Liquid oxygen
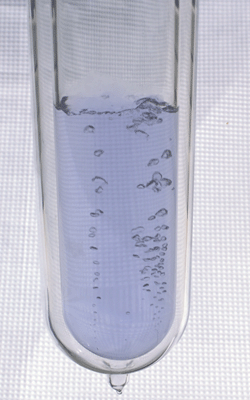
Liquid oxygen (also LOx, LOX or Lox in the aerospace, submarine and gas industry) is a form of the element oxygen. It has a pale blue color and is strongly paramagnetic. Liquid oxygen has a density of 1.141 g/cm³ (1.141 kg/L) and is moderately cryogenic (freezing point: 50.5 K (−222.65 °C), boiling point: 90.188 K (−182.96 °C) at 101.325 kPa (760 mm Hg). In commerce, liquid oxygen is classified as an industrial gas and is widely used for industrial and medical purposes. Liquid oxygen is obtained from the oxygen found naturally in air by fractional distillation. Liquid oxygen has an expansion ratio of 860:1, and because of this, is used in commercial and military aircraft today.
Due to its cryogenic nature, LOX can cause the materials it touches to become extremely brittle. Liquid oxygen is also a very powerful oxidising agent: organic materials will burn rapidly and energetically in liquid oxygen. Further; if soaked in LOX some can detonate unpredictably on subsequent contact. Petrochemicals often exhibit this behavior, including asphalt.
LOX is a common liquid oxidizer propellant for spacecraft rocket applications, usually in combination with liquid hydrogen or kerosene. It was used in the very first rocket applications like the V2 missile and Redstone, R-7 Semyorka or Atlas boosters. LOX is useful in this role because it creates a high specific impulse. LOX was also used in some early ICBMs, although more modern ICBMs do not use LOX because its cryogenic properties and need for regular replenishment to replace boiloff make it harder to maintain and launch quickly. Despite this, many modern rockets do use LOX, including the main engines on the Space Shuttle. During World War II, liquid oxygen was used as an oxidizer in several Nazi Germany military rocket designs, under name A-Stoff and Sauerstoff.
LOX also had extensive use in making oxyliquit explosives, but is rarely used now due to a high rate of accidents.
The tetraoxygen molecule (O4) was first predicted in 1924 by Gilbert N. Lewis, who proposed it as an explanation for the failure of liquid oxygen to obey Curie's law.[1] Today it seems Lewis was off, but not by far: computer simulations indicate that although there are no stable O4 molecules in liquid oxygen, O2 molecules do tend to associate in pairs with antiparallel spins, forming transient O4 units.[2]
Liquid nitrogen has a significantly lower boiling point, at −196 °C (77 K) than oxygen's −183 °C (90 K), and vessels containing liquid nitrogen can condense oxygen from air: when most of the nitrogen has evaporated from such a vessel there is a risk that liquid oxygen remaining can react violently with organic material. Conversely, liquid nitrogen or liquid air can be oxygen-enriched by letting it stand in open air; atmospheric oxygen dissolves in it, while nitrogen evaporates preferentially.
See also
- List of Stoffs
- Oxygen
- Karol Olszewski and Zygmunt Florenty Wróblewski
- Rocket fuel
- Solid oxygen
- Tetraoxygen
References
- ↑ Lewis, Gilbert N. (1924). "The Magnetism of Oxygen and the Molecule O2". Journal of the American Chemical Society. 46 (9): 2027&ndash, 2032. doi:10.1021/ja01674a008. Unknown parameter
|month=ignored (help) - ↑ Oda, Tatsuki (2004). "Noncollinear magnetism in liquid oxygen: A first-principles molecular dynamics study". Physical Review B. 70 (134402): 1&ndash, 19. doi:10.1103/PhysRevB.70.134402. Unknown parameter
|coauthors=ignored (help); Unknown parameter|month=ignored (help)
ar:أكسجين سائل lv:Šķidrais skābeklis ms:Oksigen cair fi:Nestehappi