Methscopolamine bromide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Turky Alkathery, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Methscopolamine bromide is an anticholinergic that is FDA approved for the treatment of peptic ulcer. Common adverse reactions include anticholinergic adverse reaction and headache.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Adjunctive therapy for the treatment of peptic ulcer.
- Methscopolamine bromide has not been shown to be effective in contributing to the healing of peptic ulcer, decreasing the rate of recurrence or preventing complications.
Dosage
- The average dosage of Methscopolamine Bromide Tablets is 2.5 mg one-half hour before meals and 2.5 fo 5 mg at bedtime. A starling dose of 12.5 mg daily will be clinically effective in most patients without the production of appreciable side effects.
- If the patient is experiencing symptoms such as severe abdominal pain or cramping which demand prompt relief, the drug may be started on a daily dosage of 20 mg, administered in doses of 5 mg one-half hour before meals and at bedtime. If very unpleasant side effects develop promptly, the daily dosage should be reduced. If neither symptomatic relief nor side effects appear, the daily dosage may be increased. Some patients have tolerated 30 mg daily with no unpleasant reactions.
- Patients whose dosage has been reduced to eliminate or modify side effects often continue to show adequate response both subjectively in relief of symptoms and objectively as measured by antisecretory effects.
- The ultimate aim of therapy is to arrive at a dosage which provides maximal clinical effectiveness with a minimum of unpleasant side effects. Many patients report no side effects on a dosage which gives complete relief of symptoms. On the other hand, some patients have reported severe side effects without appreciable symptomatic relief. Such patients must be considered unsuited for this therapy. Usually they have been or will prove to be similarly intolerant to other anticholinergic drugs. If methscopolamine bromide is to be used in a patient who gives a history of such intolerance, it should be started at a low dosage.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Methscopolamine bromide in adult patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Methscopolamine bromide in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Safety and efficacy in children have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Methscopolamine bromide in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Methscopolamine bromide in pediatric patients.
Contraindications
- Glaucoma; obstructive uropathy (e.g., bladder neck obstruction due to prostatic hypertrophy); obstructive disease of the gastrointestinal tract (e.g., pyloroduodenal stenosis); paralytic ileus; intestinal atony of the elderly or debilitated patient; unstable cardiovascular status in acute hemorrhage; severe ulcerative colitis; toxic megacolon complicating ulcerative colitis; myasthenia gravis.
- Methscopolamine Bromide 2.5 mg Tablets/ Methscopolamine Bromide 5 mg Tablets are contraindicated in patients who are hypersensitive to methscopolamine bromide or related drugs.
Warnings
- In the presence of high environmental temperature, heat prostration (fever and heat stroke due to decreased sweating) can occur with drug use.
- Diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy. In this instance treatment with this drug would be inappropriate and possibly harmful.
- Methscopolamine bromide may produce drowsiness or blurred vision. The patient should be cautioned regarding activities requiring mental alertness such as operating a motor vehicle or other machinery or performing hazardous work while taking this drug.
- With overdosage, a curare-like action may occur, i.e., neuromuscular blockade leading to muscular weakness and possible paralysis.
Precautions
General precautions
- Use Methscopolamine Bromide 2.5 mg Tablets/Methscopolamine Bromide 5 mg Tablets with caution in the elderly and in all patients with: autonomic neuropathy; hepatic or renal disease; or ulcerative colitis-large doses may suppress intestinal motility to the point of producing a paralytic ileus and for this reason precipitate or aggravate "toxic megacolon," a serious complication of the disease.
- The drug also should be used with caution in patients having hyperthyroidism, coronary heart disease, congestive heart failure, tachyrhythmia, Tachycardia, hypertension, or prostatic hypertrophy.
- Laboratory tests: Progress of the peptic ulcer under treatment should be followed by upper gastrointestinal contrast radiology or endoscopy to insure healing. Stool tests for occult blood and blood hemoglobin or hematocrit values should be followed to rule out bleeding from the ulcer.
Adverse Reactions
Clinical Trials Experience
- The following-adverse reactions have been observed, but there is not enough data to support an estimate of frequency.
- Cardiovascular: Tachycardia, palpitation.
- Allergic: Severe allergic reaction or drug idiosyncrasies including anaphylaxis.
- CNS: Headaches, nervousness, mental confusion, drowsiness, dizziness.
- Special Senses: Blurred vision, dilation of the pupil, cycloplegia, increased ocular tension, loss of taste.
- Renal: Urinary hesitancy and retention.
- Gastrointestinal: Nausea, vomiting, constipation, bloated feeling.
- Dermatologic: Decreased sweating, urticaria and other dermal manifestations.
- Miscellaneous: Xerostomia, weakness, insomnia, impotence, suppression of lactation.
Postmarketing Experience
- There is limited information regarding Postmarketing Experience
Drug Interactions
- Additive anticholinergic effects may result from concomitant use with antipsychotics, tricyclic antidepressants, and other drugs with anticholinergic effects. Concomitant administration with antacids may interfere with the absorption of methscopolamine bromide.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C
Animal reproduction studies have not been conducted with methscopolamine bromide. It also is not known whether methscopolamine bromide can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Methscopolamine bromide should be given to a pregnant woman only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Methscopolamine bromide in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Methscopolamine bromide during labor and delivery.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when methscopolamine bromide is administered to a nursing woman.
Pediatric Use
Safety and efficacy in children have not been established.
Geriatic Use
There is no FDA guidance on the use of Methscopolamine bromide in geriatric settings.
Gender
There is no FDA guidance on the use of Methscopolamine bromide with respect to specific gender populations.
Race
There is no FDA guidance on the use of Methscopolamine bromide with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Methscopolamine bromide in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Methscopolamine bromide in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Methscopolamine bromide in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Methscopolamine bromide in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral.
Monitoring
- There is limited information regarding drug monitoring
IV Compatibility
- There is limited information regarding IV Compatibility.
Overdosage
- The symptoms of overdosage with Methscopolamine Bromide 2.5 mg Tablets/Methscopolamine Bromide 5 mg Tablets progress from intensification of the usual side effects to CNS disturbances (from restlessness and excitement to psychotic behavior), circulatory changes (flushing, fall in blood pressure, circulatory failure), respiratory failure, paralysis, and coma.
- Measures to be taken are (1) induction of emesis and (2) injection of physostigmine 0.5 to 2 mg intravenously, and repeated as necessary up to a total of 5 mg. Fever may be treated symptomatically (alcohol sponging, ice packs). Excitement of a degree which demands attention may be managed with sodium thiopental 2% solution given slowly intravenously or chloral hydrate (100-200 ml of a 2% solution) by rectal infusion. In the event of progression of the curare-like effect to paralysis of the respiratory muscles, artificial respiration should be instituted and maintained until effective respiratory action returns.
- The oral LD50 in rats is 1,352 to 2,617 mg/kg.
- No data is available on the dialyzability of methscopolamine bromide.
Pharmacology
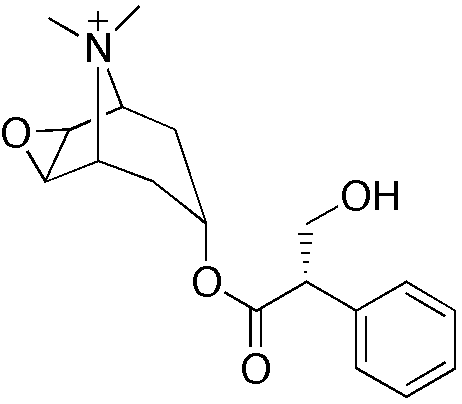
| |
Methscopolamine bromide
| |
| Systematic (IUPAC) name | |
| (1R,2S,4R,5S,7R)-{[(2R)-3-hydroxy-2-phenylpropanoyl]oxy}-9,9-dimethyl-3-oxa-9-azoniatricyclo[3.3.1.02,4]nonane | |
| Identifiers | |
| CAS number | |
| ATC code | A03 S01FA03 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 318.388 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | 3–4 hrs |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
- There is limited information regarding Mechanism of action.
Structure
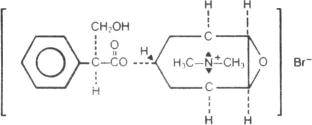
Methscopolamine Bromide 2.5 mg Tablets/Methscopolamine Bromide 5 mg Tablets contain methscopolamine bromide, an anticholinergic, which occurs as white crystals, or as a white odorless crystalline powder. Methscopolamine bromide melts at about 225°C with decomposition. The drug is freely soluble in water, slightly soluble in alcohol, and insoluble in acetone and in chloroform.
The chemical name for methscopolamine bromide is 3-Oxa-9-azoniatricyclo [3.3.1.02,4] nonane, 7-(3-hydroxy-1 -oxo-2-phenylpropoxy)-9, 9-dimethyl-, bromide, [7(S)-(1α, 2β, 4β, 5α, 7β]- and the molecular weight is 398.30.
The structural formula is represented below:
Pharmacodynamics
- There is limited information regarding pharmacodynamics.
Pharmacokinetics
- Methscopolamine bromide is a quaternary ammonium derivative of scopolamine. As a class, these agents are poorly and unreliably absorbed.1,2 Total absorption of quaternary ammonium derivatives of the alkaloids is 10-25%. Rate of absorption is not available. Quaternary ammonium salts have limited absorption from intact skin, and conjunctival penetration is poor.1 Little is known of the fate and excretion of most of these agents.1 Following oral administration, drug effects appear in about one hour and persist for 4 to 6 hours.2 Methscopolamine bromide has limited ability to cross the blood-brain barrier.34'5 The drug is excreted primarily in the urine and bile, or as unabsorbed drug in feces.2 There is no data on the presence of methscopolamine in breast milk; traces of atropine have been found after administration of atropine.
Nonclinical Toxicology
Carcinogenesis, mutagenesis, impairment of fertility
- No long-term studies in animals have been performed to evaluate carcinogenic potential.
Clinical Studies
- There is limited information regarding Clinical Studies.
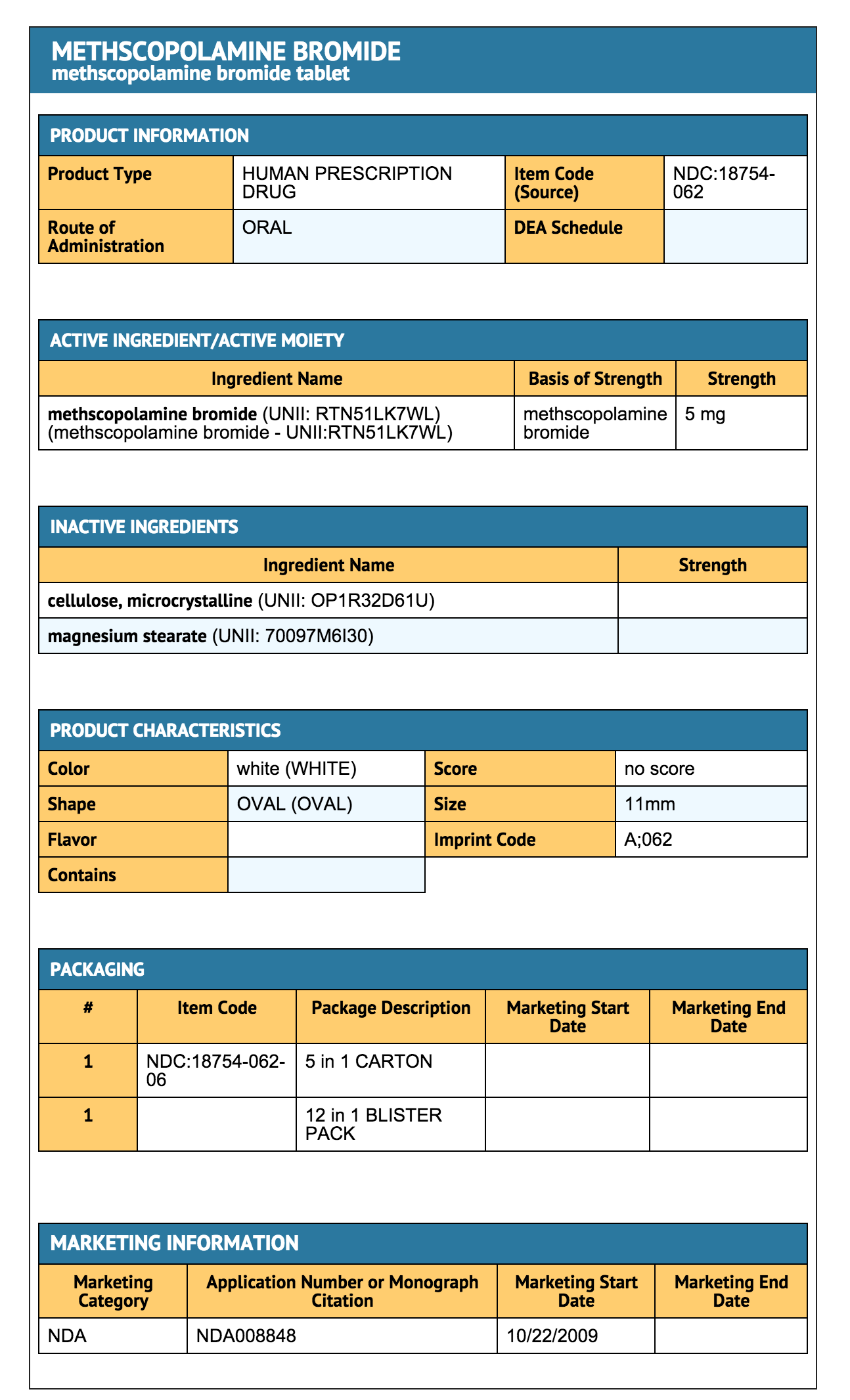
How Supplied
- Methscopolamine Bromide 2.5 mg Tablets are available as white, round tablets, debossed with "A" on one side and "061" on the other side, in the following package size:
- Bottles of 100 (NDC 18754-061-01)
- Methscopolamine Bromide 5 mg Tablets are available as white, oval tablets, debossed with "A" on one side and "062" on the other side, in the following package size:
- Dose Pack (5 blisters of 12 tablets)
- Box of 60 (NDC 18754-062-06)
Storage
- Store at controlled room temperature 15°-30°C (59°-86°F).
- KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Images
Drug Images
{{#ask: Page Name::Methscopolamine bromide |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel



{{#ask: Label Page::Methscopolamine bromide |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- There is limited information regarding Patient Counseling Information.
Precautions with Alcohol
Alcohol-Methscopolamine bromide interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- METHSCOPOLAMINE BROMIDE ®[1]
Look-Alike Drug Names
- There is limited information regarding Look-Alike Drug Names.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.