Macitentan
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Gerald Chi
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: EMBRYO-FETAL TOXICITY
See full prescribing information for complete Boxed Warning.
* Do not administer OPSUMIT to a pregnant female because it may cause fetal harm.
|
Overview
Macitentan is an endothelin receptor antagonist and antihypertensive that is FDA approved for the {{{indicationType}}} of pulmonary artery hypertension. There is a Black Box Warning for this drug as shown here. Common adverse reactions include anemia, influenza, headache, urinary tract infections, bronchitis, nasopharyngitis, and pharyngitis.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Pulmonary Arterial Hypertension
- OPSUMIT® is an endothelin receptor antagonist (ERA) indicated for the treatment of pulmonary arterial hypertension (PAH, WHO Group I) to delay disease progression. Disease progression included: death, initiation of intravenous (IV) or subcutaneous prostanoids, or clinical worsening of PAH (decreased 6-minute walk distance, worsened PAH symptoms and need for additional PAH treatment). OPSUMIT also reduced hospitalization for PAH.
- Effectiveness was established in a long-term study in PAH patients with predominantly WHO Functional Class II-III symptoms treated for an average of 2 years. Patients were treated with OPSUMIT monotherapy or in combination with phosphodiesterase-5 inhibitors or inhaled prostanoids. Patients had idiopathic and heritable PAH (57%), PAH caused by connective tissue disorders (31%), and PAH caused by congenital heart disease with repaired shunts (8%).
- Dosing Information
- The recommended dosage of OPSUMIT is 10 mg once daily for oral administration. Doses higher than 10 mg once daily have not been studied in patients with PAH and are not recommended.
- Initiate treatment with OPSUMIT in females of reproductive potential only after a negative pregnancy test. Obtain monthly pregnancy test during treatment.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Macitentan in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Macitentan in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Macitentan in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- The safety and efficacy of OPSUMIT in children have not been established.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Macitentan in pediatric patients.
Contraindications
- Pregnancy
- OPSUMIT may cause fetal harm when administered to a pregnant woman. OPSUMIT is contraindicated in females who are pregnant. OPSUMIT was consistently shown to have teratogenic effects when administered to animals. If OPSUMIT is used during pregnancy, apprise the patient of the potential hazard to a fetus.
Warnings
|
WARNING: EMBRYO-FETAL TOXICITY
See full prescribing information for complete Boxed Warning.
* Do not administer OPSUMIT to a pregnant female because it may cause fetal harm.
|
- Embryo-fetal Toxicity
- OPSUMIT may cause fetal harm when administered during pregnancy and is contraindicated for use in females who are pregnant. In females of reproductive potential, exclude pregnancy prior to initiation of therapy, ensure use of acceptable contraceptive methods and obtain monthly pregnancy tests.
- OPSUMIT is available for females through the OPSUMIT REMS Program, a restricted distribution program.
- OPSUMIT REMS Program
- For all females, OPSUMIT is available only through a restricted program called the OPSUMIT REMS Program, because of the risk of embryo-fetal toxicity.
- Notable requirements of the OPSUMIT REMS Program include the following:
- Prescribers must be certified with the program by enrolling and completing training.
- All females, regardless of reproductive potential, must enroll in the OPSUMIT REMS Program prior to initiating OPSUMIT. Male patients are not enrolled in the REMS.
- Females of reproductive potential must comply with the pregnancy testing and contraception requirements.
- Pharmacies must be certified with the program and must only dispense to patients who are authorized to receive OPSUMIT.
- Further information is available at www.OPSUMITREMS.com or 1-866-228-3546. Information on OPSUMIT certified pharmacies or wholesale distributors is available through Actelion Pathways at 1-866-228-3546.
- Hepatotoxicity
- Other ERAs have caused elevations of aminotransferases, hepatotoxicity, and liver failure. The incidence of elevated aminotransferases in the study of OPSUMIT in PAH is shown in Table 1.
- In the placebo-controlled study of OPSUMIT, discontinuations for hepatic adverse events were 3.3% in the OPSUMIT 10 mg group vs. 1.6% for placebo. Obtain liver enzyme tests prior to initiation of OPSUMIT and repeat during treatment as clinically indicated.
- Advise patients to report symptoms suggesting hepatic injury (nausea, vomiting, right upper quadrant pain, fatigue, anorexia, jaundice, dark urine, fever, or itching). If clinically relevant aminotransferase elevations occur, or if elevations are accompanied by an increase in bilirubin >2 × ULN, or by clinical symptoms of hepatotoxicity, discontinue OPSUMIT. Consider re-initiation of OPSUMIT when hepatic enzyme levels normalize in patients who have not experienced clinical symptoms of hepatotoxicity.
- Hemoglobin Decrease
- Decreases in hemoglobin concentration and hematocrit have occurred following administration of other ERAs and were observed in clinical studies with OPSUMIT. These decreases occurred early and stabilized thereafter. In the placebo-controlled study of OPSUMIT in PAH, OPSUMIT 10 mg caused a mean decrease in hemoglobin from baseline to up to 18 months of about 1.0 g/dL compared to no change in the placebo group. A decrease in hemoglobin to below 10.0 g/dL was reported in 8.7% of the OPSUMIT 10 mg group and in 3.4% of the placebo group. Decreases in hemoglobin seldom require transfusion. Initiation of OPSUMIT is not recommended in patients with severe anemia. Measure hemoglobin prior to initiation of treatment and repeat during treatment as clinically indicated.
- Pulmonary Edema with Pulmonary Veno-occlusive Disease (PVOD)
- Should signs of pulmonary edema occur, consider the possibility of associated PVOD. If confirmed, discontinue OPSUMIT.
- Decreased Sperm Counts
- Other ERAs have caused adverse effects on spermatogenesis. Counsel men about potential effects on fertility.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
- Safety data for OPSUMIT were obtained primarily from one placebo-controlled clinical study in 742 patients with PAH (SERAPHIN study). The exposure to OPSUMIT in this trial was up to 3.6 years with a median exposure of about 2 years (N=542 for 1 year; N=429 for 2 years; and N=98 for more than 3 years). The overall incidence of treatment discontinuations because of adverse events was similar across OPSUMIT 10 mg and placebo treatment groups (approximately 11%).
Table 2 presents adverse reactions more frequent on OPSUMIT than on placebo by ≥3%.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Macitentan in the drug label.
Drug Interactions
- Strong CYP3A4 Inducers
- Strong inducers of CYP3A4 such as rifampin significantly reduce macitentan exposure. Concomitant use of OPSUMIT with strong CYP3A4 inducers should be avoided.
- Strong CYP3A4 Inhibitors
- Concomitant use of strong CYP3A4 inhibitors like ketoconazole approximately double macitentan exposure. Many HIV drugs like ritonavir are strong inhibitors of CYP3A4. Avoid concomitant use of OPSUMIT with strong CYP3A4 inhibitors. Use other PAH treatment options when strong CYP3A4 inhibitors are needed as part of HIV treatment.
Use in Specific Populations
Pregnancy
- Pregnancy Category X
- Risk Summary
- OPSUMIT may cause fetal harm when administered to a pregnant woman and is contraindicated during pregnancy. Macitentan was teratogenic in rabbits and rats at all doses tested. A no-effect dose was not established in either species. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, advise the patient of the potential hazard to a fetus.
- Animal Data
- In both rabbits and rats, there were cardiovascular and mandibular arch fusion abnormalities. Administration of macitentan to female rats from late pregnancy through lactation caused reduced pup survival and impairment of the male fertility of the offspring at all dose levels tested.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Macitentan in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Macitentan during labor and delivery.
Nursing Mothers
- It is not known whether OPSUMIT is present in human milk. Macitentan and its metabolites were present in the milk of lactating rats. Because many drugs are present in human milk and because of the potential for serious adverse reactions from macitentan in nursing infants, nursing mothers should discontinue nursing or discontinue OPSUMIT.
Pediatric Use
- The safety and efficacy of OPSUMIT in children have not been established.
Geriatic Use
- Of the total number of subjects in the clinical study of OPSUMIT for PAH, 14% were 65 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects.
Gender
There is no FDA guidance on the use of Macitentan with respect to specific gender populations.
Race
There is no FDA guidance on the use of Macitentan with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Macitentan in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Macitentan in patients with hepatic impairment.
Females of Reproductive Potential and Males
- Females
- Pregnancy Testing: Female patients of reproductive potential must have a negative pregnancy test prior to starting treatment with OPSUMIT and monthly pregnancy tests during treatment with OPSUMIT. Advise patients to contact their health care provider if they become pregnant or suspect they may be pregnant. Perform a pregnancy test if pregnancy is suspected for any reason. For positive pregnancy tests, counsel patients on the potential risk to the fetus.
- Contraception: Female patients of reproductive potential must use acceptable methods of contraception during treatment with OPSUMIT and for 1 month after treatment with OPSUMIT. Patients may choose one highly effective form of contraception (intrauterine devices (IUD), contraceptive implants or tubal sterilization) or a combination of methods (hormone method with a barrier method or two barrier methods). If a partner's vasectomy is the chosen method of contraception, a hormone or barrier method must be used along with this method. Counsel patients on pregnancy planning and prevention, including emergency contraception, or designate counseling by another healthcare provider trained in contraceptive counseling.
- Males
- Testicular effects: Like other endothelin receptor antagonists, OPSUMIT may have an adverse effect on spermatogenesis
Immunocompromised Patients
There is no FDA guidance one the use of Macitentan in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- Intravenous
Monitoring
There is limited information regarding Monitoring of Macitentan in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Macitentan in the drug label.
Overdosage
Acute Overdose
Signs and Symptoms
- Description
Management
- Description
Chronic Overdose
There is limited information regarding Chronic Overdose of Macitentan in the drug label.
Pharmacology
| |
Macitentan
| |
| Systematic (IUPAC) name | |
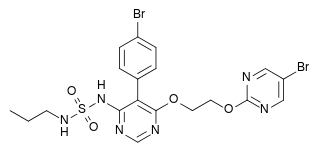
| N-[5-(4-Bromophenyl)-6-[2-[(5-bromo-2-pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]-N'-propylsulfamide | |
| Identifiers | |
| CAS number | |
| ATC code | C02 |
| PubChem | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 588.273 g/mol |
| SMILES | & |
| Synonyms | ACT-064992 |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | Hydrolysis, oxidation (CYP3A4) |
| Half life | ? |
| Excretion | 2/3 urine, 1/3 faeces |
| Therapeutic considerations | |
| Pregnancy cat. |
X(US) |
| Legal status |
[[Prescription drug|Template:Unicode-only]](US) FDA approved drug |
| Routes | Oral |
Mechanism of Action
Structure
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Macitentan in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Macitentan in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Macitentan in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Macitentan in the drug label.
How Supplied
Storage
There is limited information regarding Macitentan Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Macitentan |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Macitentan |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Macitentan in the drug label.
Precautions with Alcohol
- Alcohol-Macitentan interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Opsumit®[1]
Look-Alike Drug Names
- A® — B®[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "OPSUMIT (macitentan) tablet, film coated".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Macitentan |Pill Name=No image.jpg |Drug Name= |Pill Ingred=|+sep=; |Pill Imprint= |Pill Dosage= |Pill Color=|+sep=; |Pill Shape= |Pill Size (mm)= |Pill Scoring= |Pill Image= |Drug Author= |NDC=
}}
{{#subobject:
|Label Page=Macitentan |Label Name=Macitentan11.png
}}
{{#subobject:
|Label Page=Macitentan |Label Name=Macitentan11.png
}}


