Ursodiol
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Adeel Jamil, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Ursodiol is a bile acid and gastrointestinal agent that is FDA approved for the treatment of chemodissolution of gall bladder stone, prevention of gallstone formation in obese patients experiencing rapid weight loss and primary biliary cirrhosis. Common adverse reactions include rash, constipation, diarrhea, indigestion, nausea, vomiting, backache, dizziness, bronchitis, cough, pharyngitis and upper respiratory infection.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Ursodiol is indicated for patients with radiolucent, noncalcified gallbladder stones < 20 mm in greatest diameter in whom elective cholecystectomy would be undertaken except for the presence of increased surgical risk due to systemic disease, advanced age, idiosyncratic reaction to general anesthesia, or for those patients who refuse surgery. Safety of use of ursodiol beyond 24 months is not established.
- Ursodiol is indicated for the prevention of gallstone formation in obese patients experiencing rapid weight loss.
Dosing Information
Gallstone Dissolution
- The recommended dose for ursodiol treatment of radiolucent gallbladder stones is 8 - 10 mg/kg/day given in 2 or 3 divided doses. Ultrasound images of the gallbladder should be obtained at 6-month intervals for the first year of ursodiol therapy to monitor gallstone response. If gallstones appear to have dissolved, ursodiol therapy should be continued and dissolution confirmed on a repeat ultrasound examination within 1 to 3 months. Most patients who eventually achieve complete stone dissolution will show partial or complete dissolution at the first on-treatment reevaluation. If partial stone dissolution is not seen by 12 months of ursodiol therapy, the likelihood of success is greatly reduced.
Gallstone Prevention
- The recommended dosage of ursodiol for gallstone prevention in patients undergoing rapid weight loss is 400 mg/day (300 mg b.i.d.).
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ursodiol in adult patients.
Non–Guideline-Supported Use
- Biliary cirrhosis - Cystic fibrosis
- Cholestasis of parenteral nutrition
- Cholestatic jaundice syndrome
- Congenital dilatation of lobar intrahepatic bile duct
- Chronic hepatitis
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Ursodiol in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ursodiol in pediatric patients.
Non–Guideline-Supported Use
Contraindications
- Ursodiol will not dissolve calcified cholesterol stones, radiopaque stones, or radiolucent bile pigment stones. Hence, patients with such stones are not candidates for ursodiol therapy.
- Patients with compelling reasons for cholecystectomy including unremitting acute cholecystitis, cholangitis, biliary obstruction, gallstone pancreatitis, or biliary-[[gastrointestinal fistula]] are not candidates for ursodiol therapy.
- Allergy to bile acids.
Warnings
PRECAUTIONS
Liver Tests
- Ursodiol therapy has not been associated with liver damage. Lithocholic acid, a naturally occurring bile acid, is known to be a liver-toxic metabolite. This bile acid is formed in the gut from ursodiol less efficiently and in smaller amounts than that seen from chenodiol. Lithocholic acid is detoxified in the liver by sulfation and, although man appears to be an efficient sulfater, it is possible that some patients may have a congenital or acquired deficiency in sulfation, thereby predisposing them to lithocholate-induced liver damage.
- Abnormalities in liver enzymes have not been associated with ursodiol therapy and, in fact, ursodiol has been shown to decrease liver enzyme levels in liver disease. However, patients given ursodiol should have SGOT (AST) and SGPT (ALT) measured at the initiation of therapy and thereafter as indicated by the particular clinical circumstances.
Adverse Reactions
Clinical Trials Experience
- The nature and frequency of adverse experiences were similar across all groups.
- The following tables provide comprehensive listings of the adverse experiences reported that occurred with a 5% incidence level:
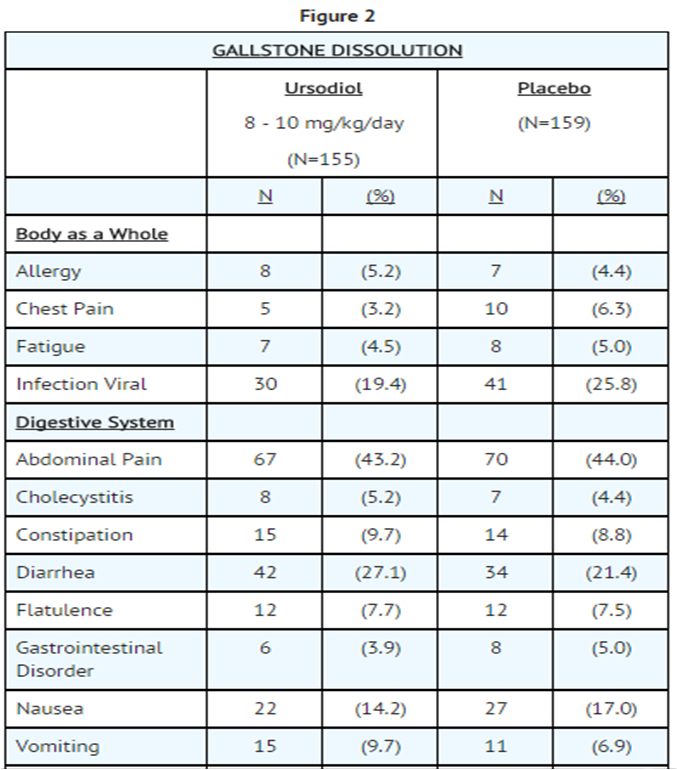
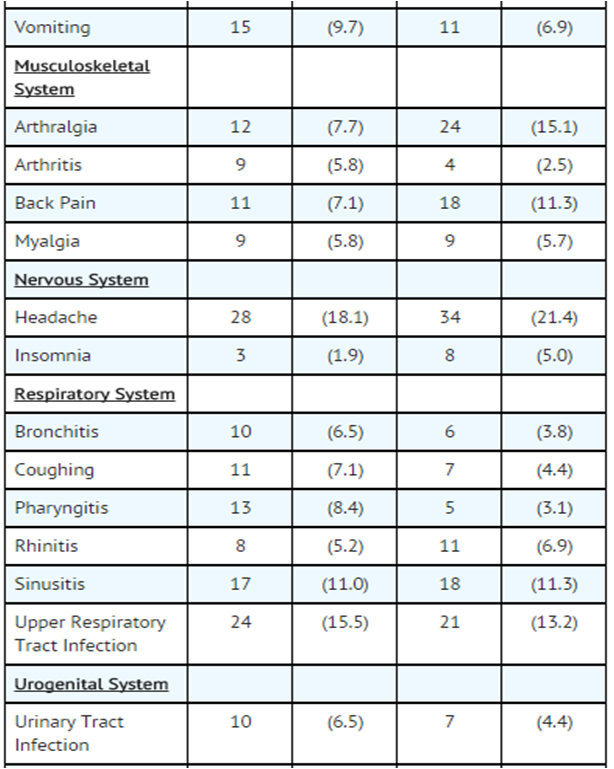
- Gallstone Dissolution In Ursodiol and Placebo Patients
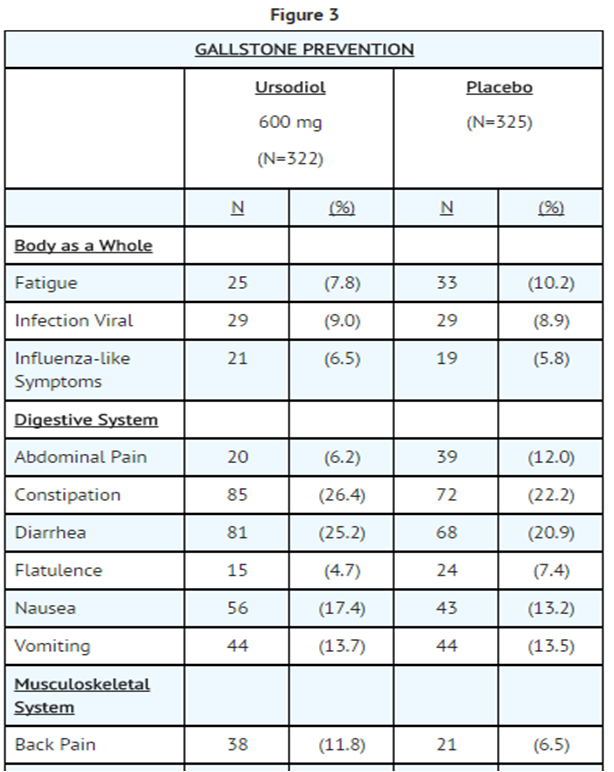
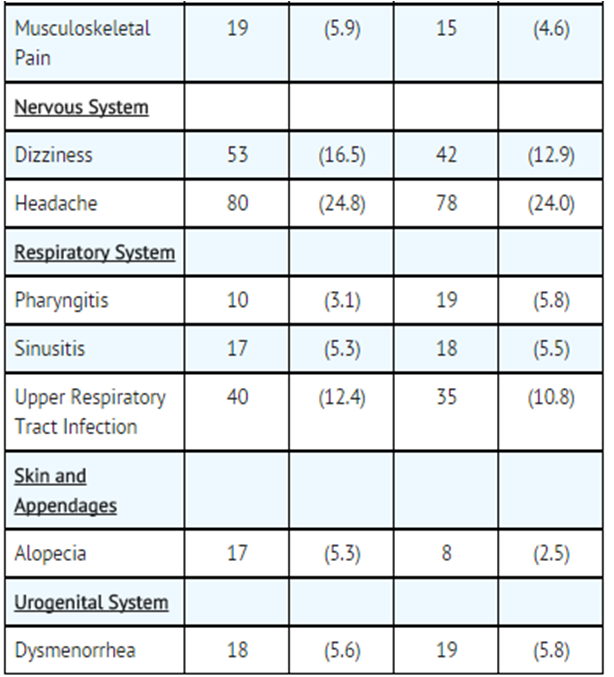
- Gallstone Prevention in Ursodiol and Placebo-Treated Patients
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Ursodiol in the drug label.
Drug Interactions
- Bile acid sequestering agents such as cholestyramine and colestipol may interfere with the action of ursodiol by reducing its absorption. Aluminum-based antacids have been shown to absorb bile acids in vitro and may be expected to interfere with ursodiol in the same manner as the bile acid sequestering agents. Estrogens, oral contraceptives, and clofibrate (and perhaps other lipid-lowering drugs) increase hepatic cholesterol secretion, and encourage cholesterol gallstone formation and hence may counteract the effectiveness of ursodiol.
Use in Specific Populations
Pregnancy
- Reproduction studies have been performed in rats and rabbits with ursodiol doses up to 200-fold the therapeutic dose and have revealed no evidence of impaired fertility or harm to the fetus at doses of 20- to 100-fold the human dose in rats and at 5-fold the human dose (highest dose tested) in rabbits. Studies employing 100- to 200-fold the human dose in rats have shown some reduction in fertility rate and litter size. There have been no adequate and well-controlled studies of the use of ursodiol in pregnant women, but inadvertent exposure of 4 women to therapeutic doses of the drug in the first trimester of pregnancy during the ursodiol trials led to no evidence of effects on the fetus or newborn baby. Although it seems unlikely, the possibility that ursodiol can cause fetal harm cannot be ruled out; hence, the drug is not recommended for use during pregnancy.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Ursodiol in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Ursodiol during labor and delivery.
Nursing Mothers
- It is not known whether ursodiol is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when ursodiol is administered to a nursing mother.
Pediatric Use
There is no FDA guidance on the use of Ursodiol with respect to pediatric patients.
Geriatic Use
- In worldwide clinical studies of ursodiol, approximately 14% of subjects were over 65 years of age (approximately 3% were over 75 years old). In a subgroup analysis of existing clinical trials, patients greater than 56 years of age did not exhibit statistically significantly different complete dissolution rates from the younger population. No age-related differences in safety and effectiveness were found. Other reported clinical experience has not identified differences in response in elderly and younger patients. However, small differences in efficacy and greater sensitivity of some elderly individuals taking ursodiol cannot be ruled out. Therefore, it is recommended that dosing proceed with caution in this population.
Gender
There is no FDA guidance on the use of Ursodiol with respect to specific gender populations.
Race
There is no FDA guidance on the use of Ursodiol with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Ursodiol in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Ursodiol in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Ursodiol in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Ursodiol in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
There is limited information regarding Monitoring of Ursodiol in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Ursodiol in the drug label.
Overdosage
- Neither accidental nor intentional overdosing with Ursodiol has been reported. Doses of Ursodiol in the range of 16 - 20 mg/kg/day have been tolerated for 6 - 37 months without symptoms by 7 patients. The LD50 for ursodiol in rats is over 5000 mg/kg given over 7 - 10 days and over 7500 mg/kg for mice. The most likely manifestation of severe overdose with ursodiol would probably be diarrhea, which should be treated symptomatically.
Pharmacology
Mechanism of Action
- Ursodiol suppresses hepatic synthesis and secretion of cholesterol, and also inhibits intestinal absorption of cholesterol. It appears to have little inhibitory effect on synthesis and secretion into bile of endogenous bile acids, and does not appear to affect secretion of phospholipids into bile.
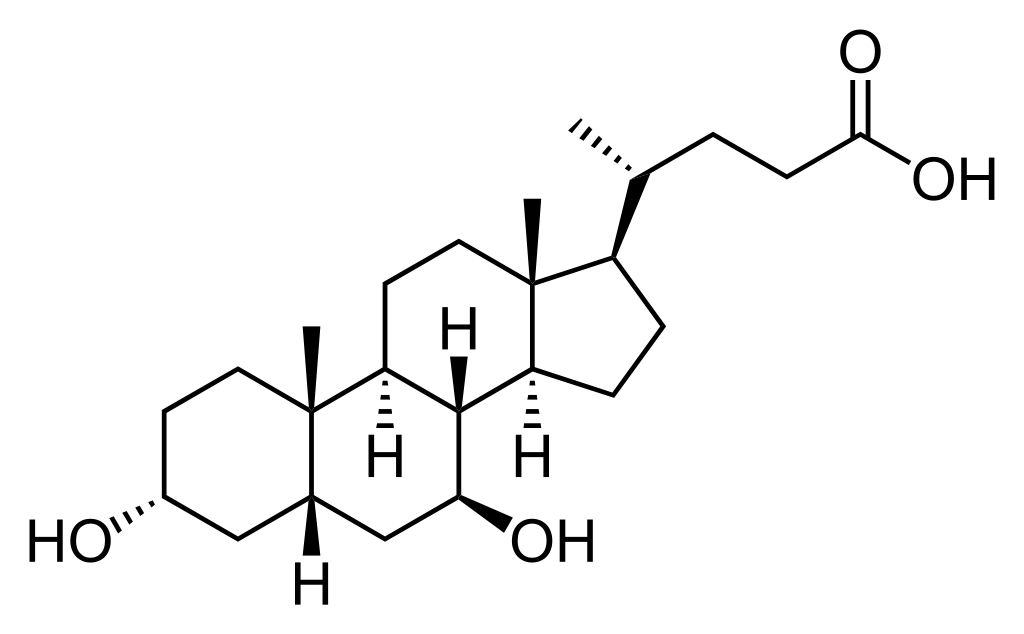
Structure
- Ursodiol is a bile acid available as 300 mg capsules suitable for oral administration.
- Ursodeoxycholic acid,is a naturally occurring bile acid found in small quantities in normal human bile and in the biles of certain other mammals. It is a bitter-tasting, white powder freely soluble in ethanol, methanol, and glacial acetic acid; sparingly soluble in chloroform; slightly soluble in ether; and insoluble in water. The chemical name for ursodiol is 3α,7β-Dihydroxy-5β-cholan-24-oic acid (C24H40O4). Ursodiol, USP has a molecular weight of 392.57. Its structure is shown below:
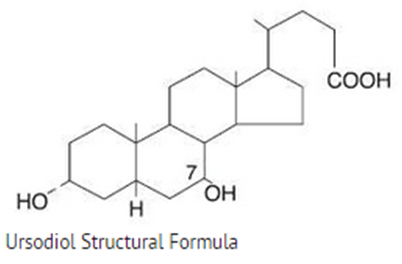
Pharmacodynamics
- Ursodiol suppresses hepatic synthesis and secretion of cholesterol, and also inhibits intestinal absorption of cholesterol. It appears to have little inhibitory effect on synthesis and secretion into bile of endogenous bile acids, and does not appear to affect secretion of phospholipids into bile.
- With repeated dosing, bile ursodeoxycholic acid concentrations reach a steady state in about 3 weeks. Although insoluble in aqueous media, cholesterol can be solubilized in at least two different ways in the presence of dihydroxy bile acids. In addition to solubilizing cholesterol in micelles, ursodiol acts by an apparently unique mechanism to cause dispersion of cholesterol as liquid crystals in aqueous media. Thus, even though administration of high doses (e.g., 15 - 18 mg/kg/day) does not result in a concentration of ursodiol higher than 60% of the total bile acid pool, ursodiol-rich bile effectively solubilizes cholesterol. The overall effect of ursodiol is to increase the concentration level at which saturation of cholesterol occurs.
- The various actions of ursodiol combine to change the bile of patients with gallstones from cholesterol-precipitating to cholesterolsolubilizing, thus resulting in bile conducive to cholesterol stone dissolution.
- After ursodiol dosing is stopped, the concentration of the bile acid in bile falls exponentially, declining to about 5% - 10% of its steady state level in about 1 week.
Pharmacokinetics
- About 90% of a therapeutic dose of ursodiol is absorbed in the small bowel after oral administration. After absorption, ursodiol enters the portal vein and undergoes efficient extraction from portal blood by the liver (i.e., there is a large “first-pass” effect) where it is conjugated with either glycine or taurine and is then secreted into the hepatic bile ducts. Ursodiol in bile is concentrated in the gallbladder and expelled into the duodenum in gallbladder bile via the cystic and common ducts by gallbladder contractions provoked by physiologic responses to eating. Only small quantities of ursodiol appear in the systemic circulation and very small amounts are excreted into urine. The sites of the drug’s therapeutic actions are in the liver, bile, and gut lumen.
- Beyond conjugation, ursodiol is not altered or catabolized appreciably by the liver or intestinal mucosa. A small proportion of orally administered drug undergoes bacterial degradation with each cycle of enterohepatic circulation. Ursodiol can be both oxidized and reduced at the 7-carbon, yielding either 7-keto-lithocholic acid or lithocholic acid, respectively. Further, there is some bacterially catalyzed deconjugation of [[glyco-ursodeoxycholic acid
|glyco-]] and tauro-ursodeoxycholic acid in the small bowel. Free ursodiol, 7-keto-lithocholic acid, and lithocholic acid are relatively insoluble in and larger proportions of these compounds are lost from the distal gut into the feces. Reabsorbed free ursodiol is reconjugated by the liver. Eighty percent of lithocholic acid formed in the small bowel is excreted in the feces, but the 20% that is absorbed is sulfated at the 3-hydroxyl group in the liver to relatively insoluble lithocholyl conjugates which are excreted into bile and lost in feces. Absorbed 7-keto-lithocholic acid is stereospecifically reduced in the liver to chenodiol.
- The 7-dehydroxylation reaction appears to be alpha-specific, i.e., chenodiol is more efficiently 7-dehydroxylated than ursodiol and, for equimolar doses of ursodiol and chenodiol, levels of lithocholic acid appearing in bile are lower with the former. Man has the capacity to sulfate lithocholic acid. Although liver injury has not been associated with ursodiol therapy, a reduced capacity to sulfate may exist in some individuals, but such a deficiency has not yet been clearly demonstrated.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Ursodiol in the drug label.
Clinical Studies
Gallstone Dissolution
- On the basis of clinical trial results in a total of 868 patients with radiolucent gallstones treated in 8 studies (three in the U.S. involving 282 patients, one in the U.K. involving 130 patients, and four in Italy involving 456 patients) for periods ranging from 6 - 78 months with ursodiol doses ranging from about 5 - 20 mg/kg/day, an ursodiol dose of about 8 - 10 mg/kg/day appeared to be the best dose.
- With an ursodiol dose of about 10 mg/kg/day, complete stone dissolution can be anticipated in about 30% of unselected patients with uncalcified gallstones < 20 mm in maximal diameter treated for up to 2 years. Patients with calcified gallstones prior to treatment, or patients who develop stone calcification or gallbladder nonvisualization on treatment, and patients with stones > 20 mm in maximal diameter rarely dissolve their stones. The chance of gallstone dissolution is increased up to 50% in patients with floating or floatable stones (i.e., those with high cholesterol content), and is inversely related to stone size for those < 20 mm in maximal diameter.
- Complete dissolution was observed in 81% of patients with stones up to 5 mm in diameter. Age, sex, weight, degree of obesity, and serum cholesterol level are not related to the chance of stone dissolution with ursodiol.
- A nonvisualizing gallbladder by oral cholecystogram prior to the initiation of therapy is not a contraindication to ursodiol therapy (the group of patients with nonvisualizing gallbladders in the ursodiol studies had complete stone dissolution rates similar to the group of patients with visualizing gallbladders). However, gallbladder nonvisualization developing during ursodiol treatment predicts failure of complete stone dissolution and in such cases therapy should be discontinued. Partial stone dissolution occurring within 6 months of beginning therapy with ursodiol appears to be associated with a > 70% chance of eventual complete stone dissolution with further treatment; partial dissolution observed within 1 year of starting therapy indicates a 40% probability of complete dissolution. Stone recurrence after dissolution with ursodiol therapy was seen within 2 years in 8/27 (30%) of patients in the U.K. studies. Of 16 patients in the U.K. study whose stones had previously dissolved on chenodiol but later recurred, 11 had complete dissolution on ursodiol. Stone recurrence has been observed in up to 50% of patients within 5 years of complete stone dissolution on ursodiol therapy. Serial ultrasonographic examinations should be obtained to monitor for recurrence of stones, bearing in mind that radiolucency of the stones should be established before another course of ursodiol is instituted. A prophylactic dose of ursodiol has not been established.
Gallstone Prevention
- Two placebo-controlled, multicenter, double-blind, randomized, parallel group trials in a total of 1,316 obese patients were undertaken to evaluate ursodiol in the prevention of gallstone formation in obese patients undergoing rapid weight loss. The first trial consisted of 1,004 obese patients with a body mass index (BMI) ≥ 38 who underwent weight loss induced by means of a very low calorie diet for a period of 16 weeks. An intent-to-treat analysis of this trial showed that gallstone formation occurred in 23% of the placebo group, while those patients on 300, 400, or 1200 mg/day of ursodiol experienced a 6%, 3%, and 2% incidence of gallstone formation, respectively. The mean weight loss for this 16-week trial was 47 lb for the placebo group, and 47, 48, and 50 lb for the 300, 400, and 1200 mg/day ursodiol groups, respectively.
- The second trial consisted of 312 obese patients (BMI ≥ 40) who underwent rapid weight loss through gastric bypass surgery. The trial drug treatment period was for 6 months following this surgery. Results of this trial showed that gallstone formation occurred in 23% of the placebo group, while those patients on 300, 400, or 1200 mg/day of Ursodiol experienced a 9%, 1%, and 5% incidence of gallstone formation, respectively. The mean weight loss for this 6-month trial was 64 lb for the placebo group, and 67, 74, and 72 lb for the 300, 400, and 1200 mg/day ursodiol groups, respectively.
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Ursodeoxycholic acid was tested in 2-year oral carcinogenicity studies in CD-1 mice and Sprague-Dawley rats at daily doses of 50, 250, and 1000 mg/kg/day. It was not tumorigenic in mice. In the rat study, it produced statistically significant dose-related increased incidences of pheochromocytomas of adrenal medulla in males (p=0.014, Peto trend test) and females (p=0.004, Peto trend test). A 78-week rat study employing intrarectal instillation of lithocholic acid and tauro-deoxycholic acid, metabolites of ursodiol and chenodiol, has been conducted. These bile acids alone did not produce any tumors. A tumor-promoting effect of both metabolites was observed when they were co-administered with a carcinogenic agent. Results of epidemiologic studies suggest that bile acids might be involved in the pathogenesis of human colon cancer in patients who had undergone a cholecystectomy, but direct evidence is lacking. Ursodiol is not mutagenic in the Ames test. Dietary administration of lithocholic acid to chickens is reported to cause hepatic adenomatous hyperplasia.
How Supplied
Ursodiol Capsules USP, 300 mg are opaque white and pink capsules, filled with white powder. They are imprinted
“Є503” in black ink on cap and body and are supplied in bottles of 100 and 1000.
Keep out of reach of children.
Manufactured By:
Epic Pharma, LLC
Laurelton, NY 11413
Revised August 2014
MF503REV08/14
OE1105
Storage
- Store at 20° - 25°C (68° - 77°F). Dispense contents in tight, light-resistant container as defined in the USP.
Images
Drug Images
{{#ask: Page Name::Ursodiol |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel

{{#ask: Label Page::Ursodiol |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Ursodiol in the drug label.
Precautions with Alcohol
- Alcohol-Ursodiol interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Actigall®
- Urso®
- Urso 250®
- Urso Forte®
Look-Alike Drug Names
There is limited information regarding Ursodiol Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Ursodiol
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}