Methacholine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Turky Alkathery, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Methacholine is a non-selective muscarinic receptor agonist that is FDA approved for the diagnosis of bronchial airway hyperreactivity in subjects who do not have clinically apparent asthma. Common adverse reactions include headache, throat irritation, Iightheadedness and itching.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Methacholine chloride powder for inhalation is indicated for the diagnosis of bronchial airway hyperreactivity in subjects who do not have clinically apparent asthma.
Dosage
- Before methacholine chloride powder for inhalation Inhalation challenge is begun, baseline pulmonary function tests must be performed. A subject to be challenged must have an FEV1 of at least 70% of the predicted value.
- The target level for a positive challenge is a 20% reduction in the FEV1 compared with the baseline value after inhalation of the control sodium chloride solution (Note: Use the same diluent that the methacholine powder has been reconstituted with for the baseline spirometry). This target value should be calculated and recorded before methacholine challenge is started.
- Dilutions: (Note: Do not inhale powder. Do not handle this material if you have asthma or hay fever.) All dilutions should be made with 0.9% sodium chloride injection or 0.9% sodium chloride injection containing 0.4% phenol (pH 7.0) using sterile, empty USP Type I borosilcate glass vials. After adding the sodium chloride solution, shake each vial to obtain a clear solution (Note: When preparing dilutions, use only the same kind of diluent to prepare all concentrations).
- Dilutions A through D should be stored at 36° to 46°F (2° to 8°C) in a refrigerator and can be stored for not more than 2 weeks. [The unreconstituted powder should be stored at 59°F to 86°F (15° to 30°C)]. After this time, discard the vials and prepare new dilutions. Freezing does not affect the stability of dilutions A through D. Vial E must be prepared on the day of challenge.
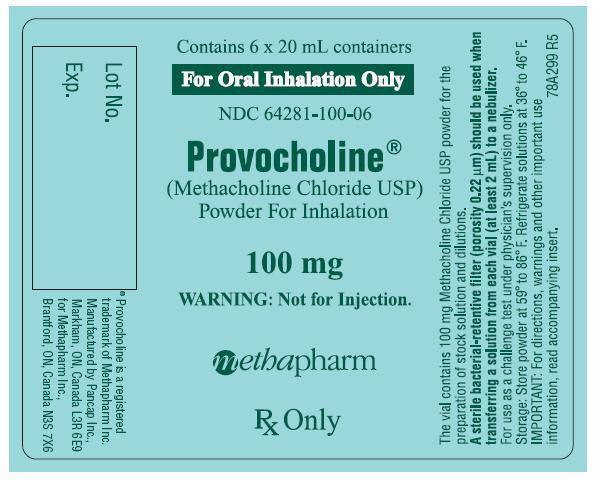
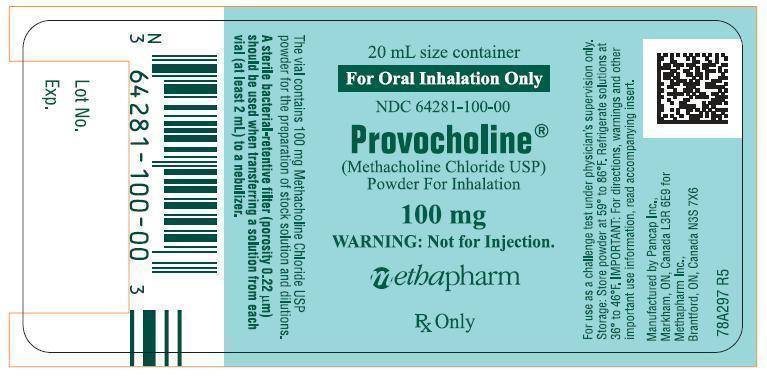
- A Sterile bacterial-retentive filter (porosity 0.22µm) should be used when transferring a solution from each vial (at least 2mL) to a nebulizer.
- Procedure: A standardized procedure for inhalation has been developed.
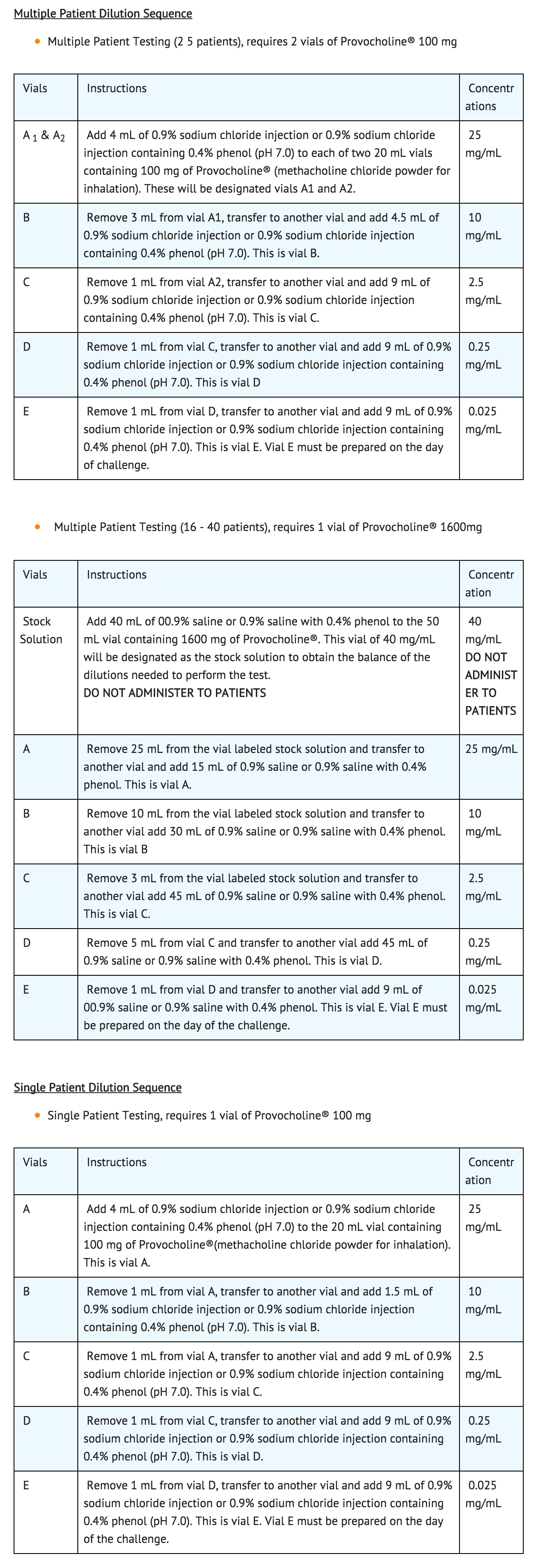
- The challenge is performed by giving a subject ascending serial concentrations of methacholine. At each concentration, five breaths are administered by a nebulizer that permits intermittent delivery time of 0.6 seconds by a breath-actuated timing device (dosimeter).
- At each of five inhalations of a serial concentration, the subject begins at functional residual capacity (FRC) and slowly and completely inhales the dose delivered. Within 5 minutes, FEV1 values are determined. The procedure ends either when there is a 20% or greater reduction in FEV1 compared with the baseline sodium chloride solution value (i.e., a positive response) or if 188.88 total cumulative units has been administered (see table below) and the FEV1 has been reduced by 14% or less (i.e., a negative response). If there is a reduction of 15% to 19% in the FEV1 compared with baseline, either the challenge may be repeated at that concentration or a higher concentration may be given as long as the dosage administered does not result in total cumulative units exceeding 188.88.
- The following is a suggested schedule for the administration of methacholine (methacholine chloride powder for inhalation) challenge. Cumulative units are calculated by multiplying the number of breaths by the concentration administered.
- An inhaled beta-agonist may be administered after methacholine challenge to expedite the return of the FEV1 to baseline and to relieve the discomfort of the subject. Most patients revert to normal pulmonary function within 5 minutes following bronchodilators or within 30 to 45 minutes without any bronchodilator.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Methacholine in adult patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Methacholine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- The safety and efficacy of methacholine inhalation challenge have not been established in children below the age of 5 years.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Methacholine in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Methacholine in pediatric patients.
Contraindications
- Methacholine chloride powder for inhalation is contraindicated in patients with known hypersensitivity to this drug or to other parasympathomimetic agents.
- Repeated administration of methacholine by inhalation other than on the day that a patient undergoes challenge with increasing doses is contraindicated.
- Inhalation challenge should not be performed in patients receiving any beta-adrenergic blocking agent because in such patients responses to methacholine chloride can be exaggerated or prolonged, and may not respond as readily to accepted modalities of treatment.
Warnings
Precations
General
- Administration of methacholine chloride powder for inhalation to patients with epilepsy, cardiovascular disease accompanied by bradycardia, vagotonia, peptic ulcer disease, thyroid disease, urinary tract obstruction or other condition that could be adversely affected by a cholinergic agent should be undertaken only if the physician feels benefit to the individual outweighs the potential risks.
Adverse Reactions
Clinical Trials Experience
- Adverse reactions associated with 153 inhaled methacholine chloride challenges include one occurrence each of headache, throat irritation, Iightheadedness and itching.
- Methacholine chloride powder for inhalation is to be administered only by inhalation. When administered orally or by injection, methacholine chloride is reported to be associated with nausea and vomiting, substernal pain or pressure, hypotension,fainting and transient complete heart block.
Postmarketing Experience
- There is limited information regarding postmarketing experience.
Drug Interactions
- There is limited information regarding Drug Interactions.
Use in Specific Populations
Pregnancy
- Animal reproduction studies have not been conducted with methacholine chloride. It is not known whether methacholine chloride can cause fetal harm when administered to a pregnant patient or affect reproductive capacity. Methacholine chloride should be given to a pregnant woman only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Methacholine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Methacholine during labor and delivery.
Nursing Mothers
- Methacholine inhalation challenge should not be administered to a nursing mother since it is not known whether methacholine chloride when inhaled is excreted in breast milk.
Pediatric Use
- The safety and efficacy of methacholine inhalation challenge have not been established in children below the age of 5 years.
Geriatic Use
There is no FDA guidance on the use of Methacholine in geriatric settings.
Gender
There is no FDA guidance on the use of Methacholine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Methacholine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Methacholine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Methacholine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Methacholine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Methacholine in patients who are immunocompromised.
Administration and Monitoring
Administration
- Respiratory (inhalation)
Monitoring
There is limited information regarding drug monitoring
IV Compatibility
- There is limited information regarding IV Compatibility.
Overdosage
- Methacholine chloride powder for inhalation is to be administered only by inhalation. When administered orally or by injection, overdosage with methacholine chloride can result in a syncopal reaction, with cardiac arrest and loss of consciousness. Serious toxic reactions should be treated with 0.5 mg to 1 mg of atropine sulfate, administered IM or IV.
- The acute (24 hour) oral LD50 of methacholine chloride and related compounds is 1100 mg/kg in the mouse and 750 mg/kg in the rat.
- Cynomolgus monkeys were exposed to a 2% (20 mg/mL) aerosol of methacholine chloride in acute (10 minute) and subchronic (7 day) inhalation toxicity studies. In the former study, animals exposed to the aerosol for up to 10 minutes demonstrated an increase in respiratory rate and decrease in tidal volume after 30 seconds. These changes peaked at 2 minutes and were followed by a rise in pulmonary resistance and a decrease in compliance. Pulmonary function returned to normal 20 to 25 minutes after exposure ended. In the 7 day study, monkeys were given daily inhalations equivalent to the maximum and roughly five times the maximum standard human dose. Although the typical pulmonary response/ recovery sequence was observed, distinct changes in airway resistance were noted at the end of the study. These changes were not rapidly reversed in the maximum equivalent standard dose group, which was observed for 9 weeks.
Pharmacology
| |
Methacholine
| |
| Systematic (IUPAC) name | |
| 2-(Acetyloxy)-N,N,N-trimethylpropan-1-aminium | |
| Identifiers | |
| CAS number | 62-51-1 (chloride) |
| ATC code | none |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox+ |
| Mol. mass | 160.234 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
- Methacholine chloride is the ß-methyl homolog of acetylcholine and differs from the latter primarily in its greater duration and selectivity of action. Bronchial smooth muscle contains significant parasympathetic (cholinergic) innervation.
- Bronchoconstriction occurs when the vagus nerve is stimulated and acetylcholine is released from the nerve endings. Muscle constriction is essentially confined to the local site of release because acetylcholine is rapidly inactivated by acetylcholinesterase.
- Compared with acetylcholine, methacholine chloride is more slowly hydrolyzed by acetylcholinesterase and is almost totally resistant to inactivation by nonspecific cholinesterase or pseudocholinesterase. When a sodium chloride solution containing methacholine chloride is inhaled, subjects with asthma are markedly more sensitive to methacholine-induced bronchoconstriction than are healthy subjects. This difference in response is the pharmacologic basis for the methacholine (methacholine chloride powder for inhalation) inhalation diagnostic challenge. However, it should be recognized that methacholine challenge may occasionally be positive after influenza, upper respiratory infections or immunizations, in very young or very old patients, or in patients with chronic lung disease (cystic fibrosis, sarcoidosis, tuberculosis, chronic obstructive pulmonary disease). The challenge may also be positive in patients with allergic rhinitis without asthma, in smokers, in patients after exposure to air pollutants, or in patients who have had or will in the future develop asthma.
Structure
- Methacholine chloride powder for inhalation is a parasympathomimetic (cholinergic)) bronchoconstrictor agent to be administered in solution only, by inhalation, for diagnostic purposes. Each 20 mL vial contains 100 mg and each 50 mL vial contains 1600 mg of methacholine chloride powder which is to be reconstituted with 0.9% sodium chloride injection or 0.9% sodium chloride injection containing 0.4% phenol (pH 7.0).
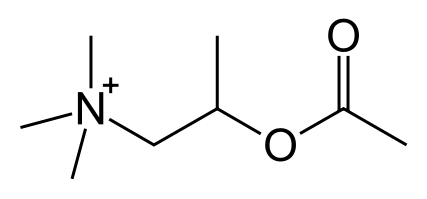
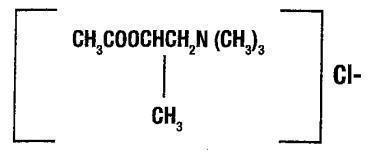
- Chemically, methacholine chloride (the active ingredient) is 1-propanaminium, 2-(acetyloxy)N,N,N, - trimethyl,-chloride. It is a white to practically white deliquescent compound, soluble in water. Methacholine chloride has an empirical formula of C8H18ClN02, a calculated molecular weight of 195.69, and the following structural formula:
Pharmacodynamics
There is limited information regarding Methacholine Pharmacodynamics in the drug label.
Pharmacokinetics
- There are no pharmacokinetic data available on methacholine chloride.
Nonclinical Toxicology
- There have been no studies with methacholine chloride that would permit an evaluation of its carcinogenic or mutagenic potential or of its effect on fertility.
Clinical Studies
- There is limited information regarding Clinical Studies.
How Supplied
- 20mL amber vial containing 100mg methacholine chloride powder which is to be reconstituted with 0.9% sodium chloride injection containing 0.4% phenol (pH 7.0) – boxes of 6 (NDC 64281-100-06).
- 50 mL amber vials containing 1600 mg of methacholine chloride powder which is to be reconstituted with 0.9% sodium chloride injection or with 0.9% sodium chloride injection containing 0.4% phenol (pH 7.0) – single vial per box (NDC 64281-100-16).
Storage
- Store the powder at 59° to 86°F (15° to 30°C). Refrigerate the reconstituted solutions (dilutions A-D) at 36° to 46°F (2° to 8°C) for not more than 2 weeks. Dilution E must be prepared on the day of the challenge.
Images
Drug Images
{{#ask: Page Name::Methacholine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Methacholine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- To assure the safe and effective use of Provocholine® inhalation challenge, the following instructions and information should be given to patients:
- Patients should be instructed regarding symptoms that may occur as a result of the test and how such symptoms can be managed.
- Female patient should inform her physician if she is pregnant, or the date of her last onset of menses, or the date and result of her last pregnancy test.
Precautions with Alcohol
- Alcohol-Methacholine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- PROVOCHOLINE®[1]
Look-Alike Drug Names
- There is limited information regarding Look-Alike Drug Names.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.