Hepatitis B immunoglobulin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Hepatitis B immunoglobulin is a human immune globulin that is FDA approved for the treatment of hepatitis B. Common adverse reactions include urticaria, angioedema, anaphylaxis, and pain at the injection site.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Recommendations on post-exposure prophylaxis are based on available efficacy data and on the likelihood of future HBV exposure for the person requiring treatment. In all exposures, a regimen combining Hepatitis B Immune Globulin (Human) with hepatitis B vaccine will provide both short- and long-term protection, will be less costly than the two-dose Hepatitis B Immune Globulin (Human) treatment alone, and is the treatment of choice.HyperHEP B S/D is indicated for post-exposure prophylaxis in the following situations:
Acute Exposure to Blood Containing HBsAg
- After either parenteral exposure, e.g., by accidental “needlestick” or direct mucous membrane contact (accidental splash), or oral ingestion (pipetting accident) involving HBsAg-positive materials such as blood, plasma or serum. For inadvertent percutaneous exposure, a regimen of two doses of Hepatitis B Immune Globulin (Human), one given after exposure and one a month later, is about 75% effective in preventing hepatitis B in this setting.
Perinatal Exposure of Infants Born to HBsAg-positive Mothers
- Infants born to HBsAg-positive mothers are at risk of being infected with hepatitis B virus and becoming chronic carriers. This risk is especially great if the mother is HBeAg-positive.For an infant with perinatal exposure to an HBsAg-positive and HBeAg-positive mother, a regimen combining one dose of Hepatitis B Immune Globulin (Human) at birth with the hepatitis B vaccine series started soon after birth is 85%–95% effective in preventing development of the HBV carrier state. Regimens involving either multiple doses of Hepatitis B Immune Globulin (Human) alone or the vaccine series alone have 70%–90% efficacy, while a single dose of Hepatitis B Immune Globulin (Human) alone has only 50% efficacy.
Sexual Exposure to an HBsAg-positive Person
- Sex partners of HBsAg-positive persons are at increased risk of acquiring HBV infection. For sexual exposure to a person with acute hepatitis B, a single dose of Hepatitis B Immune Globulin (Human) is 75% effective if administered within 2 weeks of last sexual exposure.
Household Exposure to Persons with Acute HBV Infection
- Since infants have close contact with primary care-givers and they have a higher risk of becoming HBV carriers after acute HBV infection, prophylaxis of an infant less than 12 months of age with Hepatitis B Immune Globulin (Human) and hepatitis B vaccine is indicated if the mother or primary caregiver has acute HBV infection.
- Administration of Hepatitis B Immune Globulin (Human) either preceding or concomitant with the commencement of active immunization with Hepatitis B Vaccine provides for more rapid achievement of protective levels of hepatitis B antibody, than when the vaccine alone is administered.Rapid achievement of protective levels of antibody to hepatitis B virus may be desirable in certain clinical situations, as in cases of accidental inoculations with contaminated medical instruments. Administration of Hepatitis B Immune Globulin (Human) either 1 month preceding or at the time of commencement of a program of active vaccination with Hepatitis B Vaccine has been shown not to interfere with the active immune response to the vaccine.
Dosage
Acute Exposure to Blood Containing HBsAg
- TABLE 1 summarizes prophylaxis for percutaneous (needlestick or bite), ocular, or mucous-membrane exposure to blood according to the source of exposure and vaccination status of the exposed person. For greatest effectiveness, passive prophylaxis with Hepatitis B Immune Globulin (Human) should be given as soon as possible after exposure (its value beyond 7 days of exposure is unclear). If Hepatitis B Immune Globulin (Human) is indicated (see TABLE 1), an injection of 0.06 mL/kg of body weight should be administered intramuscularly as soon as possible after exposure and within 24 hours, if possible. Consult Hepatitis B Vaccine package insert for dosage information regarding that product.
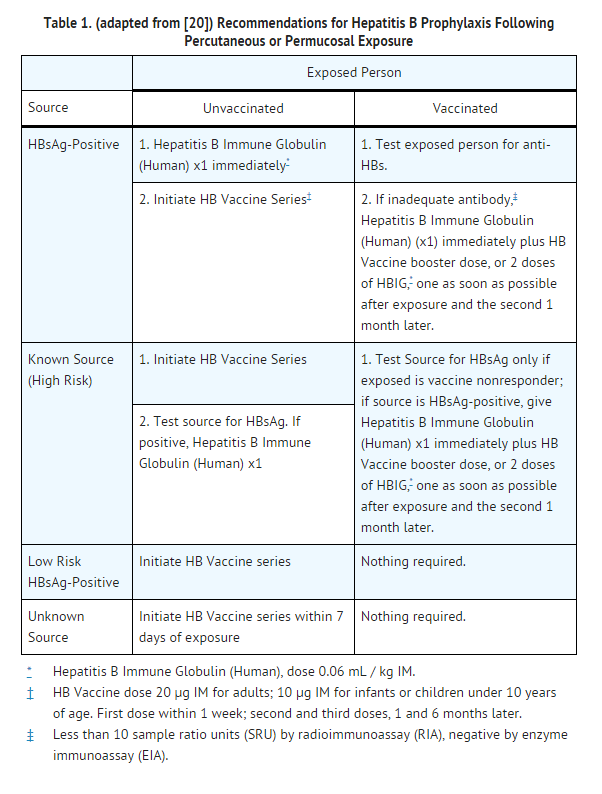
- For persons who refuse Hepatitis B Vaccine, a second dose of Hepatitis B Immune Globulin (Human) should be given 1 month after the first dose.
Prophylaxis of Infants Born to HBsAg and HBeAg Positive Mothers
- Efficacy of prophylactic Hepatitis B Immune Globulin (Human) in infants at risk depends on administering Hepatitis B Immune Globulin (Human) on the day of birth. It is therefore vital that HBsAg-positive mothers be identified before delivery.
- Hepatitis B Immune Globulin (Human) (0.5 mL) should be administered intramuscularly (IM) to the newborn infant after physiologic stabilization of the infant and preferably within 12 hours of birth. Hepatitis B Immune Globulin (Human) efficacy decreases markedly if treatment is delayed beyond 48 hours. Hepatitis B Vaccine should be administered IM in three doses of 0.5 mL of vaccine (10 μg) each. The first dose should be given within 7 days of birth and may be given concurrently with Hepatitis B Immune Globulin (Human) but at a separate site. The second and third doses of vaccine should be given 1 month and 6 months, respectively, after the first. If administration of the first dose of Hepatitis B Vaccine is delayed for as long as 3 months, then a 0.5 mL dose of Hepatitis B Immune Globulin (Human) should be repeated at 3 months. If Hepatitis B Vaccine is refused, the 0.5 mL dose of Hepatitis B Immune Globulin (Human) should be repeated at 3 and 6 months. Hepatitis B Immune Globulin (Human) administered at birth should not interfere with oral polio and diphtheria-tetanus-pertussis vaccines administered at 2 months of age.
Sexual Exposure to an HBsAg-positive Person
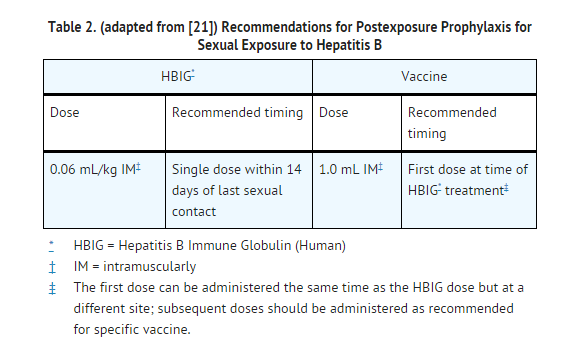
- All susceptible persons whose sex partners have acute hepatitis B infection should receive a single dose of HBIG (0.06 mL/kg) and should begin the hepatitis B vaccine series if prophylaxis can be started within 14 days of the last sexual contact or if sexual contact with the infected person will continue (see TABLE 2 below). Administering the vaccine with HBIG may improve the efficacy of postexposure treatment. The vaccine has the added advantage of conferring long-lasting protection.
Household Exposure to Persons with Acute HBV Infection
- Prophylactic treatment with a 0.5 mL dose of Hepatitis B Immune Globulin (Human) and hepatitis B vaccine is indicated for infants <12 months of age who have been exposed to a primary care-giver who has acute hepatitis B. Prophylaxis for other household contacts of persons with acute HBV infection is not indicated unless they have had identifiable blood exposure to the index patient, such as by sharing toothbrushes or razors. Such exposures should be treated like sexual exposures. If the index patient becomes an HBV carrier, all household contacts should receive hepatitis B vaccine.
- Hepatitis B Immune Globulin (Human) may be administered at the same time (but at a different site), or up to 1 month preceding Hepatitis B Vaccination without impairing the active immune response from Hepatitis B Vaccination.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Administer intramuscularly. Do not inject intravenously.
- Hepatitis B Immune Globulin (Human) — HyperHEP B® S/D is supplied in a syringe with an attached UltraSafe® Needle Guard for your protection and convenience, as well as in vials. Please follow instructions below for proper use of syringe and UltraSafe® Needle Guard.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Hepatitis B immunoglobulin in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Hepatitis B immunoglobulin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Hepatitis B immunoglobulin in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Hepatitis B immunoglobulin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Hepatitis B immunoglobulin in pediatric patients.
Contraindications
- None known.
Warnings
- HyperHEP B S/D is made from human plasma. Products made from human plasma may contain infectious agents, such as viruses, and, theoretically, the Creutzfeldt-Jakob Disease (CJD) agent that can cause disease. The risk that such products will transmit an infectious agent has been reduced by screening plasma donors for prior exposure to certain viruses, by testing for the presence of certain current virus infections, and by inactivating and/or removing certain viruses. Despite these measures, such products can still potentially transmit disease. There is also the possibility that unknown infectious agents may be present in such products. Individuals who receive infusions of blood or plasma products may develop signs and/or symptoms of some viral infections, particularly hepatitis C. ALL infections thought by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to Grifols Therapeutics Inc. [1-800-520-2807].
- The physician should discuss the risks and benefits of this product with the patient, before prescribing or administering it to the patient.
- HyperHEP B S/D should be given with caution to patients with a history of prior systemic allergic reactions following the administration of human immune globulin preparations. Epinephrine should be available.
- In patients who have severe thrombocytopenia or any coagulation disorder that would contraindicate intramuscular injections, Hepatitis B Immune Globulin (Human) should be given only if the expected benefits outweigh the risks.
Adverse Reactions
Clinical Trials Experience
- Local pain and tenderness at the injection site, urticaria and angioedema may occur; anaphylactic reactions, although rare, have been reported following the injection of human immune globulin preparations.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Hepatitis B immunoglobulin in the drug label.
Drug Interactions
- Although administration of Hepatitis B Immune Globulin (Human) did not interfere with measles vaccination, it is not known whether Hepatitis B Immune Globulin (Human) may interfere with other live virus vaccines. Therefore, use of such vaccines should be deferred until approximately 3 months after Hepatitis B Immune Globulin (Human) administration. Hepatitis B Vaccine may be administered at the same time, but at a different injection site, without interfering with the immune response.No interactions with other products are known.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Pregnancy Category C
- Animal reproduction studies have not been conducted with HyperHEP B S/D. It is also not known whether HyperHEP B S/D can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. HyperHEP B S/D should be given to a pregnant woman only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Hepatitis B immunoglobulin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Hepatitis B immunoglobulin during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Hepatitis B immunoglobulin with respect to nursing mothers.
Pediatric Use
- Safety and effectiveness in the pediatric population have not been established.
Geriatic Use
There is no FDA guidance on the use of Hepatitis B immunoglobulin with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Hepatitis B immunoglobulin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Hepatitis B immunoglobulin with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Hepatitis B immunoglobulin in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Hepatitis B immunoglobulin in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Hepatitis B immunoglobulin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Hepatitis B immunoglobulin in patients who are immunocompromised.
Administration and Monitoring
Administration
- Intramuscular
Monitoring
There is limited information regarding Monitoring of Hepatitis B immunoglobulin in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Hepatitis B immunoglobulin in the drug label.
Overdosage
- Although no data are available, clinical experience with other immunoglobulin preparations suggests that the only manifestations would be pain and tenderness at the injection site.
Pharmacology
Hepatitis B immune globulin
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | J06 |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
C(US) |
| Legal status | |
| Routes | intramuscular injection |
Mechanism of Action
- There is limited information regarding Mechanism of Action of Hepatitis B immunoglobulin in the drug label.
Structure
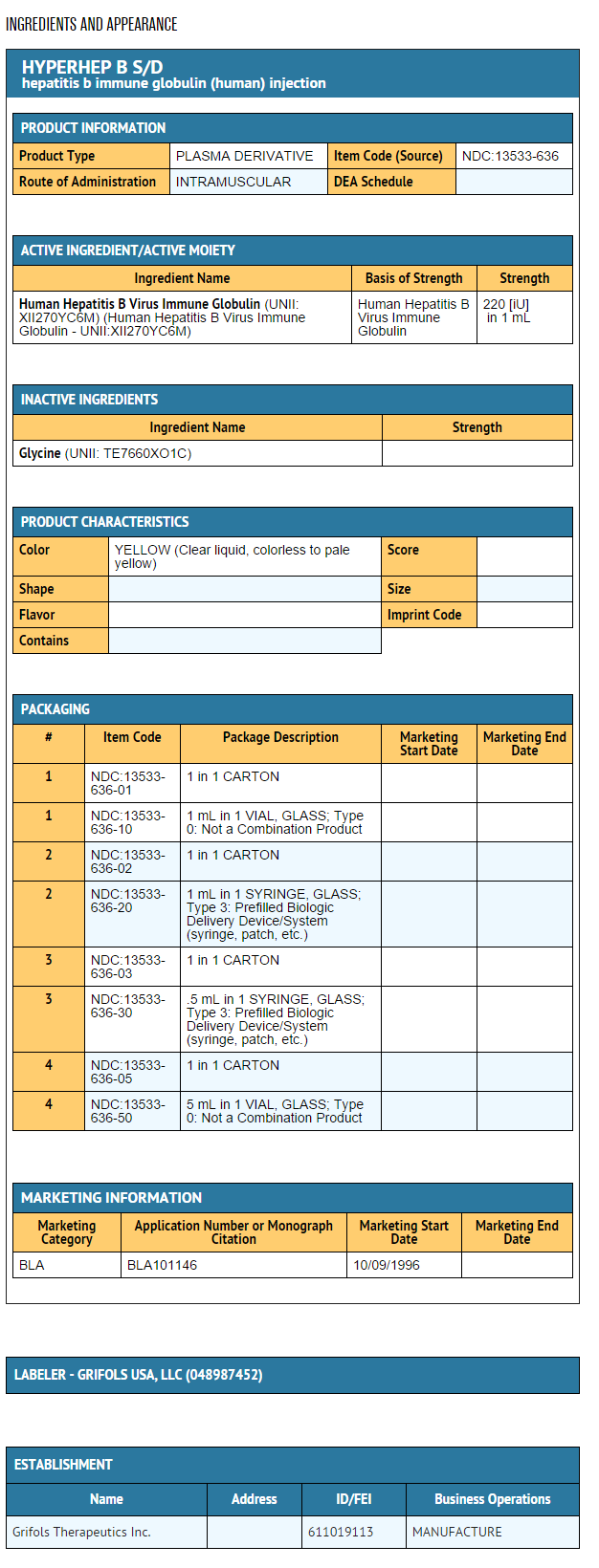
- Hepatitis B Immune Globulin (Human) — HyperHEP B®S/D treated with solvent/detergent is a colorless to pale yellow or pink sterile solution of hepatitis B hyperimmune immune globulin for intramuscular administration; it is preservative-free and latex-free. HyperHEP B S/D is prepared by cold ethanol fractionation from the plasma of donors with high titers of antibody to the hepatitis B surface antigen (anti-HBs). The immune globulin is isolated from solubilized Cohn Fraction II. The Fraction II solution is adjusted to a final concentration of 0.3% tri-n-butyl phosphate (TNBP) and 0.2% sodium cholate. After the addition of solvent (TNBP) and detergent (sodium cholate), the solution is heated to 30°C and maintained at that temperature for not less than 6 hours. After the viral inactivation step, the reactants are removed by precipitation, filtration and finally ultrafiltration and diafiltration. HyperHEP B S/D is formulated as a 15–18% protein solution at a pH of 6.4–7.2 in 0.21–0.32 M glycine. HyperHEP B S/D is then incubated in the final container for 21–28 days at 20–27°C. Each vial or syringe contains anti-HBs antibody equivalent to or exceeding the potency of anti-HBs in a U.S. reference hepatitis B immune globulin (Center for Biologics Evaluation and Research, FDA). The U.S. reference has been tested against the World Health Organization standard Hepatitis B Immune Globulin and found to be equal to 220 international units (IU) per mL.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Hepatitis B immunoglobulin in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Hepatitis B immunoglobulin in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Hepatitis B immunoglobulin in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Hepatitis B immunoglobulin in the drug label.
How Supplied
- HyperHEP B S/D is supplied in a 0.5 mL neonatal single dose syringe with attached needle, a 1 mL single dose syringe with attached needle and a 1 mL and a 5 mL single dose vial. HyperHEP B S/D is preservative-free and latex-free.
Storage
- Store at 2–8°C (36–46°F). Do not freeze. Do not use after expiration date.
Images
Drug Images
{{#ask: Page Name::Hepatitis B immunoglobulin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Hepatitis B immunoglobulin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Hepatitis B immunoglobulin in the drug label.
Precautions with Alcohol
- Alcohol-Hepatitis B immunoglobulin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- HYPERHEP B S/D®[1]
Look-Alike Drug Names
- A® — B®[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "human hepatitis b virus immune globulin injection".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Hepatitis B immunoglobulin
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Hepatitis B immunoglobulin |Label Name=Hepatitis B immunoglobulin11.png
}}
{{#subobject:
|Label Page=Hepatitis B immunoglobulin |Label Name=Hepatitis B immunoglobulin11.png
}}