Treprostinil (injection)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sheng Shi, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Treprostinil (injection) is a prostaglandin analog that is FDA approved for the treatment of pulmonary arterial hypertension. Common adverse reactions include edema, hypotension, vasodilatation, flushing, injection site pain, injection site reaction, pruritus , rash, diarrhea, nausea, jaw pain, dizziness, headache, cough, and throat irritation.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Pulmonary Arterial Hypertension
General Dosing information
- Treprostinil is supplied in 20 mL vials containing 20, 50, 100, or 200 mg of treprostinil (1 mg/mL, 2.5 mg/mL, 5 mg/mL or 10 mg/mL). Treprostinil can be administered as supplied or diluted for intravenous infusion with Sterile Water for Injection, 0.9% Sodium Chloride Injection, Sterile Diluent for Flolan, or Sterile Diluent for Epoprostenol Sodium prior to administration.
Initial Dose for Patients New to Prostacyclin Infusion Therapy
- Treprostinil is indicated for subcutaneous (SC) or intravenous (IV) use only as a continuous infusion. Treprostinil is preferably infused subcutaneously, but can be administered by a central intravenous line if the subcutaneous route is not tolerated, because of severe site pain or reaction. The infusion rate is initiated at 1.25 ng/kg/min. If this initial dose cannot be tolerated because of systemic effects, the infusion rate should be reduced to .625 ng/kg/min.
Dosage Adjustments
- The infusion rate should be increased in increments of 1.25 ng/kg/min per week for the first four weeks of treatment and then 2.5 ng/kg/min per week for the remaining duration of infusion, depending on clinical response. Dosage adjustments may be undertaken more often if tolerated. Abrupt cessation of infusion should be avoided. Restarting a Treprostinil infusion within a few hours after an interruption can be done using the same dose rate. Interruptions for longer periods may require the dose of Treprostinil to be re-titrated.
Patients with Hepatic Insufficiency
- Initial dosage: decreased to 0.625 ng/kg/min ideal body weight and should be increased cautiously. Treprostinil has not been studied in patients with severe hepatic insufficiency
Patients with Renal Insufficiency
- No studies have been performed in patients with renal insufficiency. No specific advice about dosing in patients with renal impairment can be given.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Treprostinil (injection) in adult patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Treprostinil (injection) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Safety and effectiveness in pediatric patients have not been established
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Treprostinil (injection) in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Treprostinil (injection) in pediatric patients.
Contraindications
- None
Warnings
Risks Attributable to the Drug Delivery System
- Chronic intravenous infusions of Treprostinil are delivered using an indwelling central venous catheter. This route is associated with the risk of blood stream infections (BSIs) and sepsis, which may be fatal. Therefore, continuous subcutaneous infusion (undiluted) is the preferred mode of administration.
- In an open-label study of IV treprostinil (n=47), there were seven catheter-related line infections during approximately 35 patient years, or about 1 BSI event per 5 years of use. A CDC survey of seven sites that used IV treprostinil for the treatment of PAH found approximately 1 BSI (defined as any positive blood culture) event per 3 years of use. Administration of IV Treprostinil with a high pH glycine diluent such as Sterile Diluent for Flolan or Sterile Diluent for Epoprostenol Sodium has been associated with a lower incidence of BSIs when compared to neutral diluents (sterile water, 0.9% sodium chloride) when used along with catheter care guidelines.
General Conditions of Use
- Treprostinil should be used only by clinicians experienced in the diagnosis and treatment of PAH.
- Treprostinil is a potent pulmonary and systemic vasodilator. Initiation of Treprostinil must be performed in a setting with adequate personnel and equipment for physiological monitoring and emergency care. Therapy with Treprostinil may be used for prolonged periods, and the patient's ability to administer Treprostinil and care for an infusion system should be carefully considered.
Dose Modification
- Dose should be increased for lack of improvement in, or worsening of, symptoms and it should be decreased for excessive pharmacologic effects or for unacceptable infusion site symptoms
Abrupt Withdrawal or Sudden Large Dose Reduction
- Abrupt withdrawal or sudden large reductions in dosage of Treprostinil may result in worsening of PAH symptoms and should be avoided.
Patients with Hepatic or Renal Insufficiency
- Titrate slowly in patients with hepatic or renal insufficiency, because such patients will likely be exposed to greater systemic concentrations relative to patients with normal hepatic or renal function.
Effect of Other Drugs on Treprostinil
- Co-administration of a cytochrome P450 (CYP) 2C8 enzyme inhibitor (e.g., gemfibrozil) may increase exposure (both Cmax and AUC) to treprostinil. Co-administration of a CYP2C8 enzyme inducer (e.g., rifampin) may decrease exposure to treprostinil. Increased exposure is likely to increase adverse events associated with treprostinil administration, whereas decreased exposure is likely to reduce clinical effectiveness.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse Events with Subcutaneously Administered Treprostinil
- Patients receiving Treprostinil as a subcutaneous infusion reported a wide range of adverse events, many potentially related to the underlying disease (dyspnea, fatigue, chest pain, right ventricular heart failure, and pallor). During clinical trials with subcutaneous infusion of Treprostinil, infusion site pain and reaction were the most common adverse events among those treated with Treprostinil. Infusion site reaction was defined as any local adverse event other than pain or bleeding/bruising at the infusion site and included symptoms such as erythema, induration or rash. Infusion site reactions were sometimes severe and could lead to discontinuation of treatment.

- Other adverse events included diarrhea, jaw pain, edema, vasodilatation and nausea, and these are generally considered to be related to the pharmacologic effects of Treprostinil, whether administered subcutaneously or intravenously.
Adverse Events during Chronic Dosing
- Table 3 lists adverse events that occurred at a rate of at least 3% and were more frequent in patients treated with subcutaneous Treprostinil than with placebo in controlled trials in PAH.
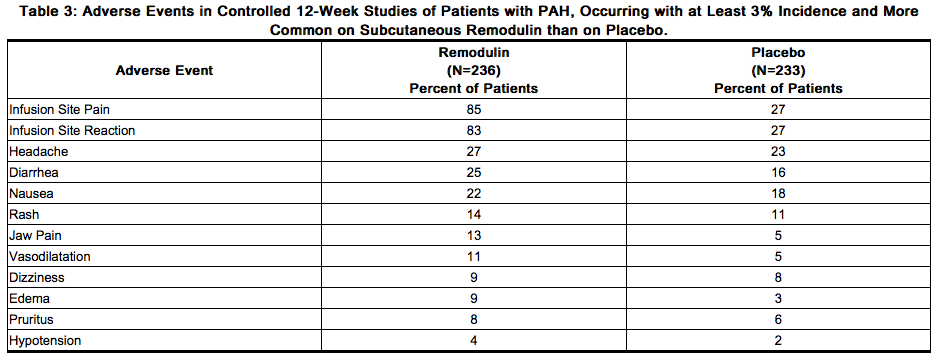
- Reported adverse events (at least 3%) are included except those too general to be informative, and those not plausibly attributable to the use of the drug, because they were associated with the condition being treated or are very common in the treated population.
- The safety of Treprostinil was also studied in a long-term, open-label extension study in which 860 patients were dosed for a mean duration of 1.6 years, with a maximum exposure of 4.6 years. Twenty-nine (29%) percent achieved a dose of at least 40 ng/kg/min (max: 290 ng/kg/min). The safety profile during this chronic dosing study was similar to that observed in the 12-week placebo controlled study except for the following suspected adverse drug reactions (occurring in at least 3% of patients): anorexia, vomiting, infusion site infection, asthenia, and abdominal pain.
Adverse Events Attributable to the Drug Delivery System
- In controlled studies of Treprostinil administered subcutaneously, there were no reports of infection related to the drug delivery system. There were 187 infusion system complications reported in 28% of patients (23% Treprostinil, 33% placebo); 173 (93%) were pump related and 14 (7%) related to the infusion set. Eight of these patients (4 Treprostinil, 4 Placebo) reported non-serious adverse events resulting from infusion system complications. Adverse events resulting from problems with the delivery systems were typically related to either symptoms of excess Treprostinil (e.g., nausea) or return of PAH symptoms (e.g., dyspnea). These events were generally resolved by correcting the delivery system pump or infusion set problem such as replacing the syringe or battery, reprogramming the pump, or straightening a crimped infusion line. Adverse events resulting from problems with the delivery system did not lead to clinical instability or rapid deterioration. In addition to these adverse events due to the drug delivery system during subcutaneous administration, the following adverse events may be attributable to the IV mode of infusion including arm swelling, paresthesias, hematoma and pain.
Postmarketing Experience
- In addition to adverse reactions reported from clinical trials, the following events have been identified during post-approval use of Treprostinil. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The following events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, and potential connection to Treprostinil. These events are thrombophlebitis associated with peripheral intravenous infusion, thrombocytopenia and bone pain. In addition, generalized rashes, sometimes macular or papular in nature, and cellulitis have been infrequently reported.
Drug Interactions
- Pharmacokinetic/pharmacodynamic interaction studies have been conducted with treprostinil administered subcutaneously (Treprostinil) and orally (treprostinil diethanolamine).
Pharmacodynamics
Antihypertensive Agents or Other Vasodilators
- Concomitant administration of Treprostinil with diuretics, antihypertensive agents or other vasodilators may increase the risk of symptomatic hypotension.
Anticoagulants
- Since treprostinil inhibits platelet aggregation, there may be an increased risk of bleeding, particularly among patients receiving anticoagulants.
Pharmacokinetics
Bosentan
- In a human pharmacokinetic study conducted with bosentan (250 mg/day) and an oral formulation of treprostinil (treprostinil diethanolamine), no pharmacokinetic interactions between treprostinil and bosentan were observed.
Sildenafil
- In a human pharmacokinetic study conducted with sildenafil (60 mg/day) and an oral formulation of treprostinil (treprostinil diethanolamine), no pharmacokinetic interactions between treprostinil and sildenafil were observed.
Effect of Treprostinil on Cytochrome P450 Enzymes
- In vitro studies of human hepatic microsomes showed that treprostinil does not inhibit cytochrome P450 (CYP) isoenzymes CYP1A2, CYP2A6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1 and CYP3A. Additionally, treprostinil does not induce cytochrome P450 isoenzymes CYP1A2, CYP2B6, CYP2C9, CYP2C19, and CYP3A. Thus Treprostinil is not expected to alter the pharmacokinetics of compounds metabolized by CYP enzymes.
Effect of Cytochrome P450 Inhibitors and Inducers on Treprostinil
- Human pharmacokinetic studies with an oral formulation of treprostinil (treprostinil diethanolamine) indicated that co-administration of the cytochrome P450 (CYP) 2C8 enzyme inhibitor gemfibrozil increases exposure (both Cmax and AUC) to treprostinil. Co-administration of the CYP2C8 enzyme inducer rifampin decreases exposure to treprostinil. It has not been determined if the safety and efficacy of treprostinil by the parenteral (subcutaneously or intravenously) route are altered by inhibitors or inducers of CYP2C8.
- Treprostinil has not been studied in conjunction with Flolan or Tracleer® (bosentan).
Effect of Other Drugs on Treprostinil
- Drug interaction studies have been carried out with treprostinil (oral or subcutaneous) co-administered with acetaminophen (4 g/day), warfarin (25 mg/day), and fluconazole (200 mg/day), respectively in healthy volunteers. These studies did not show a clinically significant effect on the pharmacokinetics of treprostinil. Treprostinil does not affect the pharmacokinetics or pharmacodynamics of warfarin. The pharmacokinetics of R- and S- warfarin and the INR in healthy subjects given a single 25 mg dose of warfarin were unaffected by continuous subcutaneous infusion of treprostinil at an infusion rate of 10 ng/kg/min.
Use in Specific Populations
Pregnancy
- In pregnant rats, continuous subcutaneous infusions of treprostinil during organogenesis and late gestational development, at rates as high as 900 ng treprostinil/kg/min (about 117 times the starting human rate of infusion, on a ng/m2 basis and about 16 times the average rate achieved in clinical trials), resulted in no evidence of harm to the fetus. In pregnant rabbits, effects of continuous subcutaneous infusions of treprostinil during organogenesis were limited to an increased incidence of fetal skeletal variations (bilateral full rib or right rudimentary rib on lumbar 1) associated with maternal toxicity (reduction in body weight and food consumption) at an infusion rate of 150 ng treprostinil/kg/min (about 41 times the starting human rate of infusion, on a ng/m2 basis, and 5 times the average rate used in clinical trials). In rats, continuous subcutaneous infusion of treprostinil from implantation to the end of lactation, at rates of up to 450 ng treprostinil/kg/min, did not affect the growth and development of offspring. Because animal reproduction studies are not always predictive of human response, Treprostinil should be used during pregnancy only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Treprostinil (injection) in women who are pregnant.
Labor and Delivery
- No treprostinil treatment-related effects on labor and delivery were seen in animal studies. The effect of treprostinil sodium on labor and delivery in humans is unknown.
Nursing Mothers
- It is not known whether treprostinil is excreted in human milk or absorbed systemically after ingestion. Because many drugs are excreted in human milk, caution should be exercised when Treprostinil is administered to nursing women.
Pediatric Use
- Safety and effectiveness in pediatric patients have not been established. Clinical studies of Treprostinil did not include sufficient numbers of patients aged ≤16 years to determine whether they respond differently from older patients. In general, dose selection should be cautious.
Geriatic Use
- Clinical studies of Treprostinil did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Gender
- There is no FDA guidance on the use of Treprostinil (injection) with respect to specific gender populations.
Race
- There is no FDA guidance on the use of Treprostinil (injection) with respect to specific racial populations.
Renal Impairment
- No studies have been performed in patients with renal insufficiency. No specific advice about dosing in patients with renal impairment can be given
Hepatic Impairment
- Treprostinil clearance is reduced in patients with hepatic insufficiency. In patients with mild or moderate hepatic insufficiency, decrease the initial dose of Treprostinil to 0.625 ng/kg/min ideal body weight, and monitor closely. Treprostinil has not been studied in patients with severe hepatic insufficiency
Females of Reproductive Potential and Males
- There is no FDA guidance on the use of Treprostinil (injection) in women of reproductive potentials and males.
Immunocompromised Patients
- There is no FDA guidance one the use of Treprostinil (injection) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. If either particulate matter or discoloration is noted, Treprostinil should not be administered.
Subcutaneous Infusion
- Treprostinil is administered subcutaneously by continuous infusion, via a self-inserted subcutaneous catheter, using an infusion pump designed for subcutaneous drug delivery. To avoid potential interruptions in drug delivery, the patient must have immediate access to a backup infusion pump and subcutaneous infusion sets. The ambulatory infusion pump used to administer Treprostinil should: (1) be small and lightweight, (2) be adjustable to approximately 0.002 mL/hr, (3) have occlusion/no delivery, low battery, programming error and motor malfunction alarms, (4) have delivery accuracy of ±6% or better and (5) be positive pressure driven. The reservoir should be made of polyvinyl chloride, polypropylene or glass.
- For subcutaneous infusion, Treprostinil is delivered without further dilution at a calculated Subcutaneous Infusion Rate (mL/hr) based on a patients Dose (ng/kg/min), Weight (kg), and the Vial Strength (mg/mL) of Treprostinil being used. During use, a single reservoir (syringe) of undiluted Treprostinil can be administered up to 72 hours at 37°C. The Subcutaneous Infusion rate is calculated using the following formula:

- Example 1:
For a 60 kg person at the recommended initial dose of 1.25 ng/kg/min using the 1 mg/mL Treprostinil Vial Strength, the infusion rate would be calculated as follows:
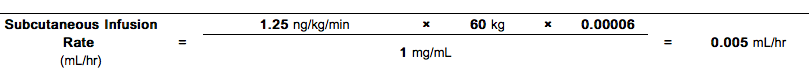
- Example 2:
For a 65 kg person at a dose of 40 ng/kg/min using the 5 mg/mL Treprostinil Vial Strength, the infusion rate would be calculated as follows:

Intravenous Infusion
- Treprostinil must be diluted with either Sterile Water for Injection, 0.9% Sodium Chloride Injection, Sterile Diluent for Flolan, or Sterile Diluent for Epoprostenol Sodium and is administered intravenously by continuous infusion, via a surgically placed indwelling central venous catheter, using an infusion pump designed for intravenous drug delivery. If clinically necessary, a temporary peripheral intravenous cannula, preferably placed in a large vein, may be used for short term administration of Treprostinil. Use of a peripheral intravenous infusion for more than a few hours may be associated with an increased risk of thrombophlebitis. To avoid potential interruptions in drug delivery, the patient must have immediate access to a backup infusion pump and infusion sets. The ambulatory infusion pump used to administer Treprostinil should: (1) be small and lightweight, (2) have occlusion/no delivery, low battery, programming error and motor malfunction alarms, (3) have delivery accuracy of ±6% or better of the hourly dose, and (4) be positive pressure driven. The reservoir should be made of polyvinyl chloride, polypropylene or glass.
- Infusion sets with an in-line 0.22 or 0.2 micron pore size filter should be used.
- Diluted Treprostinil has been shown to be stable at ambient temperature for up to 48 hours at concentrations as low as 0.004 mg/mL (4,000 ng/mL).
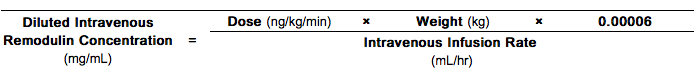
- When using an appropriate infusion pump and reservoir, a predetermined intravenous infusion rate should first be selected to allow for a desired infusion period length of up to 48 hours between system changeovers. Typical intravenous infusion system reservoirs have volumes of 50 or 100 mL. With this selected Intravenous Infusion Rate (mL/hr) and the patient's Dose (ng/kg/min) and Weight (kg), the Diluted Intravenous Treprostinil Concentration (mg/mL) can be calculated using the following formula:
Step 1
- The Amount of Treprostinil Injection needed to make the required Diluted Intravenous Treprostinil Concentration for the given reservoir size can then be calculated using the following formula:
Step 2

- The calculated amount of Treprostinil Injection is then added to the reservoir along with the sufficient volume of diluent (Sterile Water for Injection, 0.9% Sodium Chloride Injection, Sterile Diluent for Flolan, or Sterile Diluent for Epoprostenol Sodium) to achieve the desired total volume in the reservoir.
Example calculations for Intravenous Infusion are as follows:
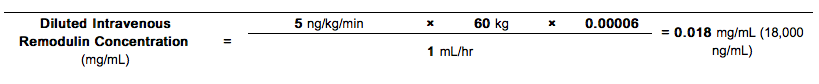
- Example 3:
For a 60 kg person at a dose of 5 ng/kg/min, with a predetermined intravenous infusion rate of 1 mL/hr and a reservoir of 50 mL, the Diluted Intravenous Treprostinil Solution Concentration would be calculated as follows:
Step 1
- The Amount of Treprostinil Injection (using 1 mg/mL Vial Strength) needed for a total Diluted Treprostinil Concentration of 0.018 mg/mL and a total volume of 50 mL would be calculated as follows:
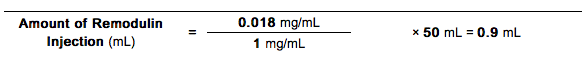
- The Diluted Intravenous Treprostinil Concentration for the person in Example 3 would thus be prepared by adding 0.9 mL of 1 mg/mL Treprostinil Injection to a suitable reservoir along with a sufficient volume of diluent to achieve a total volume of 50 mL in the reservoir. The pump flow rate for this example would be set at 1 mL/hr.
- Example 4:
For a 75 kg person at a dose of 30 ng/kg/min, with a predetermined intravenous infusion rate of 2 mL/hr, and a reservoir of 100 mL, the Diluted Intravenous Treprostinil Solution Concentration would be calculated as follows:
Step 1

- The Amount of Treprostinil Injection (using 2.5 mg/mL Vial Strength) needed for a total Diluted Treprostinil Concentration of 0.0675 mg/mL and a total volume of 100 mL would be calculated as follows:
Step 2
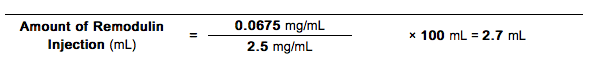
- The Diluted Intravenous Treprostinil Concentration for the person in Example 4 would thus be prepared by adding 2.7 mL of 2.5 mg/mL Treprostinil Injection to a suitable reservoir along with a sufficient volume of diluent to achieve a total volume of 100 mL in the reservoir. The pump flow rate for this example would be set at 2 mL/hr.
Patients Requiring Transition from Flolan
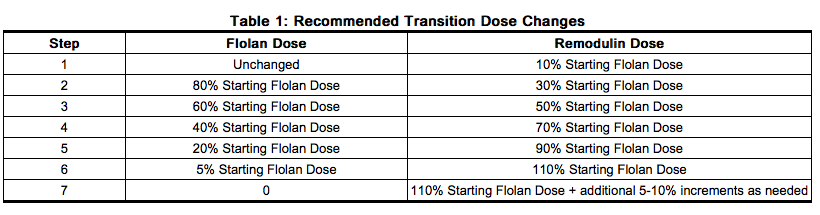
- Transition from Flolan to Treprostinil is accomplished by initiating the infusion of Treprostinil and increasing it, while simultaneously reducing the dose of intravenous Flolan. The transition to Treprostinil should take place in a hospital with constant observation of response (e.g., walk distance and signs and symptoms of disease progression). During the transition, Treprostinil is initiated at a recommended dose of 10% of the current Flolan dose, and then escalated as the Flolan dose is decreased (see Table 1 for recommended dose titrations).
- Patients are individually titrated to a dose that allows transition from Flolan therapy to Treprostinil while balancing prostacyclin-limiting adverse events. Increases in the patient's symptoms of PAH should be first treated with increases in the dose of Treprostinil. Side effects normally associated with prostacyclin and prostacyclin analogs are to be first treated by decreasing the dose of Flolan.
Monitoring
- There is limited information regarding Monitoring of Treprostinil (injection) in the drug label.
IV Compatibility
- There is limited information regarding IV Compatibility of Treprostinil (injection) in the drug label.
Overdosage
- Signs and symptoms of overdose with Treprostinil during clinical trials are extensions of its dose-limiting pharmacologic effects and include flushing, headache, hypotension, nausea, vomiting, and diarrhea. Most events were self-limiting and resolved with reduction or withholding of Treprostinil.
- In controlled clinical trials, seven patients received some level of overdose and in open-label follow-on treatment seven additional patients received an overdose; these occurrences resulted from accidental bolus administration of Treprostinil, errors in pump programmed rate of administration, and prescription of an incorrect dose. In only two cases did excess delivery of Treprostinil produce an event of substantial hemodynamic concern (hypotension, near-syncope).
- One pediatric patient was accidentally administered 7.5 mg of Treprostinil via a central venous catheter. Symptoms included flushing, headache, nausea, vomiting, hypotension and seizure-like activity with loss of consciousness lasting several minutes. The patient subsequently recovered.
Pharmacology
Mechanism of Action
- The major pharmacologic actions of treprostinil are direct vasodilation of pulmonary and systemic arterial vascular beds, and inhibition of platelet aggregation.
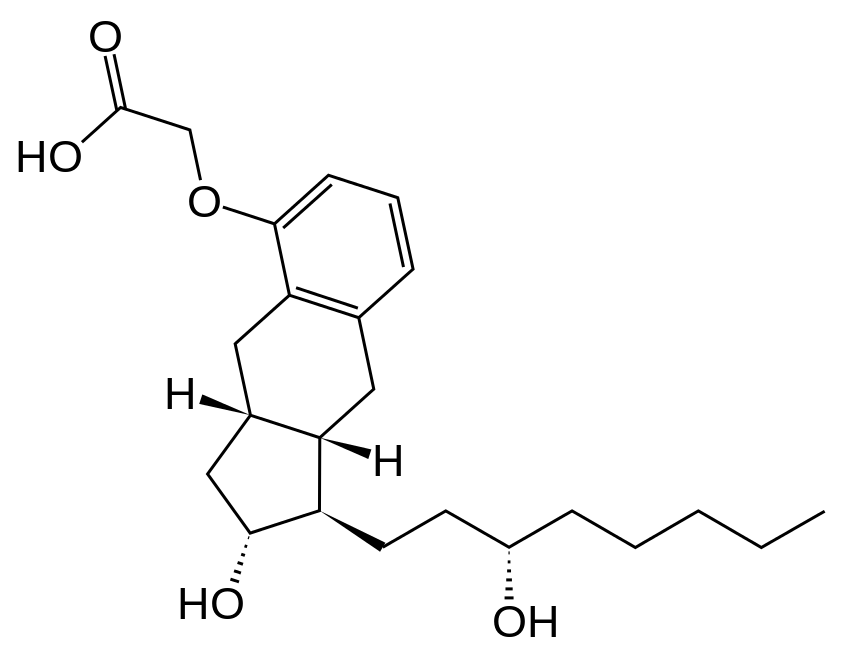
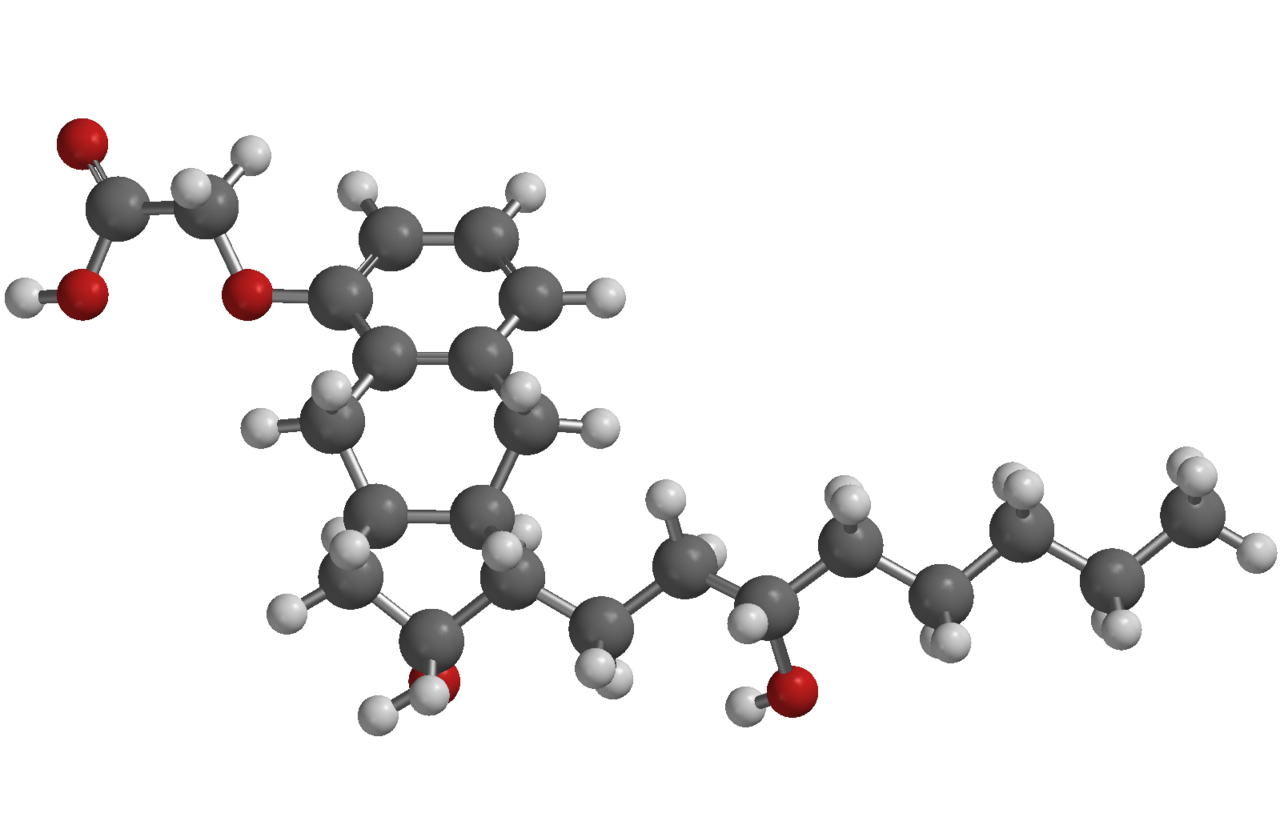
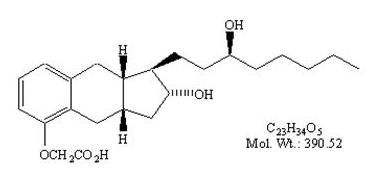
Structure
Pharmacodynamics
- In animals, the vasodilatory effects reduce right and left ventricular afterload and increase cardiac output and stroke volume. Other studies have shown that treprostinil causes a dose-related negative inotropic and lusitropic effect. No major effects on cardiac conduction have been observed.
- Treprostinil produces vasodilation and tachycardia. Single doses of treprostinil up to 84 mcg by inhalation produce modest and short-lasting effects on QTc, but this is apt to be an artifact of the rapidly changing heart rate. Treprostinil administered by the subcutaneous or intravenous routes has the potential to generate concentrations many-fold greater than those generated via the inhaled route; the effect on the QTc interval when treprostinil is administered parenterally has not been established.
Pharmacokinetics
- The pharmacokinetics of continuous subcutaneous Treprostinil are linear over the dose range of 1.25 to 125 ng/kg/min (corresponding to plasma concentrations of about 15 pg/mL to 18,250 pg/mL) and can be described by a two-compartment model. Dose proportionality at infusion rates greater than 125 ng/kg/min has not been studied.
- Subcutaneous and intravenous administration of Treprostinil demonstrated bioequivalence at steady state at a dose of 10 ng/kg/min.
Absorption
- Treprostinil is relatively rapidly and completely absorbed after subcutaneous infusion, with an absolute bioavailability approximating 100%. Steady-state concentrations occurred in approximately 10 hours. Concentrations in patients treated with an average dose of 9.3 ng/kg/min were approximately 2,000 pg/mL.
Distribution
- The volume of distribution of the drug in the central compartment is approximately 14L/70 kg ideal body weight. Treprostinil at in vitro concentrations ranging from 330-10,000 mcg/L was 91% bound to human plasma protein.
Metabolism and Excretion
- Treprostinil is substantially metabolized by the liver, primarily by CYP2C8. In a study conducted in healthy volunteers using [14C] treprostinil, 78.6% and 13.4% of the subcutaneous dose was recovered in the urine and feces, respectively, over 10 days. Only 4% was excreted as unchanged treprostinil in the urine. Five metabolites were detected in the urine, ranging from 10.2% to 15.5% and representing 64.4% of the dose administered. Four of the metabolites are products of oxidation of the 3-hydroxyloctyl side chain and one is a glucuroconjugated derivative (treprostinil glucuronide). The identified metabolites do not appear to have activity.
- The elimination of treprostinil (following subcutaneous administration) is biphasic, with a terminal elimination half-life of approximately 4 hours using a two compartment model. Systemic clearance is approximately 30 L/hr for a 70 kg person.
Based on in vitro studies treprostinil does not inhibit or induce major CYP enzymes
Special Populations
Hepatic Insufficiency
- In patients with portopulmonary hypertension and mild (n=4) or moderate (n=5) hepatic insufficiency, Treprostinil at a subcutaneous dose of 10 ng/kg/min for 150 minutes had a Cmax that was increased 2-fold and 4-fold, respectively, and an AUC 0-∞ that was increased 3-fold and 5-fold, respectively, compared to healthy subjects. Clearance in patients with hepatic insufficiency was reduced by up to 80% compared to healthy adults.
Renal Insufficiency
- No studies have been performed in patients with renal insufficiency, so no specific advice about dosing in such patients can be given. Although only 4% of the administered dose is excreted unchanged in the urine, the five identified metabolites are all excreted in the urine.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Long-term studies have not been performed to evaluate the carcinogenic potential of treprostinil. In vitro and in vivo genetic toxicology studies did not demonstrate any mutagenic or clastogenic effects of treprostinil. Treprostinil did not affect fertility or mating performance of male or female rats given continuous subcutaneous infusions at rates of up to 450 ng treprostinil/kg/min [about 59 times the recommended starting human rate of infusion (1.25 ng/kg/min) and about 8 times the average rate (9.3 ng/kg/min) achieved in clinical trials, on a ng/m2 basis]. In this study, males were dosed from 10 weeks prior to mating and through the 2-week mating period. Females were dosed from 2 weeks prior to mating until gestational day 6.
Clinical Studies
Clinical Trials in Pulmonary Arterial Hypertension (PAH)
- Two 12-week, multicenter, randomized, double-blind studies compared continuous subcutaneous infusion of Treprostinil to placebo in a total of 470 patients with NYHA Class II (11%), III (81%), or IV (7%) pulmonary arterial hypertension (PAH). PAH was idiopathic/heritable in 58% of patients, associated with connective tissue diseases in 19%, and the result of congenital systemic-to-pulmonary shunts in 23%. The mean age was 45 (range 9 to 75 years). About 81% were female and 84% were Caucasian. Pulmonary hypertension had been diagnosed for a mean of 3.8 years. The primary endpoint of the studies was change in 6-minute walking distance, a standard measure of exercise capacity. There were many assessments of symptoms related to heart failure, but local discomfort and pain associated with Treprostinil may have substantially unblinded those assessments. The 6-minute walking distance and an associated subjective measurement of shortness of breath during the walk (Borg dyspnea score) were administered by a person not participating in other aspects of the study. Treprostinil was administered as a subcutaneous infusion,and the dose averaged 9.3 ng/kg/min at Week 12. Few subjects received doses > 40 ng/kg/min. Background therapy, determined by the investigators, could include anticoagulants, oral vasodilators, diuretics, digoxin, and oxygen but not an endothelin receptor antagonist or epoprostenol. The two studies were identical in design and conducted simultaneously, and the results were analyzed both pooled and individually.
Hemodynamic Effects
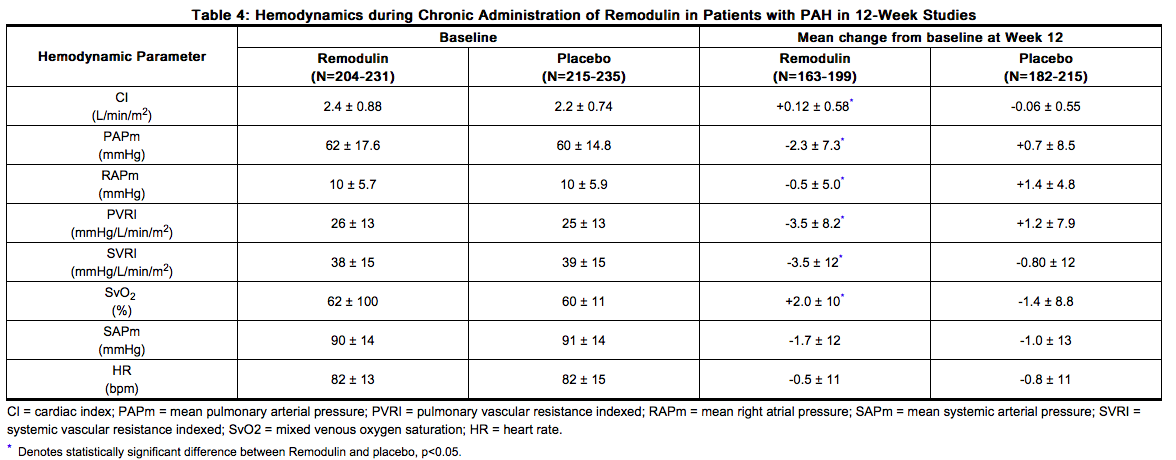
- As shown in Table 4, chronic therapy with Treprostinil resulted in small hemodynamic changes consistent with pulmonary and systemic vasodilation.

Clinical Effects
- The effect of Treprostinil on 6-minute walk, the primary end point of the 12-week studies, was small and did not achieve conventional levels of statistical significance. For the combined populations, the median change from baseline on Treprostinil was 10 meters and the median change from baseline on placebo was 0 meters from a baseline of approximately 345 meters. Although it was not the primary endpoint of the study, the Borg dyspnea score was significantly improved by Treprostinil during the 6-minute walk, and Treprostinil also had a significant effect, compared with placebo, on an assessment that combined walking distance with the Borg dyspnea score. Treprostinil also consistently improved indices of dyspnea, fatigue and signs and symptoms of pulmonary hypertension, but these indices were difficult to interpret in the context of incomplete blinding to treatment assignment resulting from infusion site symptoms.
Flolan-To-Treprostinil Transition Study
- In an 8-week, multicenter, randomized, double-blind, placebo-controlled study, patients on stable doses of Flolan were randomly withdrawn from Flolan to placebo or Treprostinil. Fourteen Treprostinil and 8 placebo patients completed the study. The primary endpoint of the study was the time to clinical deterioration, defined as either an increase in Flolan dose, hospitalization due to PAH, or death. No patients died during the study.
- During the study period, Treprostinil effectively prevented clinical deterioration in patients transitioning from Flolan therapy compared to placebo (Figure 1). Thirteen of 14 patients in the Treprostinil arm were able to transition from Flolan successfully, compared to only 1 of 8 patients in the placebo arm (p=0.0002).
How Supplied
- Treprostinil is supplied in 20 mL multidose vials containing 20, 50, 100, or 200 mg of treprostinil at concentrations of 1 mg/mL, 2.5 mg/mL, 5 mg/mL, and 10 mg/mL treprostinil, respectively, as sterile solutions in water for injection, individually packaged in cartons. Unopened vials of Treprostinil are stable until the date indicated when stored at 15 to 25°C (59 to 77°F). Store at 25°C (77°F), with excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
- Treprostinil Injection is supplied as:
- 20-mL vial containing 20 mg treprostinil (1 mg treprostinil per mL), carton of 1 (NDC 66302-101-01).
- 20-mL vial containing 50 mg treprostinil (2.5 mg treprostinil per mL), carton of 1 (NDC 66302-102-01).
- 20-mL vial containing 100 mg treprostinil (5 mg treprostinil per mL), carton of 1 (NDC 66302-105-01).
- 20-mL vial containing 200 mg treprostinil (10 mg treprostinil per mL), carton of 1 (NDC 66302-110-01).
Storage
- During use, a single reservoir (syringe) of undiluted Treprostinil can be administered up to 72 hours at 37°C. Diluted Treprostinil Solution can be administered up to 48 hours at 37°C when diluted to concentrations as low as 0.004 mg/mL in Sterile Water for Injection, 0.9% Sodium Chloride Injection, Sterile Diluent for Flolan or Sterile Diluent for Epoprostenol Sodium. A single vial of Treprostinil should be used for no more than 30 days after the initial introduction into the vial.
Images
Drug Images
{{#ask: Page Name::Treprostinil (injection) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Treprostinil (injection) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patients receiving Treprostinil should be given the following information: Treprostinil is infused continuously through a subcutaneous or surgically placed indwelling central venous catheter, via an infusion pump. Patients receiving intravenous infusion should use an infusion set with an in-line filter. Therapy with Treprostinil will be needed for prolonged periods, possibly years, and the patient's ability to accept and care for a catheter and to use an infusion pump should be carefully considered. In order to reduce the risk of infection, aseptic technique must be used in the preparation and administration of Treprostinil. Additionally, patients should be aware that subsequent disease management may require the initiation of an alternative intravenous prostacyclin therapy, Flolan® (epoprostenol sodium).
Precautions with Alcohol
- Alcohol-Treprostinil (injection) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Treprostinil[1]
Look-Alike Drug Names
- Tyvaso
- Remodulin
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Treprostinil (injection)
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Treprostinil (injection) |Label Name=Treprostinil_label_01.jpg
}}
{{#subobject:
|Label Page=Treprostinil (injection) |Label Name=Treprostinil_label_02.jpg
}}
{{#subobject:
|Label Page=Treprostinil (injection) |Label Name=Treprostinil_label_03.jpg
}}
{{#subobject:
|Label Page=Treprostinil (injection) |Label Name=Treprostinil_label_04.jpg
}}
{{#subobject:
|Label Page=Treprostinil (injection) |Label Name=Treprostinil_panel_01.png
}}
{{#subobject:
|Label Page=Treprostinil (injection) |Label Name=Treprostinil_panel_02.png
}}
{{#subobject:
|Label Page=Treprostinil (injection) |Label Name=Treprostinil_panel_03.png
}}
{{#subobject:
|Label Page=Treprostinil (injection) |Label Name=Treprostinil_panel_04.png
}}