydrochloride in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Atovaquone and proguanil hydrochloride in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Atovaquone and proguanil hydrochloride Administration in the drug label.
Monitoring
There is limited information regarding Atovaquone and proguanil hydrochloride Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Atovaquone and proguanil hydrochloride and IV administrations.
Overdosage
- There is no information on overdoses of atovaquone and proguanil hydrochloride substantially higher than the doses recommended for treatment.
- There is no known antidote for atovaquone, and it is currently unknown if atovaquone is dialyzable. Overdoses up to 31,500 mg of atovaquone have been reported. In one such patient who also took an unspecified dose of dapsone, methemoglobinemia occurred. Rash has also been reported after overdose.
- Overdoses of proguanil hydrochloride as large as 1,500 mg have been followed by complete recovery, and doses as high as 700 mg twice daily have been taken for over 2 weeks without serious toxicity. Adverse experiences occasionally associated with proguanil hydrochloride doses of 100 to 200 mg/day, such as epigastric discomfort and vomiting would be likely to occur with overdose. There are also reports of reversible hair loss and scaling of the skin on the palms and/or soles, reversible aphthous ulceration, and hematologic side effects.
Pharmacology
There is limited information regarding Atovaquone and proguanil hydrochloride Pharmacology in the drug label.
Mechanism of Action
- Atovaquone and proguanil hydrochloride, interfere with 2 different pathways involved in the biosynthesis of pyrimidines required for nucleic acid replication. Atovaquone is a selective inhibitor of parasite mitochondrial electron transport. Proguanil hydrochloride primarily exerts its effect by means of the metabolite cycloguanil, a dihydrofolate reductase inhibitor. Inhibition of dihydrofolate reductase in the malaria parasite disrupts deoxythymidylate synthesis.
Structure
- Atovaquone and proguanil hydrochloride tablets (Adult Strength), for oral administration.are a fixed-dose combination of the antimalarial agents atovaquone USP and proguanil hydrochloride USP.
The chemical name of atovaquone USP is trans-2-[4-(4-chlorophenyl)cyclohexyl]-3-hydroxy-1,4-naphthalenedione. Atovaquone USP is a yellow crystalline solid that is freely soluble in N-methyl-2-pyrrolidone and in tetrahydrofuran; soluble in chloroform; sparingly soluble in acetone and dimethyl sulfoxide; slightly soluble in octanol, ethyl acetate, polyethylene glycol 200; very slightly soluble in 0.1N sodium hydroxide; insoluble in water. It has a molecular weight of 366.84 and the molecular formula C22H19ClO3. The compound has the following structural formula:
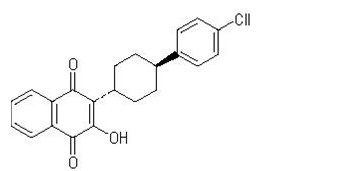
- The chemical name of proguanil hydrochloride USP is 1-(4-chlorophenyl)-5-isopropyl-biguanide hydrochloride. Proguanil hydrochloride USP is a white crystalline powder slightly soluble in water, sparingly soluble in alcohol, practically insoluble in methylene chloride. It has a molecular weight of 290.22 and the molecular formula C11H16ClN5•HCl. The compound has the following structural formula:

- Each atovaquone and proguanil hydrochloride tablet contains 250 mg of atovaquone USP and 100 mg of proguanil hydrochloride USP. The inactive ingredients in the tablet are colloidal silicon dioxide, ferric oxide red, hypromellose 2910, low substituted hydroxypropyl cellulose, magnesium stearate, microcrystalline cellulose, poloxamer 188, polyethylene glycol 400, polyethylene glycol 8000, povidone K30, sodium starch glycolate and titanium dioxide.
Pharmacodynamics
- No trials of the pharmacodynamics of atovaquone and proguanil hydrochloride have been conducted.
Pharmacokinetics
Absorption: Atovaquone is a highly lipophilic compound with low aqueous solubility. The bioavailability of atovaquone shows considerable inter-individual variability.
- Dietary fat taken with atovaquone increases the rate and extent of absorption, increasing AUC 2 to 3 times and Cmax 5 times over fasting. The absolute bioavailability of the tablet formulation of atovaquone when taken with food is 23%. Atovaquone and proguanil hydrochloride tablets should be taken with food or a milky drink.
- Distribution: Atovaquone is highly protein bound (>99%) over the concentration range of 1 to 90 mcg/mL. A population pharmacokinetic analysis demonstrated that the apparent volume of distribution of atovaquone (V/F) in adult and pediatric patients after oral administration is approximately 8.8 L/kg.
- Proguanil is 75% protein bound. A population pharmacokinetic analysis demonstrated that the apparent V/F of proguanil in adult and pediatric patients >15 years of age with body weights from 31 to 110 kg ranged from 1,617 to 2,502 L. In pediatric patients ≤15 years of age with body weights from 11 to 56 kg, the V/F of proguanil ranged from 462 to 966 L.
- In human plasma, the binding of atovaquone and proguanil was unaffected by the presence of the other.
Metabolism: In a study where 14C-labeled atovaquone was administered to healthy volunteers, greater than 94% of the dose was recovered as unchanged atovaquone in the feces over 21 days. There was little or no excretion of atovaquone in the urine (less than 0.6%). There is indirect evidence that atovaquone may undergo limited metabolism; however, a specific metabolite has not been identified. Between 40% to 60% of proguanil is excreted by the kidneys. Proguanil is metabolized to cycloguanil (primarily via CYP2C19) and 4-chlorophenylbiguanide. The main routes of elimination are hepatic biotransformation and renal excretion.
Elimination: The elimination half-life of atovaquone is about 2 to 3 days in adult patients.
- The elimination half-life of proguanil is 12 to 21 hours in both adult patients and pediatric patients, but may be longer in individuals who are slow metabolizers.
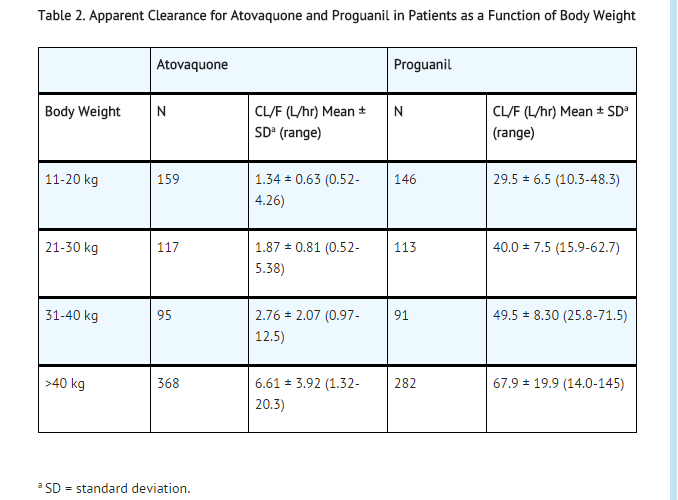
- A population pharmacokinetic analysis in adult and pediatric patients showed that the apparent clearance (CL/F) of both atovaquone and proguanil are related to the body weight. The values CL/F for both atovaquone and proguanil in subjects with body weight ≥11 kg are shown in Table 2.
Nonclinical Toxicology
There is limited information regarding Atovaquone and proguanil hydrochloride Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Atovaquone and proguanil hydrochloride Clinical Studies in the drug label.
How Supplied
There is limited information regarding Atovaquone and proguanil hydrochloride How Supplied in the drug label.
Storage
There is limited information regarding Atovaquone and proguanil hydrochloride Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Atovaquone and proguanil hydrochloride |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Atovaquone and proguanil hydrochloride |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Atovaquone and proguanil hydrochloride Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Atovaquone and proguanil hydrochloride interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Atovaquone and proguanil hydrochloride Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Atovaquone and proguanil hydrochloride Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.