Superantigen
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Superantigens (SAgs) are secreted proteins (exotoxins) that exhibit highly potent lymphocyte-transforming (mitogenic) activity directed towards T lymphocytes [2,4,6]. Compared to a normal antigen-induced T-cell response where .001-.0001% of the body’s T-cells are activated, SAgs are capable of activating up to 20% of the body’s T-cells [23]. This causes a massive immune response that is not specific to any particular epitope on the SAg. Since one of the fundamental strengths of the adaptive immune system is its ability to target antigens with high specificity, SAgs produce an immune response that is effectively useless. Microbes (including viruses, mycoplasma, and bacteria [2]) produce SAgs as a defense mechanism to aid them in evading the immune system [4].
Types of Superantigens
The most well characterized superantigens are secreted by the bacteria Staphylococcus aureus, and Streptococcus pyogenes [1]. These bacteria produce more than 20 different SAgs [9, 15]. Five groups have been proposed for classifying these toxins based on the specific variable region of the β chain of the human T cell receptor (TCR) to which they bind. Group I, for example, contains Toxic Shock Syndrome Toxin 1 (TSST-1) [1, 2]. Other non-bacterial SAgs have been discovered and are discussed in the section on endogenous superantigens.
Most of the genes encoding SAgs are located in close proximity to each other on mobile elements of bacterial genomes such as plasmids or “pathogenicity islands” [15]. An operon known as the enterotoxin gene cluster was found to be common in most SAg-producing bacterial strains [15]. Table 1 illustrates the main groups of enterotoxins and nomenclature used to describe them.
| Toxins | Amino acids | Molecular weight | motifs |
|---|
Structure
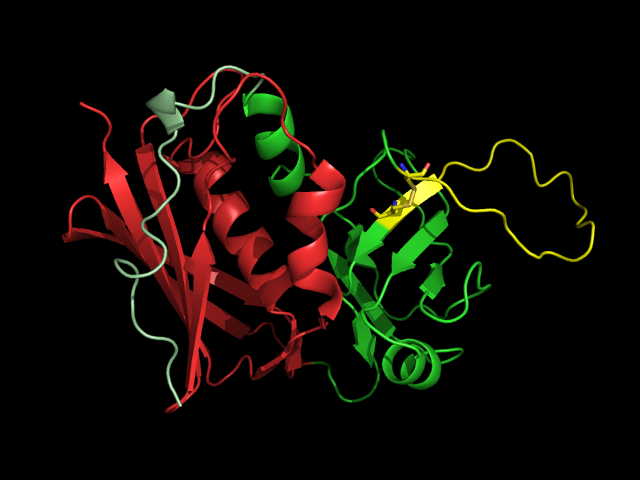
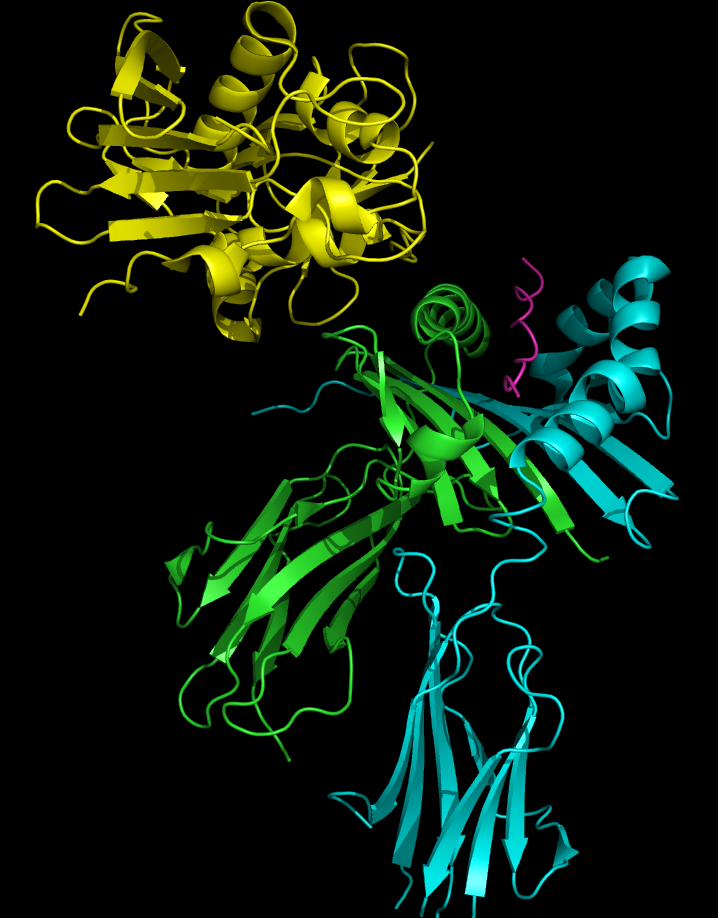
SAgs are produced intracellularly by bacteria and are released upon infection as extracellular mature toxins [5]. The sequences of these toxins are relatively conserved among the different subgroups. More important than sequence homology, the 3D structure is very similar among different SAgs resulting in similar functional effects among different groups [12, 14]. Crystal structures of the enterotoxins reveals that they are compact, ellipsoidal proteins sharing a characteristic two-domain folding pattern comprising an NH2-terminal β barrel globular domain known as the oligosaccharide / oligonucleotide fold, a long α-helix that diagonally spans the center of the molecule, and a COOH terminal globular domain [12]. The domains have binding regions for the Major Histocompatibility Complex Class II (MHC Class II) and the T-cell Receptor (TCR), respectively (see Figure 2) [13].
Binding
Superantigens bind first to the MHC Class II and then coordinate to a T-cell Receptor (TCR) with a specific Variable β motif [4,14,15].
MHC Class II
Binding to the MHC class II can involve interactions between the SAg and the peptide in the cleft of the MHC, as is the case with TSST-1 of the Group I SAgs. Group II SAgs exhibit peptide independent binding. Most SAgs bind preferentially to the α-chain of MHC class II molecules in the NH2-terminal domain [1, 15]. There are several different forms of the Human Leukocyte Antigen (HLA) that exhibit some variation on the heavy chains, and most SAgs show preference for the HLA-DQ form of the molecule [15]. Binding to the α-chain puts the SAg in the appropriate position to coordinate to the TCR. Less commonly, SAgs attach to the polymorphic MHC class II β-chain in an interaction mediated by a zinc ion coordination complex between three SAg residues and a highly conserved region of the HLA-DR β chain [14]. The use of a zinc ion in binding leads to a higher affinity interaction [12]. Several staphylococcal SAgs are capable of cross-linking MHC molecules by binding to both the α and β chains [12,14]. This mechanism stimulates cytokine expression and release in antigen presenting cells as well as inducing the production of costimulatory molecules that allow the cell to bind to and activate T cells more effectively [14].
T-cell Receptor
The T-cell binding region of the SAg interacts with the Variable region on the Beta chain of the T-cell Receptor. A given SAg can activate a large proportion of the T-cell population because the human T-cell repertoire comprises only about 50 types of Vβ elements and some SAgs are capable of binding to multiple types of VB regions. This interaction varies slightly among the different groups of SAgs [13]. Variability among different people in the types of T-cell regions that are prevalent explains why some people respond more strongly to certain SAgs. Group I SAgs contact the Vβ at the CDR2 and framework region of the molecule [1,9]. SAgs of Group II interact with the Vβ region using mechanisms that are conformation-dependent. These interactions are for the most part independent of specific Vβ amino acid side-chains. Group IV SAgs have been shown to engage all three CDR loops of certain Vβ forms [1,9]. The interaction takes place in a cleft between the small and large domains of the SAg and allows the SAg to act as a wedge between the TCR and MHC. This displaces the antigenic peptide away from the TCR and circumvents the normal mechanism for T-cell activation [14, 23].
The biological strength of the SAg (its ability to stimulate) is determined by its affinity for the TCR. SAgs with the highest affinity for the TCR elicit the strongest response [22]. SPMEZ-2 is the most potent SAg discovered to date [22].
T-cell Signaling
The SAg cross-links the MHC and the TCR inducing a signaling pathway that results in the proliferation of the cell and production of cytokines. Low levels of Zap-70 have been found in T-cells activated by SAgs, indicating that the normal signaling pathway of T-cell activation is impaired [10]. It is hypothesized that Fyn rather than Lck is activated by a tyrosine kinase, leading to the adaptive induction of anergy [18]. Both the protein kinase C pathway and the protein tyrosine kinase pathways are activated, resulting in upregulating production of proinflammatory cytokines [11]. This alternative signaling pathway impairs the calcium/calcineurin and Ras/MAPkinase pathways slightly [18], but allows for a focused inflammatory response.
Direct Effects
SAg stimulation of antigen presenting cells and T-cells elicits a response that is mainly inflammatory, focused on the action of Th1 T-helper cells. Some of the major products are IL-1, IL-2, IL-6, TNF-α, gamma interferon (IFN-γ), macrophage inflammatory protein 1α (MIP-1α), MIP-1β, and monocyte chemoattractant protein 1 (MCP-1) [11]. This excessive uncoordinated release of cytokines, (especially TNF-α), overloads the body and results in to rashes, fever, and can lead to multi-organ failure, coma and death [9, 15]. Deletion or anergy of activated T-cells follows infection. This results from production of IL-10 from prolonged exposure to the toxin. IL-10 downregulates production of IL-2, MHC Class II, and costimulatory molecules on the surface of APCs. These effects produce memory cells that are unresponsive to antigen stimulation [8, 19]. One mechanism by which this is possible involves cytokine-mediated suppression of T-cells. MHC crosslinking also activates a signaling pathway that suppresses hematopoiesis and upregulates Fas-mediated apoptosis [21]. IFN-α is another product of prolong SAg exposure. This cytokine is closely linked with induction of autoimmunity [20], and the autoimmune disease Kawasaki Disease is known to be caused by SAg infection [22]. SAg activation in T-cells leads to production of CD40 ligand which activates isotype switching in B cells from to IgG and IgM and IgE [17]. To summarize, the T-cells are stimulated and produce excess amounts of cytokine resulting in cytokine-mediated suppression of T-cells and deletion of the activated cells as the body returns to homeostasis. The toxic effects of the microbe and SAg also damage tissue and organ systems, a condition known as Toxic Shock Syndrome [17]. If the initial inflammation is survived, the host cells become anergic or are deleted, resulting in a severely compromised immune system.
Superantigenicity independent Effects (Indirect Effects)
Apart from their mitogenic activity, SAgs are able to cause symptoms that are characteristic of infection [as cited by 2].
One such effect is emesis. This effect is felt in cases of food poisoning, when SAg-producing bacteria release the toxin, which is highly resistant to heat. There is a distinct region of the molecule that is active in inducing gastrointestinal toxicity [as cited by 2]. This activity is also highly potent, and quantities as small as 20-35ug of SAg are able to induce vomiting [15].
SAgs are able to stimulate recruitment of neutrophils to the site of infection in a way that is independent of T-cell stimulation. This effect is due to the ability of SAgs to activate monocytic cells, stimulating the release of the cytokine TNF-α, leading to increased expression of adhesion molecules that recruit leukocytes to infected regions. This causes inflammation in the lungs, intestinal tissue, and any place that the bacteria have colonized [3]. While small amounts of inflammation are natural and helpful, excessive inflammation can lead to tissue destruction.
One of the more dangerous indirect effects of SAg infection concerns the ability of SAgs to augment the effects of endotoxins in the body. This is accomplished by reducing the threshold for endotoxicity. Schlievert demonstrated that, when administered conjunctively, the effects of SAg and endotoxin are magnified as much as 50 000 times [4]. This could be due to the reduced immune system efficiency induced by SAg infection. Aside from the synergistic relationship between endotoxin and SAg, the “double hit” effect of the activity of the endotoxin and the SAg result in effects more deleterious that those seen in a typical bacterial infection. This also implicates SAgs in the progression of sepsis in patients with bacterial infections [17].
Diseases Associated with Superantigen production [2]
- Toxic Shock Syndrome
- Kawasaki Disease
- Eczema
- Guttate psoriasis
- Rheumatoid arthritis
- Diabetes mellitus
- Scarlet fever [15]
Treatment
The primary goal of medical treatment is to eliminate the microbe that is producing the SAgs. This is accomplished through the use of vasopressors, fluid resuscitation and antibiotics [2]. The body naturally produces antibodies to some SAgs, and this effect can be augmented by stimulating B-cell production of these antibodies [16]. Immunoglobulin pools are able to neutralize specific antibodies and prevent T-cell activation. Synthetic antibodies and peptides have been created to mimic SAg-binding regions on the MHC class II, blocking the interaction and preventing T cell activation [as cited by 2]. Immunosuppressants are also employed to prevent T-cell activation and the release of cytokines. Corticosteroids are used to reduce inflammatory effects [17].
Evolution of Superantigen Production
SAg production effectively corrupts the immune response, allowing the microbe secreting the SAg to be carried and transmitted unchecked. One mechanism by which this is done system is through inducing anergy of the T-cells to antigens and SAgs [8, 10]. Lussow and MacDonald demonstrated this by systematically exposing animals to a streptococcal antigen. They found that exposure to other antigens after SAg infection failed to elicit an immune response [8]. In another experiment, Watson and Lee discovered that memory T-cells created by normal antigen stimulation were anergic to SAg stimulation and that memory T-cells created after a SAg infection were anergic to all antigen stimulation. The mechanism by which this occurred was undetermined [10]. The genes that regulate SAg expression also regulate mechanisms of immune evasion such as M protein and capsule expression, supporting the hypothesis that SAg production evolved primarily as a mechanism of immune evasion [6].
When the structure of individual SAg domains has been compared to other immunoglobulin-binding streptococcal proteins (such as those toxins produced by ''E. coli'') it was found that the domains separately resemble members of these families. This homology suggests that the SAgs evolved through the recombination of two smaller B-strand motifs [24].
Endogenous SAgs
Minor lymphocyte stimulating (Mls) exotoxins were originally discovered in the thymic stromal cells of mice. These toxins are encoded by SAg genes that were incorporated into the mouse genome from the mouse mammary tumour virus (MMTV). The presence of these genes in the mouse genome allows the mouse to express the antigen in the thymus as a means of negatively selecting for lymphocytes with a variable Beta region that is susceptible to stimulation by the viral SAg. The result is that these mice are immune to infection by the virus later in life [as cited by 2].
Similar endogenous SAg-dependent selection has yet to be identified in the human genome, but endogenous SAgs have been discovered and are suspected of playing an integral role in viral infection. Infection by theEpstein-Barr virus, for example, is known to cause production of a SAg in infected cells, yet no gene for the toxin has been found on the genome of the virus. The virus manipulates the infected cell to express its own SAg genes, and this helps it to evade the host immune system. Similar results have been found with rabies, cytomegalovirus, and HIV [as cited by 2].
Conclusion
Much detailed research has been conducted to determine the specific structures and binding mechanisms of each type of SAg. This paper presents a general overview of some of these that are more common and well-understood mechanisms of SAg infection. Recent research attempts to harness the immunostimulatory properties of these molecules by using specific SAgs to induce inflammatory cytokines that could target and prevent tumour growth [16]. As the microbial population develops more effective ways of infecting other organisms the human population uses intelligence to combat the advancing lines
References
- Jean-Nicholas P. Brouillard, Sebastian Günther, Ashok K. Varma, Irene Gryski, Christine A. Herfst, A.K.M. Nur-ur Rahman, Donald Y.M. Leung, Patrick M. Schlievert, Joaquín Madrenas, Eric J. Sundberg, and John K. McCormick. Crystal Structure of the Streptococcal Superantigen SpeI and Functional Role of a Novel Loop Domain in T Cell Activation by Group V Superantigens. Journal of Molecular Biology Volume 367;4 p 925-934
- Martin Llewelyn and Jon Cohen. 2002. Superantigens: microbial agents that corrupt immunity. THE LANCET Infectious Diseases. 2:156-162
- Diener K, Tessier P, Fraser J, Kontgen F, McColl SR. Induction of acute inflammation in vivo by staphylococcal superantigens I: leukocyte recruitment occurs independently of T lymphocytes and major histocompatibility complex class II molecules. Lab Invest 1998; 78: 647–56.
- Schlievert PM. 1982. Enhancement of host susceptibility to lethal endotoxin shock by staphylococcal pyrogenic exotoxin type C. Infect Immun; 36: 123–28.
- Shiranee Sriskandan, Lee Faulkner, and Philip Hopkins. 2007. Streptococcus pyogenes: Insight into the function of the streptococcal superantigens. The International Journal of Biochemistry & Cell Biology. 39 (1); 12-19
- Cleary PP, McLandsborough L, Ikeda L, Cue D, Krawczak J, Lam H. 1998 High-frequency intracellular infection and erythrogenic toxin A expression undergo phase variation in M1 group A streptococci. Mol Microbiol; 28: 157–67.
- O’Hehir RE, Lamb JR. 1990. Induction of specific clonal anergy in human T lymphocytes by Staphylococcus aureus enterotoxins. Proc Natl Acad Sci USA; 87: 8884–88.
- Lussow AR, MacDonald HR. 1994. Differential effects of superantigen-induced “anergy” on priming and effector stages of a T cell-dependent antibody response. Eur J Immunol; 24: 445–49.
- Rebecca A. Buonpane1, Beenu Moza, Eric J. Sundberg and David M. Kranz. 2005. Characterization of T Cell Receptors Engineered for High Affinity Against Toxic Shock Syndrome Toxin-1. J. Mol. Biol. 353; 308–321
- Andrew R.O. Watson a, William T. Lee. 2006. Defective T cell receptor-mediated signal transduction in memory CD4 T lymphocytes exposed to superantigen or anti-T cell receptor antibodies. Cellular Immunology 242: 80–90
- Bradley G. Stiles, Ph.D. and Teresa Krakauer, Ph.D. 2005 Staphylococcal Enterotoxins: a Purging Experience in Review, Part I. Clinical Microbiology Newsletter. 27;23
- K. Petersson, G. Forsberg & B. Walse. 2004. Interplay Between Superantigens and Immunoreceptors. Scandinavian Journal of Immunology 59; 345–355.
- Anastassios C. Papageorgiou, Howard S. Tranter and K. Ravi Acharya1. 1998. Crystal Structure of Microbial Superantigen Staphylococcal Enterotoxin B at 1.5 AÊ Resolution: Implications for Superantigen Recognition by MHC Class II Molecules and T-cell Receptors. J. Mol. Biol. 277; 61±79
- By Khalil Mehindate, Jacques Thibodeau, Mikael Dohlsten,Terje Kalland, Rafick-Pierre S4kaly, and Walid Mourad. 1995. Cross-linking of Major Histocompatibility Complex Class II Molecules by Staphylococcal Enterotoxin A Superantigen Is a Requirement for Inflammatory Cytokine Gene Expression. J. Exp. Med. 182; 1573-1577
- Joseph E. Alouf, Heide Muller-Alouf. 2003. Staphylococcal and streptococcal superantigens: molecular, biological, and clinical aspects. Int. J. Med. Microbiol. 292; 429 ± 440
- Eva Erlandsson, Kerstin Andersson, Anders Cavallin, Anneli Nilsson, Ulla Larsson-Lorek, Ulf Niss, Annelie Sjöberg, Marie Wallén-Öhman, Per Antonsson, Björn Walse and Göran Forsberg. 2003. Identification of the Antigenic Epitopes in Staphylococcal Enterotoxins A and E and Design of a Superantigen for Human Cancer Therapy. Journal of Molecular Biology. 333 (5); 893-905
- Haifa H. Jabara and Raif S. Geha. 1996. The superantigen toxic shock syndrome toxin-1 induces CD40 ligand expression and modulates IgE isotype switching. International Immunology. 8 (10); 1503-1510
- Seeyoung Choi and Ronald H. Schwartz. 2007. Molecular mechanisms for adaptive tolerance and other T cell anergy models. Seminars in Immunology; Article in Press, Corrected Proof
- Carla Miller, Jack A. Ragheb, and Ronald H. Schwartz.1999. Anergy and Cytokine-mediated Suppression as Distinct Superantigen-induced Tolerance Mechanisms In Vivo. J. Exp. Med. 190 (1); 53-64
- Yves Stauffer, Samuel Marguerat, Françoise Meylan, Catherine Ucla, Natalie Sutkowski, Brigitte Huber, Thierry Pelet and Bernard Conrad. 2001. Interferon-α-Induced Endogenous Superantigen A Model Linking Environment and Autoimmunity. Immunity. 15 (4);591-601
- Masaki Yamaguchi, Steve Nadler, Jong-Wook Lee, and H. Joachim Deeg. 1999 Induction of negative regulators of hematopoiesis in human bone marrow cells by HLA-DR cross-linking. Transplant Immunology. 7; 159-168.
- Vickery L. Arcus, Thomas Proft, Jill A. Sigrell, Heather M. Baker, John D. Fraser and Edward N. Baker. 2000. Conservation and variation in superantigen structure and activity highlighted by the three-dimensional structures of two new superantigens from Streptococcus pyogenes. Journal of Molecular Biology. 299 (1); 157-168
- Hongmin Li, Andrea Llera, Daisuke Tsuchiya, Lukas Leder, Xavier Ysern, Patrick M. Schlievert, Klaus Karjalainen, and Roy A. Mariuzza. 1998. Three-Dimensional Structure of the Complex between a T Cell Receptor b Chain and the Superantigen Staphylococcal Enterotoxin B. Immunity, 9; 807–816
- C Bachert, P Gevaert, P van Cauwenberge 2002. Staphylococcus aureus enterotoxins: a key in airway disease? Allergy 57 (6), 480–487.
Further reading
- Superantigen Web Database at Birkbeck College
- List of Superantigen Proteins from UniProt
- Superantigens at the US National Library of Medicine Medical Subject Headings (MeSH)