Sumatriptan (nasal)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Turky Alkathery, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Sumatriptan (nasal) is a serotonin (5-HT1B/1D) receptor agonist that is FDA approved for the treatment of migraine with or without aura in adults. Common adverse reactions include burning sensation, disorder/discomfort of nasal cavity/sinuses, throat discomfort, nausea and/or vomiting, bad/unusual taste, and dizziness/vertigo.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indication
- Sumatriptan Nasal Spray is indicated for the acute treatment of migraine with or without aura in adults.
- Limitations of Use:
- Use only if a clear diagnosis of migraine headache has been established. If a patient has no response to the first migraine attack treated with sumatriptan, reconsider the diagnosis of migraine before sumatriptan is administered to treat any subsequent attacks.
- Sumatriptan is not indicated for the prevention of migraine attacks.
- Safety and effectiveness of Sumatriptan Nasal Spray have not been established for cluster headache.
Dosage
- The recommended adult dose of Sumatriptan Nasal Spray for the acute treatment of migraine is 5 mg, 10 mg, or 20 mg. The 20-mg dose may provide a greater effect than the 5-mg and 10-mg doses, but may have a greater risk of adverse reactions.
- The 5-mg and 20-mg doses are given as a single spray in 1 nostril. The 10-mg dose may be achieved by the administration of a single 5-mg dose in each nostril.
- If the migraine has not resolved by 2 hours after taking Sumatriptan Nasal Spray, or returns after a transient improvement, 1 additional dose may be administered at least 2 hours after the first dose. The maximum daily dose is 40 mg in a 24-hour period.
- The safety of treating an average of more than 4 headaches in a 30‑day period has not been established.
Dosage Forms and Strengths
- Unit dose nasal spray devices containing 5 mg or 20 mg sumatriptan.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- Cluster headache.[1]
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Sumatriptan (nasal) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Safety and effectiveness in pediatric patients have not been established. Sumatriptan Nasal Spray is not recommended for use in patients younger than 18 years of age.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Sumatriptan (nasal) in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Sumatriptan (nasal) in pediatric patients.
Contraindications
- Sumatriptan Nasal Spray is contraindicated in patients with:
- Ischemic coronary artery disease (CAD) (angina pectoris, history of myocardial infarction, or documented silent ischemia) or coronary artery vasospasm, including Prinzmetal’s angina.
- Wolff-Parkinson-White syndrome or arrhythmias associated with other cardiac accessory conduction pathway disorders.
- History of stroke, transient ischemic attack (TIA), or history of hemiplegic or basilar migraine because these patients are at a higher risk of stroke
- Peripheral vascular disease
- Ischemic bowel disease
- Uncontrolled hypertension
- Recent use (i.e., within 24 hours) of ergotamine-containing medication, ergot-type medication (such as dihydroergotamine or methysergide), or another 5-hydroxytryptamine 1 (5-HT 1) agonist
- Concurrent administration of a monoamine oxidase (MAO)-A inhibitor or recent (within 2 weeks) use of an MAO-A inhibitor
- Hypersensitivity to sumatriptan (angioedema and anaphylaxis seen)
- Severe hepatic impairment
Warnings
Myocardial Ischemia, Myocardial Infarction, and Prinzmetal’s Angina
- The use of Sumatriptan Nasal Spray is contraindicated in patients with ischemic or vasospastic CAD. There have been rare reports of serious cardiac adverse reactions, including acute myocardial infarction, occurring within a few hours following administration of Sumatriptan Nasal Spray. Some of these reactions occurred in patients without known CAD. Sumatriptan Nasal Spray may cause coronary artery vasospasm (Prinzmetal’s angina), even in patients without a history of CAD.
- Perform a cardiovascular evaluation in triptan-naive patients who have multiple cardiovascular risk factors (e.g., increased age, diabetes, hypertension, smoking, obesity, strong family history of CAD) prior to receiving Sumatriptan Nasal Spray. If there is evidence of CAD or coronary artery vasospasm, Sumatriptan Nasal Spray is contraindicated. For patients with multiple cardiovascular risk factors who have a negative cardiovascular evaluation, consider administering the first dose of Sumatriptan Nasal Spray in a medically supervised setting and performing an electrocardiogram (ECG) immediately following administration of Sumatriptan Nasal Spray. For such patients, consider periodic cardiovascular evaluation in intermittent long-term users of Sumatriptan Nasal Spray.
Arrhythmias
- Life-threatening disturbances of cardiac rhythm, including ventricular tachycardia and ventricular fibrillation leading to death, have been reported within a few hours following the administration of 5-HT1 agonists. Discontinue Sumatriptan Nasal Spray if these disturbances occur. Sumatriptan Nasal Spray is contraindicated in patients with Wolff-Parkinson-White syndrome or arrhythmias associated with other cardiac accessory conduction pathway disorders.
Chest, Throat, Neck, and/or Jaw Pain/Tightness/Pressure
- Sensations of tightness, pain, pressure, and heaviness in the precordium, throat, neck, and jaw may occur after treatment with Sumatriptan Nasal Spray and are usually non-cardiac in origin. However, perform a cardiac evaluation if these patients are at high cardiac risk. The use of Sumatriptan Nasal Spray is contraindicated in patients with CAD and those with Prinzmetal’s variant angina.
Cerebrovascular Events
- Cerebral hemorrhage, subarachnoid hemorrhage, and stroke have occurred in patients treated with 5-HT1 agonists, and some have resulted in fatalities. In a number of cases, it appears possible that the cerebrovascular events were primary, the 5-HT1 agonist having been administered in the incorrect belief that the symptoms experienced were a consequence of migraine when they were not. Also, patients with migraine may be at increased risk of certain cerebrovascular events (e.g., stroke, hemorrhage, TIA). Discontinue Sumatriptan Nasal Spray if a cerebrovascular event occurs.
- Before treating headaches in patients not previously diagnosed as migraineurs, and in migraineurs who present with atypical symptoms, exclude other potentially serious neurological conditions. Sumatriptan Nasal Spray is contraindicated in patients with a history of stroke or TIA.
Other Vasospasm Reactions
- Sumatriptan Nasal Spray may cause non-coronary vasospastic reactions, such as peripheral vascular ischemia, gastrointestinal vascular ischemia and infarction (presenting with abdominal pain and bloody diarrhea), splenic infarction, and Raynaud’s syndrome. In patients who experience symptoms or signs suggestive of non-coronary vasospasm reaction following the use of any 5-HT1 agonist, rule out a vasospastic reaction before using additional Sumatriptan Nasal Spray.
- Reports of transient and permanent blindness and significant partial vision loss have been reported with the use of 5‑HT1 agonists. Since visual disorders may be part of a migraine attack, a causal relationship between these events and the use of 5-HT1 agonists have not been clearly established.
Medication Overuse Headache
- Overuse of acute migraine drugs (e.g., ergotamine, triptans, opioids, or combination of these drugs for 10 or more days per month) may lead to exacerbation of headache (medication overuse headache). Medication overuse headache may present as migraine-like daily headaches or as a marked increase in frequency of migraine attacks. Detoxification of patients, including withdrawal of the overused drugs, and treatment of withdrawal symptoms (which often includes a transient worsening of headache) may be necessary.
Serotonin Syndrome
- Serotonin syndrome may occur with Sumatriptan Nasal Spray, particularly during co-administration with selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), and MAO inhibitors. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). The onset of symptoms usually occurs within minutes to hours of receiving a new or a greater dose of a serotonergic medication. Discontinue Sumatriptan Nasal Spray if serotonin syndrome is suspected.
Increase in Blood Pressure
- Significant elevation in blood pressure, including hypertensive crisis with acute impairment of organ systems, has been reported on rare occasions in patients treated with 5-HT1 agonists, including patients without a history of hypertension. Monitor blood pressure in patients treated with sumatriptan. Sumatriptan Nasal Spray is contraindicated in patients with uncontrolled hypertension.
Local Irritation
- Local irritative symptoms such as burning, numbness, paresthesia, discharge, and pain or soreness were reported in approximately 5% of patients in controlled clinical trials and were noted to be severe in about 1%. The symptoms were transient and generally resolved in less than 2 hours. Limited examinations of the nose and throat did not reveal any clinically noticeable injury in these patients. The consequences of extended and repeated use of Sumatriptan Nasal Spray on the nasal and/or respiratory mucosa have not been systematically evaluated in patients.
Anaphylactic/Anaphylactoid Reactions
- Anaphylactic/anaphylactoid reactions have occurred in patients receiving sumatriptan. Such reactions can be life threatening or fatal. In general, anaphylactic reactions to drugs are more likely to occur in individuals with a history of sensitivity to multiple allergens. Sumatriptan Nasal Spray is contraindicated in patients with a history of hypersensitivity reaction to sumatriptan.
Seizures
- Seizures have been reported following administration of sumatriptan. Some have occurred in patients with either a history of seizures or concurrent conditions predisposing to seizures. There are also reports in patients where no such predisposing factors are apparent. Sumatriptan Nasal Spray should be used with caution in patients with a history of epilepsy or conditions associated with a lowered seizure threshold.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- Table 1 lists adverse reactions that occurred in worldwide placebo-controlled clinical trials in 3,419 patients with migraine. Only treatment-emergent adverse reactions that occurred at a frequency of 1% or more in the group treated with Sumatriptan Nasal Spray 20 mg and that occurred at a frequency greater than the placebo group are included in Table 1.

- The incidence of adverse reactions in controlled clinical trials was not affected by gender, weight, or age of the patients; use of prophylactic medications; or presence of aura. There were insufficient data to assess the impact of race on the incidence of adverse reactions.
Postmarketing Experience
- The following adverse reactions have been identified during postapproval use of Sumatriptan Tablets, Sumatriptan Nasal Spray, and Sumatriptan Injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reactions have been chosen for inclusion due to either their seriousness, frequency of reporting, or causal connection to Sumatriptan or a combination of these factors.
- Cardiovascular: Hypotension, palpitations.
- Neurological: Dystonia, tremor.
Drug Interactions
Ergot-Containing Drugs
- Ergot-containing drugs have been reported to cause prolonged vasospastic reactions. Because these effects may be additive, use of ergotamine-containing or ergot-type medications (like dihydroergotamine or methysergide) and Sumatriptan Nasal Spray within 24 hours of each other is contraindicated.
Monoamine Oxidase-A Inhibitors
- MAO-A inhibitors increase systemic exposure by up to 7-fold. Therefore, the use of Sumatriptan Nasal Spray in patients receiving MAO-A inhibitors is contraindicated.
Other 5-HT1 Agonists
- Because their vasospastic effects may be additive, co-administration of Sumatriptan Nasal Spray and other 5-HT1 agonists (e.g., triptans) within 24 hours of each other is contraindicated.
Selective Serotonin Reuptake Inhibitors/Serotonin Norepinephrine Reuptake Inhibitors and Serotonin Syndrome
- Cases of serotonin syndrome have been reported during co-administration of triptans and SSRIs, SNRIs, TCAs, and MAO inhibitors.
Use in Specific Populations
Pregnancy
- There are no adequate and well-controlled trials in pregnant women. In developmental toxicity studies in rats and rabbits, oral administration of sumatriptan to pregnant animals was associated with embryolethality, fetal abnormalities, and pup mortality. When administered by the intravenous route to pregnant rabbits, sumatriptan was embryolethal. Developmental toxicity studies of sumatriptan by the intranasal route have not been conducted. Sumatriptan Nasal Spray should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Oral administration of sumatriptan to pregnant rats during the period of organogenesis resulted in an increased incidence of fetal blood vessel (cervicothoracic and umbilical) abnormalities. The highest no-effect dose for embryofetal developmental toxicity in rats was 60 mg/kg/day. Oral administration of sumatriptan to pregnant rabbits during the period of organogenesis resulted in increased incidences of embryolethality and fetal cervicothoracic vascular and skeletal abnormalities. Intravenous administration of sumatriptan to pregnant rabbits during the period of organogenesis resulted in an increased incidence of embryolethality. The highest oral and intravenous no-effect doses for developmental toxicity in rabbits were 15 and 0.75 mg/kg/day, respectively.
- Oral administration of sumatriptan to rats prior to and throughout gestation resulted in embryofetal toxicity (decreased body weight, decreased ossification, increased incidence of skeletal abnormalities). The highest no-effect dose was 50 mg/kg/day. In offspring of pregnant rats treated orally with sumatriptan during organogenesis, there was a decrease in pup survival. The highest no-effect dose for this effect was 60 mg/kg/day. Oral treatment of pregnant rats with sumatriptan during the latter part of gestation and throughout lactation resulted in a decrease in pup survival. The highest no-effect dose for this finding was 100 mg/kg/day.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Sumatriptan (nasal) in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Sumatriptan (nasal) during labor and delivery.
Nursing Mothers
- Sumatriptan is excreted in human milk following subcutaneous administration. Infant exposure to sumatriptan can be minimized by avoiding breastfeeding for 12 hours after treatment with Sumatriptan Nasal Spray.
Pediatric Use
- Safety and effectiveness in pediatric patients have not been established. Sumatriptan Nasal Spray is not recommended for use in patients younger than 18 years of age.
- Two controlled clinical trials evaluated Sumatriptan Nasal Spray (5 to 20 mg) in 1,248 adolescent migraineurs aged 12 to 17 years who treated a single attack. The trials did not establish the efficacy of Sumatriptan Nasal Spray compared with placebo in the treatment of migraine in adolescents. Adverse reactions observed in these clinical trials were similar in nature to those reported in clinical trials in adults.
- Five controlled clinical trials (2 single-attack trials, 3 multiple-attack trials) evaluating oral Sumatriptan (25 to 100 mg) in pediatric patients aged 12 to 17 years enrolled a total of 701 adolescent migraineurs. These trials did not establish the efficacy of oral Sumatriptan compared with placebo in the treatment of migraine in adolescents. Adverse reactions observed in these clinical trials were similar in nature to those reported in clinical trials in adults. The frequency of all adverse reactions in these patients appeared to be both dose- and age-dependent, with younger patients reporting reactions more commonly than older adolescents.
- Postmarketing experience documents that serious adverse reactions have occurred in the pediatric population after use of subcutaneous, oral, and/or intranasal Sumatriptan. These reports include reactions similar in nature to those reported rarely in adults, including stroke, visual loss, and death. A myocardial infarction has been reported in a 14‑year‑old male following the use of oral Sumatriptan; clinical signs occurred within 1 day of drug administration. Clinical data to determine the frequency of serious adverse reactions in pediatric patients who might receive subcutaneous, oral, or intranasal Sumatriptan are not presently available.
Geriatic Use
- Clinical trials of Sumatriptan Nasal Spray did not include sufficient numbers of patients aged 65 and older to determine whether they respond differently from younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
- A cardiovascular evaluation is recommended for geriatric patients who have other cardiovascular risk factors (e.g., diabetes, hypertension, smoking, obesity, strong family history of CAD) prior to receiving Sumatriptan Nasal Spray.
Gender
There is no FDA guidance on the use of Sumatriptan (nasal) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Sumatriptan (nasal) with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Sumatriptan (nasal) in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Sumatriptan (nasal) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Sumatriptan (nasal) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Sumatriptan (nasal) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Nasal.
Monitoring
- Monitor blood pressure in patients treated with sumatriptan.
IV Compatibility
- There is limited information regarding IV Compatibility.
Overdosage
- In clinical trials, the highest single doses of Sumatriptan Nasal Spray administered without significant reactions were 40 mg to 12 volunteers and 40 mg to 85 subjects with migraine, which is twice the highest single recommended dose. In addition, 12 volunteers were administered a total daily dose of 60 mg (20 mg 3 times daily) for 3.5 days without significant adverse reactions.
- Overdose in animals has been fatal and has been heralded by convulsions, tremor, paralysis, inactivity, ptosis, erythema of the extremities, abnormal respiration, cyanosis, ataxia, mydriasis, salivation, and lacrimation.
- The elimination half‑life of sumatriptan is approximately 2 hours, and therefore monitoring of patients after overdose with Sumatriptan Nasal Spray should continue for at least 10 hours or while symptoms or signs persist.
- It is unknown what effect hemodialysis or peritoneal dialysis has on the serum concentrations of sumatriptan.
Pharmacology
Mechanism of Action
- Sumatriptan binds with high affinity to human cloned 5-HT1B/1D receptors. Sumatriptan presumably exerts its therapeutic effects in the treatment of migraine headache through agonist effects at the 5‑HT1B/1D receptors on intracranial blood vessels and sensory nerves of the trigeminal system, which result in cranial vessel constriction and inhibition of pro-inflammatory neuropeptide release.
Structure
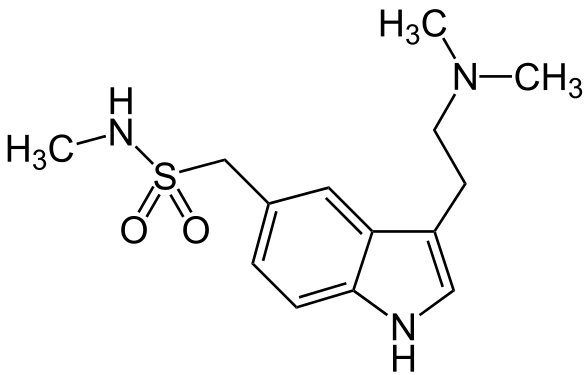
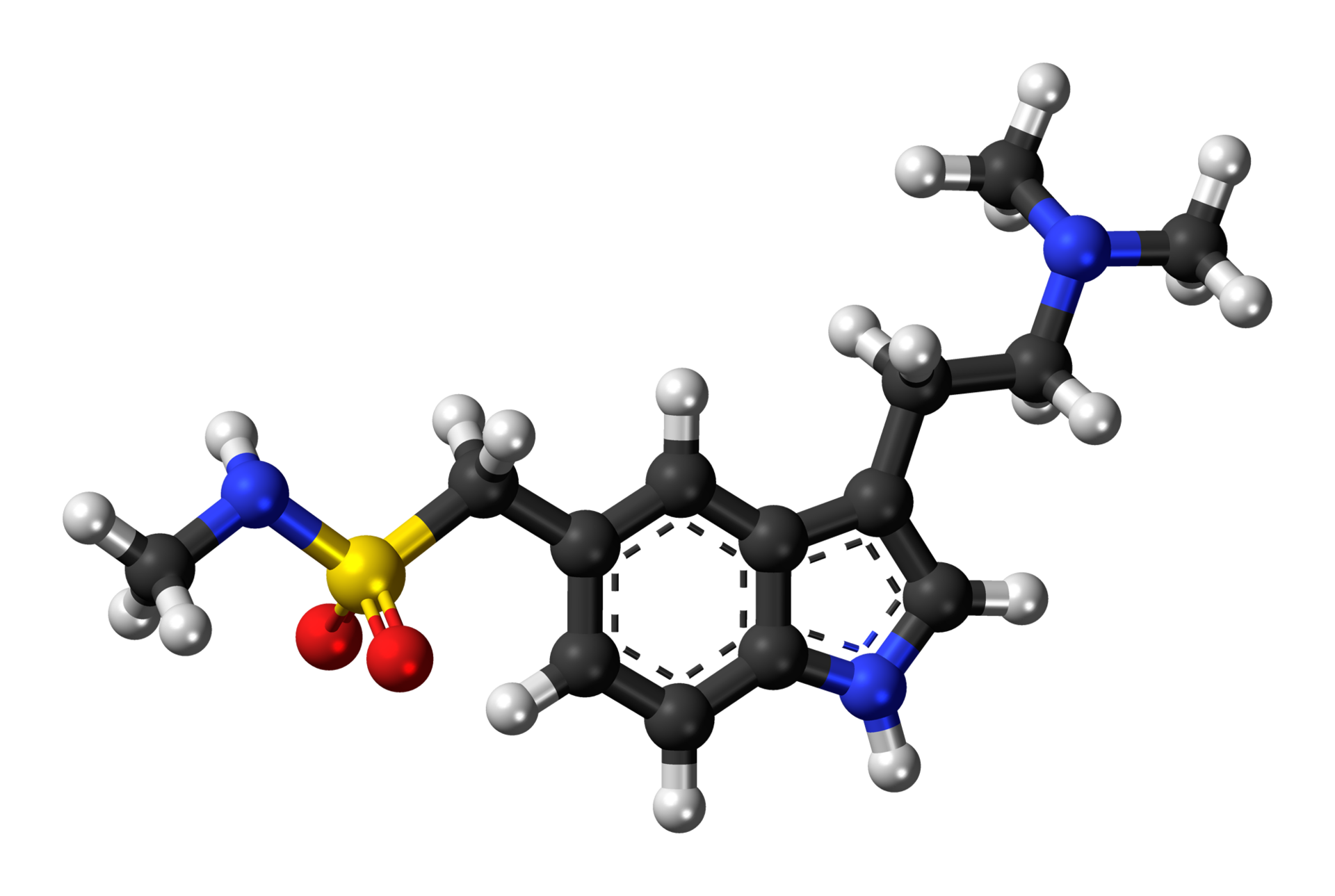
- Sumatriptan Nasal Spray contains sumatriptan, a selective 5-HT1B/1D receptor agonist. Sumatriptan is chemically designated as 3-[2-(dimethylamino)ethyl]-N-methyl-indole-5-methanesulfonamide, and it has the following structure:
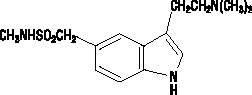
- The empirical formula is C14H21N3O2S, representing a molecular weight of 295.4. Sumatriptan is a white to off-white powder that is readily soluble in water and in saline.
- Each Sumatriptan Nasal Spray contains 5 or 20 mg of sumatriptan in a 100-μL unit dose aqueous buffered solution containing monobasic potassium phosphate NF, anhydrous dibasic sodium phosphate USP, sulfuric acid NF, sodium hydroxide NF, and purified water USP. The pH of the solution is approximately 5.5. The osmolality of the solution is 372 or 742 mOsmol for the 5- and 20-mg Sumatriptan Nasal Spray, respectively.
Pharmacodynamics
- Blood Pressure: Significant elevation in blood pressure, including hypertensive crisis, has been reported in patients with and without a history of hypertension.
- Peripheral (Small) Arteries: In healthy volunteers (N = 18), a trial evaluating the effects of sumatriptan on peripheral (small vessel) arterial reactivity failed to detect a clinically significant increase in peripheral resistance.
- Heart Rate: Transient increases in blood pressure observed in some patients in clinical trials carried out during sumatriptan’s development as a treatment for migraine were not accompanied by any clinically significant changes in heart rate.
Pharmacokinetics
- Absorption and Bioavailability: In a trial of 20 female volunteers, the mean maximum concentration following a 5- and 20-mg intranasal dose was 5 and 16 ng/mL, respectively. The mean Cmax following a 6‑mg subcutaneous injection is 71 ng/mL (range: 49 to 110 ng/mL). The mean Cmax is 18 ng/mL (range: 7 to 47 ng/mL) following oral dosing with 25 mg and 51 ng/mL (range: 28 to 100 ng/mL) following oral dosing with 100 mg of sumatriptan. In a trial of 24 male volunteers, the bioavailability relative to subcutaneous injection was low, approximately 17%, primarily due to presystemic metabolism and partly due to incomplete absorption.
- Clinical and pharmacokinetic data indicate that administration of two 5-mg doses, 1 dose in each nostril, is equivalent to administration of a single 10-mg dose in 1 nostril.
- Distribution: Protein binding, determined by equilibrium dialysis over the concentration range of 10 to 1,000 ng/mL, is low, approximately 14% to 21%. The effect of sumatriptan on the protein binding of other drugs has not been evaluated. The apparent volume of distribution is 2.7 L/kg.
- Metabolism:In vitro studies with human microsomes suggest that sumatriptan is metabolized by MAO, predominantly the A isoenzyme. Most of a radiolabeled dose of sumatriptan excreted in the urine is the major metabolite indole acetic acid (IAA) or the IAA glucuronide, both of which are inactive.
- Elimination: The elimination half-life of sumatriptan administered as a nasal spray is approximately 2 hours, similar to the half-life seen after subcutaneous injection. Only 3% of the dose is excreted in the urine as unchanged sumatriptan; 42% of the dose is excreted as the major metabolite, the indole acetic acid analogue of sumatriptan. The total plasma clearance is approximately 1,200 mL/min.
- Special Populations: Age: The pharmacokinetics of sumatriptan in the elderly (mean age: 72 years, 2 males and 4 females) and in subjects with migraine (mean age: 38 years, 25 males and 155 females) were similar to that in healthy male subjects (mean age: 30 years). Intranasal sumatriptan has not been evaluated for age differences.
- Renal Impairment: The effect of renal impairment on the pharmacokinetics of sumatriptan has not been examined.
- Hepatic Impairment: The effect of mild to moderate hepatic disease on the pharmacokinetics of the intranasal formulation of sumatriptan has not been evaluated. Sumatriptan bioavailability following intranasal administration is 17%, similar to that after oral administration (15%). Following oral administration, an approximately 70% increase in Cmax and AUC was observed in one small trial of patients with moderate liver impairment (n = 8) matched for sex, age and weight with healthy subjects (n = 8). Similar changes can be expected following intranasal administration.
- The pharmacokinetics of sumatriptan in patients with severe hepatic impairment has not been studied. The use of Sumatriptan Nasal Spray in patients with severe hepatic impairment is contraindicated.
- Race: The systemic clearance and Cmax of subcutaneous sumatriptan were similar in black (n = 34) and Caucasian (n = 38) healthy male subjects. Intranasal sumatriptan has not been evaluated for race differences.
- Drug Interaction Studies: Monoamine Oxidase-A Inhibitors:Treatment with MAO-A inhibitors generally leads to an increase of sumatriptan plasma levels. MAO inhibitors interaction studies have not been performed with intranasal sumatriptan.
- Due to gut and hepatic metabolic first-pass effects, the increase of systemic exposure after co-administration of an MAO-A inhibitor with oral sumatriptan is greater than after co-administration of the MAO inhibitors with subcutaneous sumatriptan. The effects of an MAO inhibitor on systemic exposure after intranasal sumatriptan would be expected to be greater than the effect after subcutaneous sumatriptan but smaller than the effect after oral sumatriptan because only swallowed drug would be subject to first-pass effects.
- In a trial of 14 healthy females, pretreatment with an MAO-A inhibitor decreased the clearance of subcutaneous sumatriptan, resulting in a 2-fold increase in the area under the sumatriptan plasma concentration-time curve (AUC), corresponding to a 40% increase in elimination half‑life.
- A small trial evaluating the effect of pretreatment with an MAO-A inhibitor on the bioavailability from a 25-mg oral sumatriptan tablet resulted in an approximately 7-fold increase in systemic exposure.
- Xylometazoline: An in vivo drug interaction trial indicated that 3 drops of xylometazoline (0.1% w/v), a decongestant, administered 15 minutes prior to a 20-mg nasal dose of sumatriptan did not alter the pharmacokinetics of sumatriptan.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Carcinogenesis: In carcinogenicity studies in mouse and rat in which sumatriptan was administered orally for 78 and 104 weeks, respectively, there was no evidence in either species of an increase in tumors related to sumatriptan administration.
- Carcinogenicity studies of sumatriptan using the nasal route have not been conducted.
- Mutagenesis: Sumatriptan was negative in in vitro(bacterial reverse mutation [Ames], gene cell mutation in Chinese hamster V79/HGPRT, chromosomal aberration in human lymphocytes) and in vivo (rat micronucleus) assays.
- Impairment of Fertility: When sumatriptan was administered by subcutaneous injection to male and female rats prior to and throughout the mating period, there was no evidence of impaired fertility at doses up to 60 mg/kg/day. When sumatriptan (5, 50, or 500 mg/kg/day) was administered orally to male and female rats prior to and throughout the mating period, there was a treatment-related decrease in fertility secondary to a decrease in mating in animals treated with doses greater than 5 mg/kg/day. It is not clear whether this finding was due to an effect on males or females or both.
- Fertility studies of sumatriptan using the intranasal route have not been conducted.
Animal Toxicology and/or Pharmacology
- Corneal Opacities: Dogs receiving oral sumatriptan developed corneal opacities and defects in the corneal epithelium. Corneal opacities were seen at the lowest dose tested, 2 mg/kg/day, and were present after 1 month of treatment. Defects in the corneal epithelium were noted in a 60‑week study. Earlier examinations for these toxicities were not conducted and no-effect doses were not established.
Clinical Studies
- The efficacy of Sumatriptan Nasal Spray in the acute treatment of migraine headaches was demonstrated in 8, randomized, double-blind, placebo-controlled trials, of which 5 used the recommended dosing regimen and used the marketed formulation. Patients enrolled in these 5 trials were predominately female (86%) and Caucasian (95%), with a mean age of 41 years (range of 18 to 65 years). Patients were instructed to treat a moderate to severe headache. Headache response, defined as a reduction in headache severity from moderate or severe pain to mild or no pain, was assessed up to 2 hours after dosing. Associated symptoms such as nausea, photophobia, and phonophobia were also assessed. Maintenance of response was assessed for up to 24 hours postdose. A second dose of Sumatriptan Nasal Spray or other medication was allowed 2 to 24 hours after the initial treatment for recurrent headache. The frequency and time to use of these additional treatments were also determined. In all trials, doses of 10 and 20 mg were compared with placebo in the treatment of 1 to 3 migraine attacks. Patients received doses as a single spray into 1 nostril. In 2 trials, a 5-mg dose was also evaluated.
- In all 5 trials utilizing the market formulation and recommended dosage regimen, the percentage of patients achieving headache response 2 hours after treatment was significantly greater among patients receiving Sumatriptan Nasal Spray at all doses (with one exception) compared with those who received placebo. In 4 of the 5 trials, there was a statistically significant greater percentage of patients with headache response at 2 hours in the 20-mg group when compared with the lower dose groups (5 and 10 mg). There were no statistically significant differences between the 5- and 10-mg dose groups in any trial. The results from the 5 controlled clinical trials are summarized in Table 2. Note that, in general, comparisons of results obtained in trials conducted under different conditions by different investigators with different samples of patients are ordinarily unreliable for purposes of quantitative comparison

- The estimated probability of achieving an initial headache response over the 2 hours following treatment is depicted in Figure 1.
Figure 1. Estimated Probability of Achieving Initial Headache Response Within 120 Minutes a
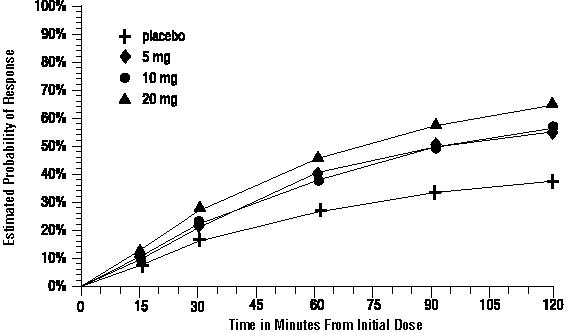
- The figure shows the probability over time of obtaining headache response (no or mild pain) following treatment with intranasal sumatriptan. The averages displayed are based on pooled data from the 5 clinical controlled trials providing evidence of efficacy. Kaplan-Meier plot with patients not achieving response within 120 minutes censored to 120 minutes.
- For patients with migraine-associated nausea, photophobia, and phonophobia at baseline, there was a lower incidence of these symptoms at 2 hours following administration of Sumatriptan Nasal Spray compared with placebo.
- Two to 24 hours following the initial dose of study treatment, patients were allowed to use additional treatment for pain relief in the form of a second dose of study treatment or other medication. The estimated probability of patients taking a second dose or other medication for migraine over the 24 hours following the initial dose of study treatment is summarized in Figure 2.
Figure 2. The Estimated Probability of Patients Taking a Second Dose or Other Medication for Migraine Over the 24 Hours Following the Initial Dose of Study Treatment

- Kaplan-Meier plot based on data obtained in the 3 clinical controlled trials providing evidence of efficacy with patients not using additional treatments censored to 24 hours. Plot also includes patients who had no response to the initial dose. No remedication was allowed within 2 hours postdose.
- There is evidence that doses above 20 mg do not provide a greater effect than 20 mg. There was no evidence to suggest that treatment with sumatriptan was associated with an increase in the severity of recurrent headaches. The efficacy of Sumatriptan Nasal Spray was unaffected by presence of aura; duration of headache prior to treatment; gender, age, or weight of the subject; or concomitant use of common migraine prophylactic drugs (e.g., beta-blockers, calcium channel blockers, tricyclic antidepressants). There were insufficient data to assess the impact of race on efficacy.
How Supplied
- IMITREX Nasal Spray 5 mg (NDC 0173-0524-00) and 20 mg (NDC 0173-0523-00) are each supplied in boxes of 6 nasal spray devices. Each unit dose spray supplies 5 mg and 20 mg, respectively, of sumatriptan.
Storage
- Store between 2°C and 30°C (36°F and 86°F). Protect from light.
Images
Drug Images
{{#ask: Page Name::Sumatriptan (nasal) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel



{{#ask: Label Page::Sumatriptan (nasal) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Risk of Myocardial Ischemia and/or Infarction, Prinzmetal’s Angina, Other Vasospasm-Related Events, Arrhythmias, and Cerebrovascular Events: Inform patients that IMITREX Nasal Spray may cause serious cardiovascular side effects such as myocardial infarction or stroke. Although serious cardiovascular events can occur without warning symptoms, patients should be alert for the signs and symptoms of chest pain, shortness of breath, irregular heartbeat, significant rise in blood pressure, weakness, and slurring of speech and should ask for medical advice if any indicative sign or symptoms are observed. Apprise patients of the importance of this follow-up.
- Anaphylactic/Anaphylactoid Reactions: Inform patients that anaphylactic/anaphylactoid reactions have occurred in patients receiving IMITREX Nasal Spray. Such reactions can be life threatening or fatal. In general, anaphylactic reactions to drugs are more likely to occur in individuals with a history of sensitivity to multiple allergens.
- Concomitant Use With Other Triptans or Ergot Medications: Inform patients that use of IMITREX Nasal Spray within 24 hours of another triptan or an ergot-type medication (including dihydroergotamine or methysergide) is contraindicated.
- Serotonin Syndrome: Caution patients about the risk of serotonin syndrome with the use of IMITREX Nasal Spray or other triptans, particularly during combined use with SSRIs, SNRIs, TCAs, and MAO inhibitors.
- Medication Overuse Headache: Inform patients that use of acute migraine drugs for 10 or more days per month may lead to an exacerbation of headache and encourage patients to record headache frequency and drug use (e.g., by keeping a headache diary).
- Pregnancy: Inform patients that IMITREX Nasal Spray should not be used during pregnancy unless the potential benefit justifies the potential risk to the fetus.
- Nursing Mothers: Advise patients to notify their healthcare provider if they are breastfeeding or plan to breastfeed.
- Ability to Perform Complex Tasks: Treatment with IMITREX Nasal Spray may cause somnolence and dizziness; instruct patients to evaluate their ability to perform complex tasks after administration of IMITREX Nasal Spray.
- Local Irritation: Inform patients that they may experience local irritation of their nose and throat. The symptoms will generally resolve in less than 2 hours.
- How to Use IMITREX Nasal Spray: Provide patients instruction on the proper use of IMITREX Nasal Spray. Caution patients to avoid spraying the contents of the device in their eyes.
Patient Information

Precautions with Alcohol
- Alcohol-Sumatriptan (nasal) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- IMITREX®[2]
Look-Alike Drug Names
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ van Vliet JA, Bahra A, Martin V, Ramadan N, Aurora SK, Mathew NT; et al. (2003). "Intranasal sumatriptan in cluster headache: randomized placebo-controlled double-blind study". Neurology. 60 (4): 630–3. PMID 12601104.
- ↑ "IMITREX- sumatriptan spray".
- ↑ 3.0 3.1 "https://www.ismp.org". External link in
|title=(help)