Topotecan (injection)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: BONE MARROW SUPPRESSION
See full prescribing information for complete Boxed Warning.
|
Overview
Topotecan (injection) is a topoisomerase I inhibitor that is FDA approved for the treatment of small cell lung cancer, stage IV-B, recurrent, or persistent carcinoma of the cervix. There is a Black Box Warning for this drug as shown here. Common adverse reactions include bleeding, anaphylactoid reactions, abdominal pain, Interstitial lung disease, and pruritus.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Topotecan Injection is indicated for the treatment of
- Small cell lung cancer sensitive disease after failure of first-line chemotherapy. In clinical studies submitted to support approval, sensitive disease was defined as disease responding to chemotherapy but subsequently progressing at least 60 days (in the Phase 3 study) or at least 90 days (in the Phase 2 studies) after chemotherapy.
- Topotecan Injection in combination with cisplatin is indicated for the treatment of
- stage IV-B, recurrent, or persistent carcinoma of the cervix which is not amenable to curative treatment with surgery and/or radiation therapy.
- Prior to administration of the first course of Topotecan Injection, patients must have a baseline neutrophil count of >1,500 cells/mm3 and a platelet count of >100,000 cells/mm3.
Small Cell Lung Cancer
Recommended Dosage
- The recommended dose of topotecan is 1.5 mg/m2 by intravenous infusion over 30 minutes daily for 5 consecutive days, starting on day 1 of a 21-day course.
- In the absence of tumor progression, a minimum of 4 courses is recommended because tumor response may be delayed. The median time to response in 4 small cell lung cancer trials was 5 to 7 weeks.
Dosage Modification Guidelines
- In the event of severe neutropenia (defined as <500 cells/mm3) during any course, reduce the dose by 0.25 mg/m2 (to 1.25 mg/m2) for subsequent courses.
- Alternatively, in the event of severe neutropenia, administer G-CSF (granulocyte-colony stimulating factor) following the subsequent course (before resorting to dose reduction) starting from day 6 of the course (24 hours after completion of topotecan administration).
- In the event the platelet count falls below 25,000 cells/mm3, reduce doses by 0.25 mg/m2 (to 1.25 mg/m2) for subsequent courses.
Cervical Cancer
Recommended Dosage
- The recommended dose of Topotecan Injection is 0.75 mg/m2 by intravenous infusion over 30 minutes daily on days 1, 2, and 3; followed by cisplatin 50 mg/m2 by intravenous infusion on day 1 repeated every 21 days (a 21-day course).
Dosage Modification Guidelines
- Dosage adjustments for subsequent courses of Topotecan Injection in combination with cisplatin are specific for each drug. See manufacturer’s prescribing information for cisplatin administration and hydration guidelines and for cisplatin dosage adjustment in the event of hematologic toxicity.
- In the event of severe febrile neutropenia (defined as <1000 cells/mm3 with temperature of 38°C or 100.4°F), reduce the dose of Topotecan Injection to 0.60 mg/m2 for subsequent courses.
- Alternatively, in the event of severe febrile neutropenia, administer G-CS following the subsequent course (before resorting to dose reduction) starting from day 4 of the course (24 hours after completion of administration of Topotecan Injection).
- If febrile neutropenia occurs despite the use of G-CSF, reduce the dose of Topotecan Injection to 0.45 mg/m2 for subsequent courses.
- In the event the platelet count falls below 25,000 cells/mm3, reduce doses to 0.60 mg/m2 for subsequent courses.
Dosage Adjustment in Specific Populations
Renal Impairment
- No dosage adjustment of Topotecan Injection appears to be required for patients with mild renal impairment (Clcr 40 to 60 mL/min.). Dosage adjustment of Topotecan Injection to 0.75 mg/m2 is recommended for patients with moderate renal impairment (20 to 39 mL/min.). Insufficient data are available in patients with severe renal impairment to provide a dosage recommendation for Topotecan Injection.
- Topotecan Injection in combination with cisplatin for the treatment of cervical cancer should only be initiated in patients with serum creatinine ≤1.5 mg/dL. In the clinical trial, cisplatin was discontinued for a serum creatinine >1.5 mg/dL. Insufficient data are available regarding continuing monotherapy with Topotecan Injection after cisplatin discontinuation in patients with cervical cancer.
Instructions for Handling, Preparation and Intravenous Administration
- Handling
- Topotecan is a cytotoxic anticancer drug. Prepare Topotecan Injection under a vertical laminar flow hood while wearing gloves and protective clothing. If Topotecan Injection solution contacts the skin, wash the skin immediately and thoroughly with soap and water. If Topotecan Injection contacts mucous membranes, flush thoroughly with water.
- Use procedures for proper handling and disposal of anticancer drugs. Several guidelines on this subject have been published.1-4
Preparation and Administration
- The appropriate volume of the Topotecan Injection is diluted in a minimum of 50 mL of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP prior to administration. Infuse over 30 minutes. Topotecan Injection diluted for infusion is stable for 4 hours at room temperature or 24 hours at refrigerated temperature in ambient lighting conditions.
- Topotecan Injection is supplied as a multiple dose vial. Studies have shown the product is stable for 28 days after initial puncture when stored under refrigerated conditions.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Topotecan (injection) in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Topotecan (injection) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Topotecan (injection) in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Topotecan (injection) in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Topotecan (injection) in pediatric patients.
Contraindications
- Topotecan Injection is contraindicated in patients who have a history of severe hypersensitivity reactions (e.g., anaphylactoid reactions) to topotecan or to any of its ingredients. Topotecan Injection should not be used in patients with severe bone marrow depression.
Warnings
|
WARNING: BONE MARROW SUPPRESSION
See full prescribing information for complete Boxed Warning.
|
Bone Marrow Suppression
- Bone marrow suppression (primarily neutropenia) is the dose-limiting toxicity of Topotecan Injection. Neutropenia is not cumulative over time. In the comparative study, in small cell lung cancer, the treatment-related death rates were 5% for topotecan and 4% for CAV (cyclophosphamide-doxorubicin-vincristine).
Neutropenia
- From a combined experience of patients receiving topotecan which included patients treated for small cell lung cancer: Grade 4 neutropenia (<500 cells/mm3) was most common during course 1 of treatment (60% of patients) and occurred in 39% of all courses, with a median duration of 7 days. The nadir neutrophil count occurred at a median of 12 days. Therapy-related sepsis or ebrile neutropenia occurred in 23% of patients, and sepsis was fatal in 1%. Pancytopenia has been reported.
- Cervical cancer experience
- Grade 3 and grade 4 neutropenia affected 26% and 48% of patients, respectively.
Thrombocytopenia
- From a combined experience of patients receiving topotecan which included patients treated for small cell lung cancer: Grade 4 thrombocytopenia (<25,000/mm3) occurred in 27% of patients and in 9% of courses, with a median duration of 5 days and platelet nadir at a median of 15 days. Platelet transfusions were given to 15% of patients in 4% of courses.
Cervical cancer experience: Grade 3 and grade 4 thrombocytopenia affected 26% and 7% of patients, respectively.
Anemia
- From a combined experience of patients receiving topotecan which included patients treated for small cell lung cancer: Grade 3/4 anemia (<8 g/dL) occurred in 37% of patients and in 14% of courses. Median nadir was at day 15. Transfusions were needed in 52% of patients in 22% of courses.
Cervical cancer experience: Grade 3 and grade 4 anemia affected 34% and 6% of patients, respectively.
Monitoring of Bone Marrow Function
- Administer Topotecan Injection only in patients with adequate bone marrow reserves, including baseline neutrophil count of at least 1,500 cells/mm3 and platelet count at least 100,000/mm3.
- Monitor peripheral blood counts frequently during treatment with Topotecan Injection. Do not treat patients with subsequent courses of Topotecan Injection until neutrophils recover to >1,000 cells/mm3, platelets recover to >100,000 cells/mm3, and hemoglobin levels recover to 9 g/dL (with transfusion if necessary). Severe myelotoxicity has been reported when Topotecan Injection is used in combination with cisplatin.
Neutropenic Colitis
- Topotecan-induced neutropenia can lead to neutropenic colitis. Fatalities due to neutropenic colitis have been reported in clinical trials with Topotecan Injection. In patients presenting with fever, neutropenia, and a compatible pattern of abdominal pain, consider the possibility of neutropenic colitis.
Interstitial Lung Disease
- Topotecan Injection has been associated with reports of interstitial lung disease (ILD), some of which have been fatal. Underlying risk factors include history of ILD, pulmonary fibrosis, lung cancer, thoracic exposure to radiation, and use of pneumotoxic drugs and/or colony stimulating factors. Monitor patients for pulmonary symptoms indicative of interstitial lung disease (e.g., cough, fever, dyspnea, and/or hypoxia), and discontinue Topotecan Injection if a new diagnosis of ILD is confirmed.
Pregnancy
- Pregnancy Category D
- Topotecan Injection can cause fetal harm when administered to a pregnant woman.
- Topotecan caused embryolethality, fetotoxicity, and teratogenicity in rats and rabbits when administered during organogenesis. There are no adequate and well controlled studies of Topotecan Injection in pregnant women. If this drug is used during pregnancy, or if a patient becomes pregnant while receiving Topotecan Injection, the patient should be apprised of the potential hazard to the fetus.
Inadvertent Extravasation
- Inadvertent extravasation with topotecan has been observed, most reactions have been mild but severe cases have been reported.
Adverse Reactions
Clinical Trials Experience
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Small Cell Lung Cancer
- Data in the following section are based on the combined experiences of the 879 patients studied, including 426 patients with small cell lung cancer treated with topotecan. Table 1 lists the principle hematologic adverse reactions and Table 2 lists non-hematologic adverse reactions occurring in at least 15% of patients.
- Table 1. Hematologic Adverse Reactions Experienced in ≥15% of Patients, Including 426 Patients With Small Cell Lung Cancer, Receiving Topotecan
Postmarketing Experience
- In addition to adverse reactions reported from clinical trials or listed in other sections of the prescribing information, the following reactions have been identified during post-marketing use of Topotecan Injection. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made.
- These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to Topotecan Injection.
- Blood and Lymphatic System Disorders
- Severe bleeding (in association with thrombocytopenia).
- Immune System Disorders
- Allergic manifestations; Anaphylactoid reactions.
- Gastrointestinal Disorders
- Abdominal pain potentially associated with neutropenic colitis.
- Pulmonary Disorders
- Skin and Subcutaneous Tissue Disorders
- Angioedema, severe dermatitis, severe pruritus
- General Disorders and Administration Site Conditions
- Inadvertent extravasation
Drug Interactions
Bone Marrow Suppression
- Bone marrow suppression (primarily neutropenia) is the dose-limiting toxicity of Topotecan Injection. Neutropenia is not cumulative over time. In the comparative study, in small cell lung cancer, the treatment-related death rates were 5% for topotecan and 4% for CAV (cyclophosphamide-doxorubicin-vincristine).
Neutropenia
- From a combined experience of patients receiving topotecan which included patients treated for small cell lung cancer: Grade 4 neutropenia occurred in 39% of all courses, with a median duration of 7 days. The nadir neutrophil count occurred at a median of 12 days. Therapy-related sepsis or febrile eutropenia occurred in 23% of patients, and sepsis was fatal in 1%. Pancytopenia has been reported.
- Cervical cancer experience
- Grade 3 and grade 4 neutropenia affected 26% and 48% of patients, respectively.
Thrombocytopenia
- From a combined experience of patients receiving topotecan which included patients treated for small cell lung cancer: Grade 4 thrombocytopenia (<25,000/mm3) occurred in 27% of patients and in 9% of courses, with a median duration of 5 days and platelet nadir at a median of 15 days. Platelet transfusions were given to 15% of patients in 4% of courses.
Cervical cancer experience: Grade 3 and grade 4 thrombocytopenia affected 26% and 7% of patients, respectively.
Anemia
- From a combined experience of patients receiving topotecan which included patients treated for small cell lung cancer: Grade 3/4 anemia (<8 g/dL) occurred in 37% of patients and in 14% of courses. Median nadir was at day 15. Transfusions were needed in 52% of patients in 22% of courses.
Cervical cancer experience: Grade 3 and grade 4 anemia affected 34% and 6% of patients, respectively.
Monitoring of Bone Marrow Function
- Administer Topotecan Injection only in patients with adequate bone marrow reserves, including baseline neutrophil count of at least 1,500 cells/mm3 and platelet count at least 100,000/mm3.
- Monitor peripheral blood counts frequently during treatment with Topotecan Injection. Do not treat patients with subsequent courses of Topotecan Injection until neutrophils recover to >1,000 cells/mm3, platelets recover to >100,000 cells/mm3, and hemoglobin levels recover to 9 g/dL (with transfusion if necessary). Severe myelotoxicity has been reported when Topotecan Injection is used in combination with cisplatin.
Neutropenic Colitis
- Topotecan-induced neutropenia can lead to neutropenic colitis. Fatalities due to neutropenic colitis have been reported in clinical trials with Topotecan Injection. In patients presenting with fever, neutropenia, and a compatible pattern of abdominal pain, consider the possibility of neutropenic colitis.
Interstitial Lung Disease
- Topotecan Injection has been associated with reports of interstitial lung disease (ILD), some of which have been fatal [see Adverse Reactions (6.2)]. Underlying risk factors include history of ILD, pulmonary fibrosis, lung cancer, thoracic exposure to radiation, and use of pneumotoxic drugs and/or colony stimulating factors. Monitor patients for pulmonary symptoms indicative of interstitial lung disease (e.g., cough, fever, dyspnea, and/or hypoxia), and discontinue Topotecan Injection if a new diagnosis of ILD is confirmed.
Pregnancy
- Pregnancy Category D
- Topotecan Injection can cause fetal harm when administered to a pregnant woman.
- Topotecan caused embryolethality, fetotoxicity, and teratogenicity in rats and rabbits when administered during organogenesis. There are no adequate and well controlled studies of Topotecan Injection in pregnant women. If this drug is used during pregnancy, or if a patient becomes pregnant while receiving Topotecan Injection, the patient should be apprised of the potential hazard to the fetus.
Inadvertent Extravasation
- Inadvertent extravasation with topotecan has been observed, most reactions have been mild but severe cases have been reported.
Use in Specific Populations
Pregnancy
- Topotecan Injection can cause fetal harm when administered to a pregnant woman. In rabbits, a dose of 0.1 mg/kg/day (about equal to the clinical dose of 1.5 mg/m2) given on days 6 through 20 of gestation caused maternal toxicity, embryolethality, and reduced fetal body weight. In the rat, a dose of 0.23 mg/kg/day (about equal to the clinical dose of 1.5 mg/m2) given for 14 days before mating through gestation day 6 caused fetal resorption, microphthalmia, pre-implant loss, and mild maternal toxicity. A dose of 0.1 mg/kg/day (about half the clinical dose of 1.5 mg/m2) given to rats on days 6 through 17 of gestation caused an increase in post-implantation mortality. This dose also caused an increase in total fetal malformations. The most frequent malformations were of the eye (microphthalmia, anophthalmia, rosette formation of the retina, coloboma of the retina, ectopic orbit), brain (dilated lateral and third ventricles), skull, and vertebrae.
- There are no adequate and well controlled studies of Topotecan Injection in pregnant women. If this drug is used during pregnancy, or if a patient becomes pregnant while receiving Topotecan Injection, the patient should be apprised of the potential hazard to the fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Topotecan (injection) in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Topotecan (injection) during labor and delivery.
Nursing Mothers
- Rats excrete high concentrations of topotecan into milk. Lactating female rats given 4.72 mg/m2 IV (about three times the clinical dose of 1.5 mg/m2) excreted topotecan into milk at concentrations up to 48-fold higher than those in plasma. It is not known whether the drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from Topotecan Injection, discontinue breastfeeding when women are receiving Topotecan Injection.
Pediatric Use
There is no FDA guidance on the use of Topotecan (injection) with respect to pediatric patients.
Geriatic Use
- Of the 879 patients in a combined experience of topotecan which included patients with small cell lung cancer, 32% (n=281) were 65 years of age and older, while 3.8% (n=33) were 75 years of age and older.
- Of the 140 patients with stage IV-B, relapsed, or refractory cervical cancer in clinical studies of Topotecan Injection who received Topotecan Injection plus cisplatin in the randomized clinical trial, 6% (n = 9) were 65 years of age and older, while 3% (n = 4) were 75 years of age and older.
- No overall differences in effectiveness or safety were observed between these patients and younger adult patients, and other reported clinical experience has not identified differences in responses between the elderly and younger adult patients, but greater sensitivity of some older individuals cannot be ruled out.
- There were no apparent differences in the pharmacokinetics of topotecan in elderly patients, once the age-related decrease in renal function was considered.
- This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Gender
There is no FDA guidance on the use of Topotecan (injection) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Topotecan (injection) with respect to specific racial populations.
Renal Impairment
- No dosage adjustment of Topotecan Injection appears to be required for patients with mild renal impairment (Clcr 40 to 60 mL/min.). Dosage reduction is recommended for patients with moderate renal impairment (Clcr 20 to 39 mL/min.).
- Insufficient data are available in patients with severe renal impairment to provide a dosage recommendation for Topotecan Injection.
Hepatic Impairment
There is no FDA guidance on the use of Topotecan (injection) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Topotecan (injection) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Topotecan (injection) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral.
Monitoring
- Inform patients that frequent blood tests will be performed while taking Topotecan Injection to monitor for the occurrence of bone marrow suppression.
Monitoring of Bone Marrow Function
- Administer Topotecan Injection only in patients with adequate bone marrow reserves, including baseline neutrophil count of at least 1,500 cells/mm3 and platelet count at least 100,000/mm3.
- Monitor peripheral blood counts frequently during treatment with Topotecan Injection. Do not treat patients with subsequent courses of Topotecan Injection until neutrophils recover to >1,000 cells/mm3, platelets recover to >100,000 cells/mm3, and hemoglobin levels recover to 9 g/dL (with transfusion if necessary). Severe myelotoxicity has been reported when Topotecan Injection is used in combination with cisplatin.
- Monitor patients for pulmonary symptoms indicative of interstitial lung disease (e.g., cough, fever, dyspnea, and/or hypoxia), and discontinue Topotecan Injection if a new diagnosis of ILD is confirmed.
- Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
IV Compatibility
There is limited information regarding IV Compatibility of Topotecan (injection) in the drug label.
Overdosage
- There is no known antidote for overdosage with Topotecan Injection. The primary anticipated complication of overdosage would consist of bone marrow suppression.
- One patient on a single-dose regimen of 17.5 mg/m2 given on day 1 of a 21-day cycle had received a single dose of 35 mg/m2. This patient experienced severe neutropenia (nadir of 320/mm3) 14 days later but recovered without incident.
- Observe patients closely for bone marrow suppression, and consider supportive measures (such as the prophylactic use of G-CSF and/or antibiotic therapy).
Pharmacology
Mechanism of Action
- Topoisomerase I relieves torsional strain in DNA by inducing reversible single strand breaks. Topotecan binds to the topoisomerase I-DNA complex and prevents religation of these single strand breaks.
- The cytotoxicity of topotecan is thought to be due to double strand DNA damage produced during DNA synthesis, when replication enzymes interact with the ternary complex formed by topotecan, topoisomerase I, and DNA. Mammalian cells cannot efficiently repair these double strand breaks.
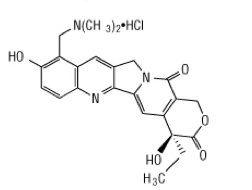
Structure
- Topotecan is a semi-synthetic derivative of camptothecin and is an anti-tumor drug with topoisomerase I-inhibitory activity.
- The chemical name for topotecan free base is (S)-10-[(dimethylamino)methyl]-4-ethyl-4,9-dihydroxy-1H-pyrano[3’,4’:6,7]indolizino[1,2-b]quinoline-3,14-(4H,12H)-dione. It has the molecular formula C23H23N3O5 and a molecular weight of 421.45.
- Topotecan has three pKa values: pKa1 = 10.50 corresponding to the benzyldimethylamino group, pKa2 = 6.99 corresponding to the phenol group and pKa3 = 0.60 corresponding to the quinoline group.
- As formulated in Topotecan Injection, topotecan has the following structural formula:
- where n is >1, corresponding to HCl added to adjust the pH to approximately 2.0 to 2.5.
- Topotecan Injection is supplied as a sterile, non-pyrogenic, clear, yellow solution at a topotecan free base concentration of 4 mg/4 mL (1 mg/mL), 3 mg/3 mL (1 mg/mL), and 1 mg/mL available in multiple dose vials.
- Each mL of Topotecan Injection contains topotecan hydrochloride equivalent to 1 mg of topotecan as free base, 5 mg tartaric acid, NF, and water for injection, USP. Hydrochloric acid and/or sodium hydroxide may be used to adjust the pH.. The hydrochloride salt of topotecan is soluble in water and melts with decomposition at 213°C to 218°C.
- The solution must be diluted before administration by intravenous infusion.
Pharmacodynamics
- The dose-limiting toxicity of topotecan is leukopenia. White blood cell count decreases with increasing topotecan dose or topotecan AUC. When topotecan is administered at a dose of 1.5 mg/m2/day for 5 days, an 80% to 90% decrease in white blood cell count at nadir is typically observed after the first cycle of therapy.
Pharmacokinetics
- The pharmacokinetics of topotecan have been evaluated in cancer patients following doses of 0.5 to 1.5 mg/m2 administered as a 30-minute infusion. Topotecan exhibits multiexponential pharmacokinetics with a terminal half-life of 2 to 3 hours. Total exposure (AUC) is approximately dose-proportional.
- Distribution
- Binding of topotecan to plasma proteins is about 35%.
- Metabolism
- Topotecan undergoes a reversible pH dependent hydrolysis of its lactone moiety; it is the lactone form that is pharmacologically active. At pH ≤ 4, the lactone is exclusively present, whereas the ring-opened hydroxy-acid form predominates at physiologic pH. In vitro studies in human liver microsomes indicate topotecan is metabolized to an N-demethylated metabolite. The mean metabolite:parent AUC ratio was about 3% for total topotecan and topotecan lactone following IV administration.
- Excretion
- Renal clearance is an important determinant of topotecan elimination.
- In a mass balance/excretion study in 4 patients with solid tumors, the overall recovery of total topotecan and its N-desmethyl metabolite in urine and feces over 9 days averaged 73.4 ± 2.3% of the administered IV dose. Mean values of 50.8 ± 2.9% as total topotecan and 3.1 ± 1.0% as N-desmethyl topotecan were excreted in the urine following IV administration. Fecal elimination of total topotecan accounted for 17.9 ± 3.6% while fecal elimination of N-desmethyl topotecan was 1.7 ± 0.6%. An O-glucuronidation metabolite of topotecan and N-desmethyl topotecan has been identified in the urine. These metabolites, topotecan-O-glucuronide and N-desmethyl topotecan-O-glucuronide, were less than 2% of the administered dose.
- Effect of Gender
- The overall mean topotecan plasma clearance in male patients was approximately 24% higher than that in female patients, largely reflecting difference in body size.
- Effect of Age
- Topotecan pharmacokinetics have not been specifically studied in an elderly population, but population pharmacokinetic analysis in female patients did not identify age as a significant factor. Decreased renal clearance, which is common in the elderly, is a more important determinant of topotecan clearance.
- Effect of Race
- The effect of race on topotecan pharmacokinetics has not been studied.
- Effect of Renal Impairment
- In patients with mild renal impairment (creatinine clearance of 40 to 60 mL/min.), topotecan plasma clearance was decreased to about 67% of the value in patients with normal renal function. In patients with moderate renal impairment (Clcr of 20 to 39 mL/min.), topotecan plasma clearance was reduced to about 34% of the value in control patients, with an increase in half-life. Mean half-life, estimated in 3 renally impaired patients, was about 5.0 hours. Dosage adjustment is recommended for these patients.
- Effect of Hepatic Impairment
- Plasma clearance in patients with hepatic impairment (serum bilirubin levels between 1.7 and 15.0 mg/dL) was decreased to about 67% of the value in patients without hepatic impairment. Topotecan half-life increased slightly, from 2.0 hours to 2.5 hours, but these hepatically impaired patients tolerated the usual recommended topotecan dosage regimen.
- Drug Interactions
- Pharmacokinetic studies of the interaction of topotecan with concomitantly administered medications have not been formally investigated.
- In vitro inhibition studies using marker substrates known to be metabolized by human P450 CYP1A2, CYP2A6, CYP2C8/9, CYP2C19, CYP2D6, CYP2E, CYP3A, or CYP4A or dihydropyrimidine dehydrogenase indicate that the activities of these enzymes were not altered by topotecan. Enzyme inhibition by topotecan has not been evaluated in vivo.
- Cisplatin
- No pharmacokinetic data are available following topotecan (0.75 mg/m2/day for 3 consecutive days) and cisplatin (50 mg/m2/day on day 1) in patients with cervical cancer.
- Myelosuppression was more severe when topotecan was given in combination with cisplatin.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Carcinogenicity testing of topotecan has not been performed. Topotecan is known to be genotoxic to mammalian cells and is a probable carcinogen. Topotecan was mutagenic to L5178Y mouse lymphoma cells and clastogenic to cultured human lymphocytes with and without metabolic activation. It was also clastogenic to mouse bone marrow. Topotecan did not cause mutations in bacterial cells.
- Topotecan given to female rats prior to mating at a dose of 1.4 mg/m2 IV (about equal to the clinical dose of 1.5 mg/m2) caused superovulation possibly related to inhibition of follicular atresia. This dose given to pregnant female rats also caused increased pre-implantation loss. Studies in dogs given 0.4 mg/m2 IV (about 1/4th the clinical dose of 1.5 mg/m2) of topotecan daily for a month suggest that treatment may cause an increase in the incidence of multinucleated spermatogonial giant cells in the testes. Topotecan may impair fertility in women and men.
Clinical Studies
Small Cell Lung Cancer
- Topotecan was studied in 426 patients with recurrent or progressive small cell lung cancer in 1 randomized, comparative study and in 3 single-arm studies.
Randomized Comparative Study
- In a randomized, comparative, Phase 3 trial, 107 patients were treated with topotecan (1.5 mg/m2/day x 5 days starting on day 1 of a 21-day course) and 104 patients were treated with CAV (1,000 mg/m2 cyclophosphamide, 45 mg/m2 doxorubicin, 2 mg vincristine administered sequentially on day 1 of a 21-day course). All patients were considered sensitive to first-line chemotherapy (responders who then subsequently progressed ≥60 days after completion of first-line therapy). A total of 77% of patients treated with topotecan and 79% of patients treated with CAV received platinum/etoposide with or without other agents as first-line chemotherapy.
- Response rates, response duration, time to progression, and survival are shown in Table 6.
- Table 6. Efficacy of Topotecan Versus CAV (cyclophosphamide-doxorubicin-vincristine) in Small Cell Lung Cancer Patients Sensitive to First-Line Chemotherapy

This image is provided by the National Library of Medicine. - a Defined as improvement sustained over at least 2 courses compared to baseline.
- b Number of patients with baseline and at least 1 post-baseline assessment.
Single-Arm Studies
- Topotecan was also studied in 3 open-label, non-comparative trials in a total of 319 patients with recurrent or progressive small cell lung cancer after treatment with first-line chemotherapy. In all 3 studies, patients were stratified as either sensitive (responders who then subsequently progressed ≥90 days after completion of first-line therapy) or refractory (no response to first-line chemotherapy or who responded to first-line therapy and then progressed within 90 days of completing first-line therapy). Response rates ranged from 11% to 31% for sensitive patients and 2% to 7% for refractory patients. Median time to progression and median survival were similar in all 3 studies and the comparative study.
Cervical Cancer
- In a comparative trial, 147 eligible women were randomized to Topotecan Injection (0.75 mg/m2/day IV over 30 minutes × 3 consecutive days starting on day 1 of a 21-day course) plus cisplatin (50 mg/m2 on day 1) and 146 eligible women were randomized to cisplatin (50 mg/m2 IV on day 1 of a 21-day course). All patients had histologically confirmed Stage IV-B, recurrent, or persistent carcinoma of the cervix considered not amenable to curative treatment with surgery and/or radiation. Fifty-six percent (56%) of patients treated with Topotecan Injection plus cisplatin and 56% of patients treated with cisplatin had received prior cisplatin with or without other agents as first-line chemotherapy.
- Median survival of eligible patients receiving Topotecan Injection plus cisplatin was 9.4 months (95% CI: 7.9 to 11.9) compared to 6.5 months (95% CI: 5.8 to 8.8) among patients randomized to cisplatin alone with a log rank P-value of 0.033 (significance level was 0.044 after adjusting for the interim analysis). The unadjusted hazard ratio for overall survival was 0.76 (95% CI: 0.59 to 0.98).
- Figure 1. Overall Survival Curves Comparing Topotecan Injection plus Cisplatin versus Cisplatin Monotherapy in Cervical Cancer Patients
How Supplied
- Topotecan Injection is supplied in the following:
- 1 mg/mL Multiple Dose Vial
- NDC 66758-051-01 (package of 1)
- 3 mg/3 mL (1 mg/mL) Multiple Dose Vial
- NDC 66758-051-03 (package of 1)
- 4 mg/4 mL (1 mg/mL) Multiple Dose Vial
- NDC 66758-051-05 (package of 1)
Storage
- Unopened vials of Topotecan Injection are stable until the date indicated on the package when stored between 2ºC and 8°C (36°F and 46°F) and protected from light in the original package.
- Topotecan Injection is supplied as a multiple dose vial. Studies have shown the product is stable for 28 days after initial puncture when stored under refrigerated conditions.
- Topotecan Injection diluted for infusion is stable for 4 hours at room temperature or 24 hours at refrigerated temperature in ambient lighting conditions.
Images
Drug Images
{{#ask: Page Name::Topotecan (injection) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel

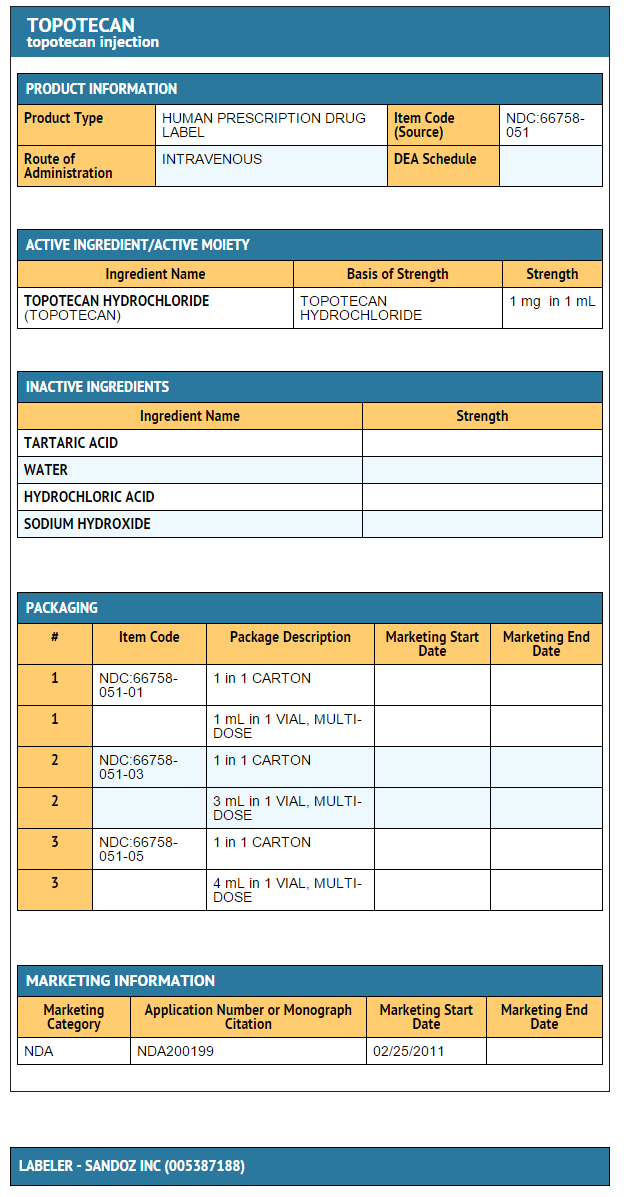
{{#ask: Label Page::Topotecan (injection) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Bone Marrow Suppression
- Inform patients that Topotecan Injection decreases blood cell counts such as white blood cells, platelets, and red blood cells. Patients who develop fever, other signs of infection (e.g., chills, cough, or burning pain on urination), or bleeding while on therapy should notify their physician promptly. Inform patients that frequent blood tests will be performed while taking Topotecan Injection to monitor for the occurrence of bone marrow suppression.
Pregnancy and Breastfeeding
- Advise patients to use effective contraceptive measures to prevent pregnancy and to avoid breastfeeding during treatment with Topotecan Injection.
Asthenia and Fatigue
- Inform patients that Topotecan Injection may cause asthenia or fatigue. If these symptoms occur, caution should be observed when driving or operating machinery.
Precautions with Alcohol
- Alcohol-Topotecan (injection) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- TOPOTECAN ®[1]
Look-Alike Drug Names
There is limited information regarding Topotecan (injection) Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Topotecan (injection) |Label Name=Topo 04.jpg
}}
{{#subobject:
|Label Page=Topotecan (injection) |Label Name=Topo 05.png
}}