Base excision repair
Base excision repair (BER) is a cellular mechanism that can repair damaged DNA during DNA replication. Repairing DNA sequence errors is necessary so that mutations are not induced during replication.
Single bases in DNA can be chemically mutated, for example by deamination or alkylation, resulting in incorrect base-pairing, and consequently, mutations in the DNA. Base excision repair involves flipping the mutated base out of the DNA helix and repairing the base alone. There are two main enzymes used, DNA glycosylases and AP endonucleases. The DNA glycosylase is used to break the β-N glycosidic bond to create an AP site. AP endonuclease recognizes this site and nicks the damaged DNA on the 5' side (upstream) of the AP site creating a free 3'-OH. DNA polymerase, Pol I, extends the DNA from the free 3'-OH using its exonuclease activity to replace the nucleotide of the damaged base, as well as a few downstream, followed by sealing of the new DNA strand by DNA ligase.
DNA glycosylases
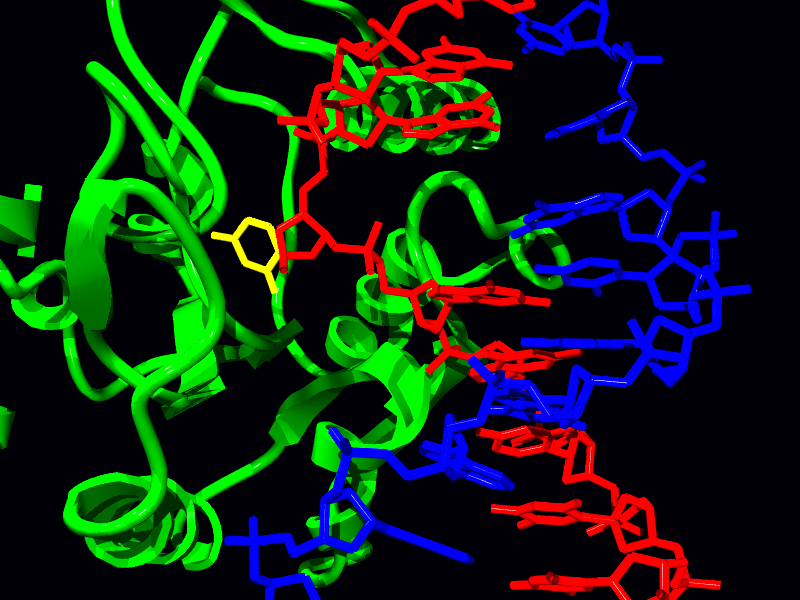
There are specific DNA glycosylases that recognize different damaged bases. For example, there are two oxoG glycosylases – the first removes oxoG that is bound to cytosine, repairing the actual damaged base, whereas the second removes an adenine bound to oxoG, removing the incorrect base before the next replication. A Uracil DNA glycosylase recognizes uracil in DNA causing its removal. Yet another glycosylase recognizes a thymine to guanine pairing, a common occurrence due to the deamination of 5-methyl cytosine, and removes the thymine to allow for its replacement with cytosine. There is also an alkyladenine DNA glycosylase that recognizes the mutated 3-methyl adenine, 7-methyl guanine, and hypoxanthine.
DNA glycosylases remove DNA bases that are cytotoxic or cause DNA polymerase errors and are part of the base excision repair pathway for DNA. DNA glycosylases cleave the N-glycosydic bond between the target base and deoxyribose. Excision of the damaged base leaves behind an apurinic/apyrimidinic (AP) site in DNA which cannot be copied by DNA polymerases. Several DNA glycosylases are bifunctional; i.e. they also display a lyase activity responsible for cleaving the phosphodiester backbone 3´ to the AP site generated by the glycosylase activity. They are called DNA glycosylase/AP lyases. Base excision repair pathway is initiated by a DNA glycosylase with the recognition of either a specific type of damaged DNA structure or an inappropriate base, such as uracil. The resulting one nucleotide gap is filled by a DNA polymerase and repair is completed by a DNA ligase that seals the remaining nick. Human cells are known to contain following DNA glycosylases:
Uracil DNA glycosylases: Uracil can arise in DNA either by spontaneous deamination of cytosine or by the misincorporation of dU opposite to a dA by a DNA polymerase during DNA synthesis. At least four different uracil-DNA glycosylase activities have been identified in mammalian cells, removing uracil and generating an abasic site. These enzymes are UNG, SMUG1, TDG and MBD4 and they vary in substrate specificity and localization in the cell. SMUG1 prefers single-stranded DNA as substrate, but also removes U from double-stranded DNA. TDG and MBD4 are strictly specific for ds DNA. Current evidence suggests that UDG and SMUG1 are the major enzymes responsible for the repair of the U:G mispairs caused by spontaneous cytosine deamination, whereas uracil arising in DNA through dU misincorporation is mainly dealt with by UNG. In contrast, MBD4 appear to have a more specialized function in correcting T:G mismatches that arise from deamination of 5-methylC to thymine in CpG sites. The structure of human UDG in complex with DNA revealed that it binds uracil by flipping the target nucleotide out of the double helix and into the active-site pocket.[1] UDG undergoes conformational change from an ‘‘open’’ unbound state to a ‘‘closed’’ DNA-bound state.[2] MBD4 (methyl binding domain 4 protein) is a DNA glycosylase that removes thymine from T:G pairs at methyl-CpG sites. It is within the HhH-GPD superfamily. Mbd4 was identified in a yeast two-hybrid screen due to its interaction with the human mismatch repair protein MLH1. The mechanism for recognition of thymine, a normal base in DNA, is based on a specific combination of hydrogen bonds and van der Waals forces.[3] MBD4 mutant mice develop normally and do not show increased cancer susceptibility or reduced survival. But they acquire more C T mutations at CpG sequences in epithelial cells of the small intestine.[4]
SMUG1 (single-strand selective monofunctional uracil-DNA glycosylase) is able to excise uracil from both single- and double-stranded in both G:U and A:U base-pairs, but show no activity against G:T mismatches.[5] Studies of UDG-knockout mice suggest that SMUG1 functions as a backup enzyme for UDG. SMUG1 also excises 5-hydroxyuracil, 5-hydroxymethyluracil and 5-formyluracil bearing an oxidized group at ring C5.[6]
TDG (thymine DNA glycosylase) repairs G:T mispair resulted form the deamination of 5-methylcytosine. TDG can also remove thymine glycol when present opposite guanine. Derivatives of U with modifications at the 5-carbon position such as 5-hydroxy-U, 5-hydroxymethyl-U, 5-fluoro- U (5-FU) and 5-bromo-U (5-BrU) are also efficiently processed. A proposed model for the binding of TDG to the DNA is that the N-terminal domain of TDG forms a flexible “clamp” that holds the enzyme to the DNA and then TDG slides along the DNA to find a mismatch.
Reactive oxygen species (ROS) are a major source of endogenous DNA damage. The most abundant lesions formed in guanine residues are 2,6-diamino-4-hydroxy-5-formamidopyrimidine (FapyG) and 8-oxoguanine. Due to mispairing with adenine during replication, 8-oxoG is highly mutagenic, resulting in G to T transversions. Repair of this lesion is initiated by the bifunctional DNA glycosylase OGG1 that recognizes 8-oxoG paired with C. hOGG1 belong to the helix-hairpin-helix (HhH) family of DNA glycosylases. For the initial recognition of the damaged base, DNA is bent at the damage site followed by a flipping of the base into the active site pockets of the enzyme. Studies with OGG1 knockout in mice show no age-dependent accumulation of 8-oxoG in spleen, spermatocytes, or renal cells but accumulated in the liver. Also OGG1 null mice do not show an increased tumor incidence.[7] The same is observed with the inactivation of MYH (Mut Y homolog DNA glycosylase) responsible for removing A opposite to 8-oxoG. But inactivation of both MYH and OGG1, accumulated 8-oxoG in the DNA of lung and small intestine.[8] This is probably because mammalian cells contain three backup DNA glycosylases, NEIL1, NEIL2 and NEIL3.
| Stub icon | This biochemistry article is a stub. You can help Wikipedia by expanding it. |
References
- ↑ Mol CD, Arvai AS, Slupphaug G, Kavli B, Alseth I, Krokan HE, Tainer JA. (1995). Crystal structure and mutational analysis of human uracil-DNA glycosylase. Cell 80(6):869-878.
- ↑ Slupphaug G, Mol CD, Kavli B, Arvai AS, Krokan HE, Tainer JA. (1996). A nucleotide-flipping mechanism from the structure of human uracil–DNA glycosylase bound to DNA. 384: 87-92.
- ↑ Wu P, Qiu C, Sohail A, Zhang X, Bhagwat, AS, Xiaodong C. (2003). Mismatch Repair in Methylated DNA. STRUCTURE AND ACTIVITY OF THE MISMATCH-SPECIFIC THYMINE GLYCOSYLASE DOMAIN OF METHYL-CpG-BINDING PROTEIN MBD4. 5285-5291.
- ↑ Wong E, Yang K, Kuraguchi M, Werling U, Avdievich E, Fan K, Fazzari M, Jin B, Brown, M.C, Lipkin M, Edelmann W. (1995). Mbd4 inactivation increases C→T transition mutations and promotes gastrointestinal tumor formation. PNAS 99(23): 14937-14942.
- ↑ Pearl L.H. (2000). Structure and function in the uracil-DNA glycosylase superfamily. Mutation Research 460 (3-4) 165-181.
- ↑ Matsubara M, Tanaka T, Terato H, Ohmae E, Izumi S, Katayanagi K, Ide H. (2004). Mutational analysis of the damage-recognition and catalytic mechanism of human SMUG1 DNA glycosylase. Nucleic Acids Res. 2004; 32(17): 5291–5302.
- ↑ Klungland A, Rosewell I, Hollenbach S, Larsen E, Daly G, Epe A, Seeberg E, Lindahl T, Barnes D. E. (1999). Accumulation of premutagenic DNA lesions in mice defective in removal of oxidative base damage. PNAS 96(23): 13300–13305.
- ↑ Russo M.T, De luca G, Degan P, Parlanti E, Dogliotti E Barnes D.E, Lindahl T, Yang H, Miller J. H, Bignami M. (2004). Accumulation of the Oxidative Base Lesion 8-Hydroxyguanine in DNA of Tumor-Prone Mice Defective in Both the Myh and Ogg1 DNA Glycosylases. Cancer Res 64(13): 4411-4414.
External links
- Base+Excision+Repair at the US National Library of Medicine Medical Subject Headings (MeSH)