Single nucleotide polymorphism
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
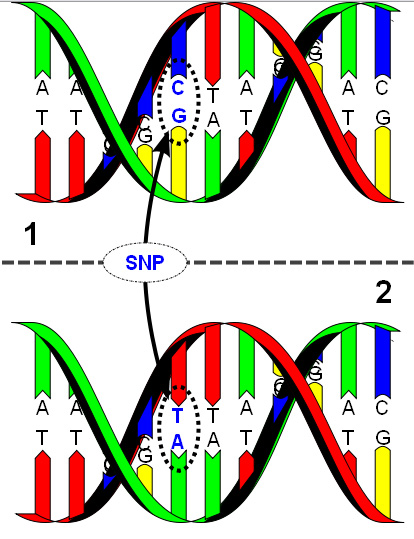
A single-nucleotide polymorphism (SNP) is a DNA sequence variation occurring when a single nucleotide — A, T, C or G — in the genome (or other shared sequence) differs between members of a biological species or paired chromosomes in a human. For example, two sequenced DNA fragments from different individuals, AAGCCTA to AAGCTTA, contain a difference in a single nucleotide. In this case we say that there are two alleles. Almost all common SNPs have only two alleles. The genomic distribution of SNPs is not homogenous; SNPs usually occur in non-coding regions more frequently than in coding regions or, in general, where natural selection is acting and fixating the allele of the SNP that constitutes the most favorable genetic adaptation.[1] Other factors, like genetic recombination and mutation rate, can also determine SNP density.[2]
SNP density can be predicted by the presence of microsatellites: AT microsatellites in particular are potent predictors of SNP density, with long (AT)(n) repeat tracts tending to be found in regions of significantly reduced SNP density and low GC content.[3]
Within a population, SNPs can be assigned a minor allele frequency — the lowest allele frequency at a locus that is observed in a particular population. This is simply the lesser of the two allele frequencies for single-nucleotide polymorphisms. There are variations between human populations, so a SNP allele that is common in one geographical or ethnic group may be much rarer in another.
These genetic variations between individuals (particularly in non-coding parts of the genome) are exploited in DNA fingerprinting, which is used in forensic science . Also, these genetic variations underlie differences in our susceptibility to disease. The severity of illness and the way our body responds to treatments are also manifestations of genetic variations. For example, a single base mutation in the APOE (apolipoprotein E) gene is associated with a higher risk for Alzheimer disease.[4]
Types
| Types of SNPs |
|---|
|
Single-nucleotide polymorphisms may fall within coding sequences of genes, non-coding regions of genes, or in the intergenic regions (regions between genes). SNPs within a coding sequence do not necessarily change the amino acid sequence of the protein that is produced, due to degeneracy of the genetic code.
SNPs in the coding region are of two types, synonymous and nonsynonymous SNPs. Synonymous SNPs do not affect the protein sequence while nonsynonymous SNPs change the amino acid sequence of protein. The nonsynonymous SNPs are of two types: missense and nonsense.
SNPs that are not in protein-coding regions may still affect gene splicing, transcription factor binding, messenger RNA degradation, or the sequence of non-coding RNA. Gene expression affected by this type of SNP is referred to as an eSNP (expression SNP) and may be upstream or downstream from the gene.
Use and importance
Variations in the DNA sequences of humans can affect how humans develop diseases and respond to pathogens, chemicals, drugs, vaccines, and other agents. SNPs are also critical for personalized medicine.[5] However, their greatest importance in biomedical research is for comparing regions of the genome between cohorts (such as with matched cohorts with and without a disease) in genome-wide association studies.
The study of SNPs is also important in crop and livestock breeding programs. See SNP genotyping for details on the various methods used to identify SNPs.
SNPs are usually biallelic and thus easily assayed.[6] A single SNP may cause a Mendelian disease. For complex diseases, SNPs do not usually function individually, rather, they work in coordination with other SNPs to manifest a disease condition as has been seen in osteoporosis.[7]
As of 26 June 2012[update], dbSNP listed 53,558,214 SNPs in humans.[8]
SNPs have been used in genome-wide association studies (GWAS), e.g. as high-resolution markers in gene mapping related to diseases or normal traits. The knowledge of SNPs will help in understanding pharmacokinetics (PK) or pharmacodynamics, i.e. how drugs act in individuals with different genetic variants. A wide range of human diseases, i.e Sickle–cell anemia, β Thalassemia and Cystic fibrosis result from SNPs.[9][10][11] Diseases with different SNPs may become relevant pharmacogenomic targets for drug therapy.[12] Some SNPs are associated with the metabolism of different drugs.[13][14][15] SNPs without an observable impact on the phenotype are still useful as genetic markers in genome-wide association studies, because of their quantity and the stable inheritance over generations.[16]
Examples
- rs6311 and rs6313 are SNPs in the HTR2A gene on human chromosome 13.
- A SNP in the F5 gene causes a hypercoagulability disorder with the variant Factor V Leiden.
- rs3091244 is an example of a triallelic SNP in the CRP gene on human chromosome 1.[17]
- TAS2R38 codes for PTC tasting ability, and contains 6 annotated SNPs.[18]
- rs148649884 and rs138055828 in the FCN1 gene encoding M-ficolin crippled the ligand-binding capability of the recombinant M-ficolin.[19]
Databases
As there are for genes, bioinformatics databases exist for SNPs. dbSNP is a SNP database from the National Center for Biotechnology Information (NCBI). SNPedia is a wiki-style database supporting personal genome annotation, interpretation and analysis. The OMIM database describes the association between polymorphisms and diseases (e.g., gives diseases in text form), the Human Gene Mutation Database provides gene mutations causing or associated with human inherited diseases and functional SNPs, and GWAS Central allows users to visually interrogate the actual summary-level association data in one or more genome-wide association studies. The International SNP Map working group mapped the sequence flanking each SNP by alignment to the genomic sequence of large-insert clones in Genebank. These Alignments were converted to chromosomal coordinates that is shown in the followign table.[20] Another database is the International HapMap Project, where researches are identifying Tag SNP to be able to determine the collection of haplotypes present in each subject.
| Chromosome | Length(bp) | All SNPs | TSC SNPs | ||
|---|---|---|---|---|---|
| SNPs | kb per SNP | SNPs | kb per SNP | ||
| 1 | 214,066,000 | 129,931 | 1.65 | 75,166 | 2.85 |
| 2 | 222,889,000 | 103,664 | 2.15 | 76,985 | 2.90 |
| 3 | 186,938,000 | 93,140 | 2.01 | 63,669 | 2.94 |
| 4 | 169,035,000 | 84,426 | 2.00 | 65,719 | 2.57 |
| 5 | 170,954,000 | 117,882 | 1.45 | 63,545 | 2.69 |
| 6 | 165,022,000 | 96,317 | 1.71 | 53,797 | 3.07 |
| 7 | 149,414,000 | 71,752 | 2.08 | 42,327 | 3.53 |
| 8 | 125,148,000 | 57,834 | 2.16 | 42,653 | 2.93 |
| 9 | 107,440,000 | 62,013 | 1.73 | 43,020 | 2.50 |
| 10 | 127,894,000 | 61,298 | 2.09 | 42,466 | 3.01 |
| 11 | 129,193,000 | 84,663 | 1.53 | 47,621 | 2.71 |
| 12 | 125,198,000 | 59,245 | 2.11 | 38,136 | 3.28 |
| 13 | 93,711,000 | 53,093 | 1.77 | 35,745 | 2.62 |
| 14 | 89,344,000 | 44,112 | 2.03 | 29,746 | 3.00 |
| 15 | 73,467,000 | 37,814 | 1.94 | 26,524 | 2.77 |
| 16 | 74,037,000 | 38,735 | 1.91 | 23,328 | 3.17 |
| 17 | 73,367,000 | 34,621 | 2.12 | 19,396 | 3.78 |
| 18 | 73,078,000 | 45,135 | 1.62 | 27,028 | 2.70 |
| 19 | 56,044,000 | 25,676 | 2.18 | 11,185 | 5.01 |
| 20 | 63,317,000 | 29,478 | 2.15 | 17,051 | 3.71 |
| 21 | 33,824,000 | 20,916 | 1.62 | 9,103 | 3.72 |
| 22 | 33,786,000 | 28,410 | 1.19 | 11,056 | 3.06 |
| X | 131,245,000 | 34,842 | 3.77 | 20,400 | 6.43 |
| Y | 21,753,000 | 4,193 | 5.19 | 1,784 | 12.19 |
| RefSeq | 15,696,674 | 14,534 | 1.08 | ||
| Totals | 2,710,164,000 | 1,419,190 | 1.91 | 887,450 | 3.05 |
Nomenclature
The nomenclature for SNPs can be confusing: several variations can exist for an individual SNP and consensus has not yet been achieved. One approach is to write SNPs with a prefix, period and "greater than" sign showing the wild-type and altered nucleotide or amino acid; for example, c.76A>T.[21][22][23] SNPs are frequently referred to by their dbSNP rs number, as in the examples above.
SNP analysis
Analytical methods to discover novel SNPs and detect known SNPs include:
- Single-strand conformation polymorphism (SSCP)
- Electrochemical analysis
- Denaturating HPLC and gel electrophoresis
- Hybridization analysis
See also
Notes
- ↑ Barreiro LB, Laval G, Quach H, Patin E, Quintana-Murci L. (2008). "Natural selection has driven population differentiation in modern humans". Nature Genetics. 40: 340–345. doi:10.1038/ng.78. PMID 18246066.
- ↑ Nachman, Michael W. (2001). "Single nucleotide polymorphisms and recombination rate in humans". Trends in genetics. 17 (9): 481–485. doi:10.1016/S0168-9525(01)02409-X. PMID 11525814.
- ↑ M.A. Varela and W. Amos (2010). "Heterogeneous distribution of SNPs in the human genome: Microsatellites as predictors of nucleotide diversity and divergence". Genomics. 95: 151–159. doi:10.1016/j.ygeno.2009.12.003. PMID 20026267.
- ↑ PMID 23159550 (PMID 23159550)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ Carlson, Bruce (2008-06-15). "SNPs — A Shortcut to Personalized Medicine". Genetic Engineering & Biotechnology News. Mary Ann Liebert, Inc. 28 (12). Retrieved 2008-07-06.
(subtitle) Medical applications are where the market's growth is expected
- ↑ Sachidanandam, Ravi; Weissman, David; Schmidt, Steven C.; Kakol, Jerzy M.; Stein, Lincoln D.; Marth, Gabor; Sherry, Steve; Mullikin, James C.; Mortimore, Beverley J. (2001). "A map of human genome sequence variation containing 1.42 million single nucleotide polymorphisms". Nature. 409 (6822): 928–33. doi:10.1038/35057149. PMID 11237013.
- ↑ Singh, Monica; Singh, Puneetpal; Juneja, Pawan Kumar; Singh, Surinder; Kaur, Taranpal (2010). "SNP–SNP interactions within APOE gene influence plasma lipids in postmenopausal osteoporosis". Rheumatology International. 31 (3): 421–3. doi:10.1007/s00296-010-1449-7. PMID 20340021.
- ↑ NCBI dbSNP build 137 for human.
- ↑ PMID 13369537 (PMID 13369537)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ PMID 88735 (PMID 88735)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ PMID 1379413 (PMID 1379413)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ Fareed, M., Afzal, M (2013) "Single nucleotide polymorphism in genome-wide association of human population: A tool for broad spectrum service". Egyptian Journal of Medical Human Genetics 14: 123–134. http://dx.doi.org/10.1016/j.ejmhg.2012.08.001.
- ↑ PMID 11678778 (PMID 11678778)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ PMID 15349140 (PMID 15349140)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ PMID 16303243 (PMID 16303243)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ PMID 21992066 (PMID 21992066)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ Morita, Akihiko; Nakayama, Tomohiro; Doba, Nobutaka; Hinohara, Shigeaki; Mizutani, Tomohiko; Soma, Masayoshi (2007). "Genotyping of triallelic SNPs using TaqMan PCR". Molecular and Cellular Probes. 21 (3): 171–6. doi:10.1016/j.mcp.2006.10.005. PMID 17161935.
- ↑ Prodi, D.A.; Drayna, D; Forabosco, P; Palmas, MA; Maestrale, GB; Piras, D; Pirastu, M; Angius, A (2004). "Bitter Taste Study in a Sardinian Genetic Isolate Supports the Association of Phenylthiocarbamide Sensitivity to the TAS2R38 Bitter Receptor Gene". Chemical Senses. 29 (8): 697–702. doi:10.1093/chemse/bjh074. PMID 15466815.
- ↑ Ammitzbøll, Christian Gytz (28). "Non-Synonymous Polymorphisms in the FCN1 Gene Determine Ligand-Binding Ability and Serum Levels of M-Ficolin". PLoS ONE. 7 (11): e50585. doi:10.1371/journal.pone.0050585. Unknown parameter
|month=ignored (help); Check date values in:|date=, |year= / |date= mismatch(help) - ↑ PMID 11237013 (PMID 11237013)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ J.T. Den Dunnen (2008-02-20). "Recommendations for the description of sequence variants". Human Genome Variation Society. Retrieved 2008-09-05.
- ↑ den Dunnen, Johan T.; Antonarakis, Stylianos E. (2000). "Mutation nomenclature extensions and suggestions to describe complex mutations: A discussion". Human Mutation. 15 (1): 7–12. doi:10.1002/(SICI)1098-1004(200001)15:1<7::AID-HUMU4>3.0.CO;2-N. PMID 10612815.
- ↑ Ogino, Shuji; Gulley, Margaret L.; Den Dunnen, Johan T.; Wilson, Robert B.; Association for Molecular Pathology Training and Education Committee (2007). "Standard Mutation Nomenclature in Molecular DiagnosticsPractical and Educational Challenges". The Journal of Molecular Diagnostics. 9 (1): 1–6. doi:10.2353/jmoldx.2007.060081. PMC 1867422. PMID 17251329.
- ↑ Altshuler, D; Pollara, V J; Cowles, C R; Van Etten, W J; Baldwin, J; Linton, L; Lander, E S (2000). "An SNP map of the human genome generated by reduced representation shotgun sequencing". Nature. 407 (6803): 513–6. doi:10.1038/35035083. PMID 11029002.
- ↑ Drabovich, A.P.; Krylov, S.N. (2006). "Identification of base pairs in single-nucleotide polymorphisms by MutS protein-mediated capillary electrophoresis". Analytical chemistry. 78 (6): 2035–8. doi:10.1021/ac0520386. PMID 16536443.
- ↑ Griffin, T J; Smith, L M (2000). "Genetic identification by mass spectrometric analysis of single-nucleotide polymorphisms: ternary encoding of genotypes". Analytical chemistry. 72 (14): 3298–302. doi:10.1021/ac991390e. PMID 10939403.
References
- Nature Reviews Glossary
- Human Genome Project Information — SNP Fact Sheet
- Relation of SNP's with Cancer
External links
- NCBI resources — Introduction to SNPs from NCBI
- The SNP Consortium LTD — SNP search
- NCBI dbSNP database — "a central repository for both single base nucleotide substitutions and short deletion and insertion polymorphisms"
- HGMD — the Human Gene Mutation Database, includes rare mutations and functional SNPs
- SNPedia - a wiki devoted to the medical consequences of DNA variations, including software to analyze personal genomes
- International HapMap Project — "a public resource that will help researchers find genes associated with human disease and response to pharmaceuticals"
- GWAS Central — a central database of summary-level genetic association findings
- 1000 Genomes Project — A Deep Catalog of Human Genetic Variation
- SIFT — "An online tool that predicts the effect of SNPs on protein function"
- PolyPhen-2 - "An online tool that predicts the effect of nonsynonymous SNPs on protein function"
- MutationTaster - "Evaluates disease-causing potential of sequence alterations"
- WatCut — an online tool for the design of SNP-RFLP assays
- SNPStats — SNPStats, a web tool for analysis of genetic association studies
- Restriction HomePage — a set of tools for DNA restriction and SNP detection, including design of mutagenic primers
- American Association for Cancer Research Cancer Concepts Factsheet on SNPs
- PharmGKB — The Pharmacogenetics and Pharmacogenomics Knowledge Base, a resource for SNPs associated with drug response and disease outcomes.
- GEN-SNiP — Online tool that identifies polymorphisms in test DNA sequences.
- Rules for Nomenclature of Genes, Genetic Markers, Alleles, and Mutations in Mouse and Rat
- HGNC Guidelines for Human Gene Nomenclature
- SNP effect predictor with galaxy integration
- Human Gene Mutation Database
- GWAS Central
- Open SNP — a portal for sharing own SNP test results
- The HapMap Project
- CS1 maint: Multiple names: authors list
- Pages with incomplete PMID references
- Pages with citations using unsupported parameters
- CS1 errors: dates
- Articles containing potentially dated statements from June 2012
- Articles with invalid date parameter in template
- All articles containing potentially dated statements
- Molecular biology
- Population genetics
- DNA
- Genetic genealogy
- Biotechnology