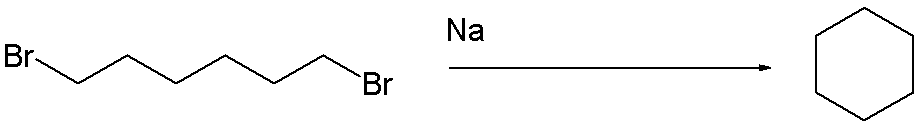Cyclohexane
|
WikiDoc Resources for Cyclohexane |
|
Articles |
|---|
|
Most recent articles on Cyclohexane Most cited articles on Cyclohexane |
|
Media |
|
Powerpoint slides on Cyclohexane |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Cyclohexane at Clinical Trials.gov Clinical Trials on Cyclohexane at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Cyclohexane
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Cyclohexane Discussion groups on Cyclohexane Patient Handouts on Cyclohexane Directions to Hospitals Treating Cyclohexane Risk calculators and risk factors for Cyclohexane
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Cyclohexane |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Cyclohexane is a cycloalkane with the molecular formula C6H12. Cyclohexane is used as a nonpolar solvent for the chemical industry, and also as a raw material for the industrial production of adipic acid and caprolactam, both of which are intermediates used in the production of nylon. On an industrial scale, cyclohexane is produced by reacting benzene with hydrogen. Due to its unique chemical and conformational properties, cyclohexane is also used in labs in analysis and as a standard.
Chemical conformation
Contrary to popular belief, the 6 vertexed ring does not conform to the shape of a perfect hexagon. The conformation of a flat 2D planar hexagon has considerable angle strain due to the fact that its bonds are not 109.5 degrees; the torsional strain would also be considerable due to all eclipsed bonds. Therefore, to reduce torsional strain, cyclohexane adopts a three-dimensional structure known as the chair conformation. The new conformation puts the carbons at an angle of 109.5°. Half of the hydrogens are in the plane of the ring (equatorial) while the other half are perpendicular to the plane (axial). This conformation allows for the most stable structure of cyclohexane. Another conformation of cyclohexane exists, known as boat conformation, but it interconverts to the slightly more stable chair formation. If cyclohexane is mono-substituted with a large substituent, then the substituent will most likely be found attached in an equatorial position, as this is the slightly more stable conformation.
Cyclohexane has the lowest angle and torsional strain of all the cycloalkanes, as a result cyclohexane has been deemed a 0 in total ring strain, a combination of angle and torsional strain. This also makes cyclohexane the most stable of the cycloalkanes and therefore will produce the least amount of heat when burned compared to the other cycloalkanes.
Reactions with cyclohexane
Pure cyclohexane in itself is rather unreactive, being a non-polar, hydrophobic hydrocarbon. It can react with very strong acids such as the superacid system HF + SbF5 which will cause forced protonation and "hydrocarbon cracking". Substituted cyclohexanes, however, may be reactive under a variety of conditions, many of which are important to organic chemistry.
Cyclohexane derivatives
The specific arrangement of functional groups in cyclohexane derivatives, and indeed in most cycloalkane molecules, is extremely important in chemical reactions, especially reactions involving nucleophiles. Substituents on the ring must be in the axial formation to react with other molecules. For example, the reaction of bromocyclohexane and a common nucleophile, a hydroxide anion (OH−), would result in cyclohexene:
C6H11Br + OH− → C6H10 + H2O + Br−
This reaction, commonly known as an elimination reaction or dehalogenation (specifically E2), requires that the bromine substituent be in the axial formation, opposing another axial H atom to react. Assuming that the bromocyclohexane was in the appropriate formation to react, the E2 reaction would commence as such:
- The electron pair bond between the C-Br moves to the Br, forming Br− and setting it free from cyclohexane
- The nucleophile (-OH) gives an electron pair to the adjacent axial H, setting H free and bonding to it to create H2O
- The electron pair bond between the adjacent axial H moves to the bond between the two C-C making it C=C
Note: All three steps happen simultaneously, characteristic of all E2 reactions.
The reaction above will generate mostly E2 reactions and as a result the product will be mostly (~70%) cyclohexene. However, the percentage varies with conditions, and generally, two different reactions (E2 and Sn2) compete. In the above reaction, an Sn2 reaction would substitute the bromine for a hydroxyl (OH-) group instead, but once again, the Br must be in axial to react. Once the SN2 substitution is complete, the newly substituted OH group would flip back to the more stable equatorial position quickly (~1 millisecond).
Uses
Commercially most of cyclohexane produced is converted into cyclohexanone-cyclohexanol mixture (or "KA oil") by catalytic oxidation. KA oil is then used as a raw material for adipic acid and caprolactam. Practically, if the cyclohexanol content of KA oil is higher than cyclohexanone, it is more likely(economical) to be converted into adipic acid, and the reverse case, caprolactam production is more likely. Such ratio in KA oil can be controlled by selecting suitable oxidation catalyts. Some of cyclohexane is used as an organic solvent.
Cyclohexane in research
The usefulness of cyclohexane in research is not to be underestimated.
Although much is already known about this cyclic hydrocarbon, research is still being done on cyclohexane and benzene mixtures and solid phase cyclohexane to determine hydrogen yields of the mix when irradiated at −195 °C.
History
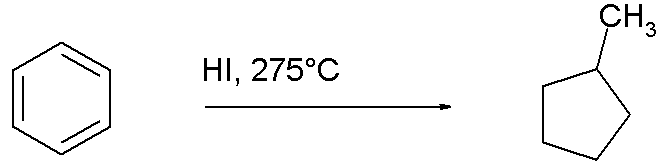
Unlike compounds like benzene, cyclohexane cannot easily be obtained from natural resources such as coal. Towards the end of the nineteenth century early chemical investigators had to depend on organic synthesis. It took them 30 years to flesh out the details[1]. In 1867 Marcellin Berthelot reduced benzene with hydroiodic acid at elevated temperatures. He incorrectly identified the reaction product as n-hexane not only because of the convenient match in boiling point (69°C) but also because he did not believe benzene was a cyclic molecule (like his contemporary August Kekule) but rather some sort of association of acetylene. In 1870 one of his sceptics Adolf von Baeyer repeated the reaction and pronounced the same reaction product hexahydrobenzene and in 1890 Vladimir Markovnikov believed he was able to distill the same compound from Caucasus petroleum calling his concoction hexanaphtene
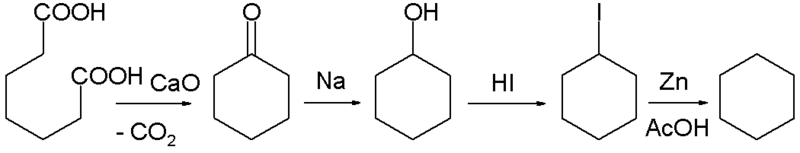
In 1894 Baeyer synthesized cyclohexane starting with a Dieckmann condensation of pimelic acid followed by multiple reductions:
and in the same year E. Haworth and W.H. Perkin Jr. (1860 - 1929) did the same in a Wurtz reaction of 1,6-dibromohexane.
Surprisingly their cyclohexanes boiled higher by 10°C than either hexahydrobenzene or hexanaphtene but this riddle was solved in 1895 by Markovnikov, N.M. Kishner and Nikolay Zelinsky when they re-diagnosed hexahydrobenzene and hexanaphtene as methylcyclopentane, the result of an unexpected rearrangement reaction.
See also
- The Flixborough disaster, a major industrial accident caused by an explosion of cyclohexane.
- Hexane
External links
- International Chemical Safety Card 0242
- National Pollutant Inventory - Cyclohexane fact sheet
- NIOSH Pocket Guide to Chemical Hazards
- Cyclohexane@3Dchem
- NLM Hazardous Substances Databank – Cyclohexane
References
- ↑ The curiously intertwined histories of benzene and cyclohexane E.W. Warnhoff J. Chem. Ed., 1996 494